chapter10
... There are three characteristics of the dipole radiation field that are important for us to consider: 1. The power radiated into a small element of area, in a direction at an angle θ with respect to the z axis, varies as sin2θ. This characteristic sin2θdependence of dipole radiation must also be a c ...
... There are three characteristics of the dipole radiation field that are important for us to consider: 1. The power radiated into a small element of area, in a direction at an angle θ with respect to the z axis, varies as sin2θ. This characteristic sin2θdependence of dipole radiation must also be a c ...
Blackbody radiation and ultra-violet catastrophe
... 1.3.1 What is blackbody? • Under thermal equilibrium, an object of a finite temperature emit radiation (supposed to be EM wave). • It is found that an object that absorbs more also emits more radiation. • A perfect absorber is an object with black surface that must of the incident energy is absorbe ...
... 1.3.1 What is blackbody? • Under thermal equilibrium, an object of a finite temperature emit radiation (supposed to be EM wave). • It is found that an object that absorbs more also emits more radiation. • A perfect absorber is an object with black surface that must of the incident energy is absorbe ...
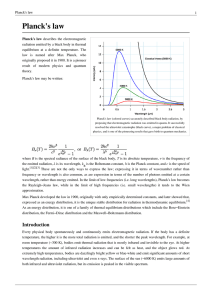
Planck`s law
... walls are not opaque, then the thermodynamic equilibrium is not isolated. It is of interest to explain how the thermodynamic equilibrium is attained. There are two main cases: (a) when the approach to thermodynamic equilibrium is in the presence of matter, when the walls of the cavity are imperfectl ...
... walls are not opaque, then the thermodynamic equilibrium is not isolated. It is of interest to explain how the thermodynamic equilibrium is attained. There are two main cases: (a) when the approach to thermodynamic equilibrium is in the presence of matter, when the walls of the cavity are imperfectl ...
Fluorescence
... by excitation of atoms in a material, which then reemit almost immediately (within about 10 8 seconds). The initial excitation is usually caused by absorption of energy from incident radiation or particles, such as X-rays or electrons. Because reemission occurs so quickly, the fluorescence ceases as ...
... by excitation of atoms in a material, which then reemit almost immediately (within about 10 8 seconds). The initial excitation is usually caused by absorption of energy from incident radiation or particles, such as X-rays or electrons. Because reemission occurs so quickly, the fluorescence ceases as ...
Document
... Assuming that the tungsten filament of a lightbulb is a blackbody, determine its peak wavelength if its temperature is 2 900 K. ...
... Assuming that the tungsten filament of a lightbulb is a blackbody, determine its peak wavelength if its temperature is 2 900 K. ...
N 2
... that the time it takes to decay from E4 to E3 is much longer than the time it takes to decay from E2 to E1. In a large population of such atoms, at equilibrium and with a continuous pumping process, a population inversion will occur between the E3 and E2 energy states, and a photon entering the popu ...
... that the time it takes to decay from E4 to E3 is much longer than the time it takes to decay from E2 to E1. In a large population of such atoms, at equilibrium and with a continuous pumping process, a population inversion will occur between the E3 and E2 energy states, and a photon entering the popu ...
original talk
... • After damping time: nonequilibrium KMS relation • Damping and prethermalization may coincide for heavy ion collisions, it gives about ¼ 0.6 fm/c • This can be an ingredient to understand the success of hydrodynamic description ...
... • After damping time: nonequilibrium KMS relation • Damping and prethermalization may coincide for heavy ion collisions, it gives about ¼ 0.6 fm/c • This can be an ingredient to understand the success of hydrodynamic description ...
Momentum Do photons carry momentum ? DeBroglie`s Relation
... or cutoff = c/ fcutoff , or cutoff = (3 x 108 m/s) / (1.09 x 1015 cycles/sec) = 276 nm (in the UV range) Any frequency lower than the cut-off (or any wavelength greater than the cut-off value) will NOT eject electrons from the metal. From Einstein’s equation: hf = W + e*Vstop , we can see that ...
... or cutoff = c/ fcutoff , or cutoff = (3 x 108 m/s) / (1.09 x 1015 cycles/sec) = 276 nm (in the UV range) Any frequency lower than the cut-off (or any wavelength greater than the cut-off value) will NOT eject electrons from the metal. From Einstein’s equation: hf = W + e*Vstop , we can see that ...
A Folded Fabry-Perot Cavity for Optical Sensing in DUAL Detector
... Measurements of thermo-elastic effects of coatings at room temperature We have performed two new series of photothermal measurements on a short cavity, changing the setup (in particular, the locking and measurement system) to verify the absence of systematic errors due to the measurement technique. ...
... Measurements of thermo-elastic effects of coatings at room temperature We have performed two new series of photothermal measurements on a short cavity, changing the setup (in particular, the locking and measurement system) to verify the absence of systematic errors due to the measurement technique. ...
9. Time-dependent Perturbation Theory
... Of course, there is a companion process in which the electron undergoes a transition from state b to state a, giving up energy Eb − Ea = ~ω0 in the process. We can calculate the probability for this process by assuming that ca(0) = 0, cb(0) = 1, and following the above derivation, yielding exactly ...
... Of course, there is a companion process in which the electron undergoes a transition from state b to state a, giving up energy Eb − Ea = ~ω0 in the process. We can calculate the probability for this process by assuming that ca(0) = 0, cb(0) = 1, and following the above derivation, yielding exactly ...
DEPARTMENT OF PHYSICS
... complete specification of the set of ME may be in general highly nontrivial for an arbitrary dielectric medium, one can expect the solution of electrodynamics problems involving dielectric medium can be tractable only for a few simple cases. We shall divide our study into the three quarter courses a ...
... complete specification of the set of ME may be in general highly nontrivial for an arbitrary dielectric medium, one can expect the solution of electrodynamics problems involving dielectric medium can be tractable only for a few simple cases. We shall divide our study into the three quarter courses a ...
Chapter 27
... and proton have the same momentum, they cannot have the same speed because of the difference in their masses. For the same reason, remembering that KE = p2/2m, they cannot have the same kinetic energy. Because the kinetic energy is the only type of energy an isolated particle can have, and we have a ...
... and proton have the same momentum, they cannot have the same speed because of the difference in their masses. For the same reason, remembering that KE = p2/2m, they cannot have the same kinetic energy. Because the kinetic energy is the only type of energy an isolated particle can have, and we have a ...
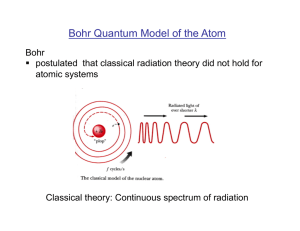
Bohr Quantum Model of the Atom
... § postulated that the electron orbital momentum is quantized Justification of Bohr’s postulates: comparison with experimental observations! ...
... § postulated that the electron orbital momentum is quantized Justification of Bohr’s postulates: comparison with experimental observations! ...
The Nature of Light - Solar Physics and Space Weather
... Kirchhoff’s Laws on Spectra • Law 1- Continuous spectrum: a hot opaque body, such as a perfect blackbody, produce a continuous spectrum – a complete rainbow of colors without any spectral line • Law 2 – emission line spectrum: a hot, transparent gas produces an emission line spectrum – a series of ...
... Kirchhoff’s Laws on Spectra • Law 1- Continuous spectrum: a hot opaque body, such as a perfect blackbody, produce a continuous spectrum – a complete rainbow of colors without any spectral line • Law 2 – emission line spectrum: a hot, transparent gas produces an emission line spectrum – a series of ...
N - INFN-LNF
... The the wave-function According to De Broglie: the wave nature of the electron is hard to figure out.... It is associated with the particles but it is not “concrete”. It’s just a mathematical description! The wave-function, Y (funcition of space and time), describes the quantum system (it is also c ...
... The the wave-function According to De Broglie: the wave nature of the electron is hard to figure out.... It is associated with the particles but it is not “concrete”. It’s just a mathematical description! The wave-function, Y (funcition of space and time), describes the quantum system (it is also c ...
Properties of blackbody radiation
... Figure shows approximate (normalized) emissions curves for the sun (blue) and the earth (red). Note that there is only about 1% overlap between the two curves. Normalized means each curve has been divided by its maximum value so both peak at 1. For the sun, T~6000 K, so from Wien’s law, m 0.5 m. ...
... Figure shows approximate (normalized) emissions curves for the sun (blue) and the earth (red). Note that there is only about 1% overlap between the two curves. Normalized means each curve has been divided by its maximum value so both peak at 1. For the sun, T~6000 K, so from Wien’s law, m 0.5 m. ...
Introduction, Introduction to lasers, Properties of light
... inside bunch of atoms oscillating inside a box with highly reflecting mirrors What do Maxwell's equations tell us about the radiation ? ...
... inside bunch of atoms oscillating inside a box with highly reflecting mirrors What do Maxwell's equations tell us about the radiation ? ...
QOLECTURE1
... inside bunch of atoms oscillating inside a box with highly reflecting mirrors What do Maxwell's equations tell us about the radiation ? ...
... inside bunch of atoms oscillating inside a box with highly reflecting mirrors What do Maxwell's equations tell us about the radiation ? ...
Radioactivity Revision_handout_20min
... F = m x a If the force is weight then use W instead and g for gravitational field strength instead of Acc (which is always 10 N/kg) Weight (N) = Mass (kg) x Gravitation field strength (N/kg) ...
... F = m x a If the force is weight then use W instead and g for gravitational field strength instead of Acc (which is always 10 N/kg) Weight (N) = Mass (kg) x Gravitation field strength (N/kg) ...
Environmental Sensors Photosynthetic Photon Flux Sensor
... quantify potential for plant photosynthesis by measuring active radiation in the wavelength ranges strongly correlated with plant growth. The sensor is calibrated for use in sunlight, and an innovative blue lens improves the accuracy of measurements. The pigments in the lens filter the incoming ligh ...
... quantify potential for plant photosynthesis by measuring active radiation in the wavelength ranges strongly correlated with plant growth. The sensor is calibrated for use in sunlight, and an innovative blue lens improves the accuracy of measurements. The pigments in the lens filter the incoming ligh ...
AP Quantum physics
... Example 3: In an experiment to determine Planck’s constant, a plot of stopping potential versus frequency is made. The slope of the curve is 4.13 x 10-15 V/Hz. What is Planck’s constant? ...
... Example 3: In an experiment to determine Planck’s constant, a plot of stopping potential versus frequency is made. The slope of the curve is 4.13 x 10-15 V/Hz. What is Planck’s constant? ...
Introduction to Quantum Mechanic
... after receiving energy goes back to the most stable state (ground state). The energy associated with the radiation is the difference in energy between these 2 states. When T increases, the average E*Mean is higher and intensity increases. E*Mean- E = kT. k is Boltzmann constant (k= 1.38 10-23 Joules ...
... after receiving energy goes back to the most stable state (ground state). The energy associated with the radiation is the difference in energy between these 2 states. When T increases, the average E*Mean is higher and intensity increases. E*Mean- E = kT. k is Boltzmann constant (k= 1.38 10-23 Joules ...
File - Mr. C at Hamilton
... •The existence of a smallest unit of light energy is one of the foundations of quantum mechanics. •The symbol (h) is used to denote Planck’s constant, which he discovered in 1899. It is used as a proportionality constant between the energy and frequency of an electromagnetic wave. (NOTE: We use it t ...
... •The existence of a smallest unit of light energy is one of the foundations of quantum mechanics. •The symbol (h) is used to denote Planck’s constant, which he discovered in 1899. It is used as a proportionality constant between the energy and frequency of an electromagnetic wave. (NOTE: We use it t ...
Schrödinger Equation
... The Schrödinger equation plays the role of Newton's laws and conservation of energy in classical mechanics - i.e., it predicts the future behavior of a dynamic system. It is a wave equation in terms of the wavefunction which predicts analytically and precisely the probability of events or outcome. T ...
... The Schrödinger equation plays the role of Newton's laws and conservation of energy in classical mechanics - i.e., it predicts the future behavior of a dynamic system. It is a wave equation in terms of the wavefunction which predicts analytically and precisely the probability of events or outcome. T ...
Quantum Nonlinear Resonances in Atom Optics
... Short wave-length modes: Phononic waves type of excitations (solved using Dyson's theory for oscillators chains with random springs) G. M. and Sh. Fishman, PRL 2004; PRE 2004. ...
... Short wave-length modes: Phononic waves type of excitations (solved using Dyson's theory for oscillators chains with random springs) G. M. and Sh. Fishman, PRL 2004; PRE 2004. ...