Fundamentals of Blackbody Radiation
... to Wien’s formula for high frequencies (i.e. hν/kB T 1) and to Rayleigh-Jeans formula for low frequencies (i.e. hν/kB T 1). An example of Planck’s radiation formula is shown in Fig. 3 along with Rayleigh-Jeans and Wien’s approximations for a blackbody of absolute temperature T = 6000◦ K. ...
... to Wien’s formula for high frequencies (i.e. hν/kB T 1) and to Rayleigh-Jeans formula for low frequencies (i.e. hν/kB T 1). An example of Planck’s radiation formula is shown in Fig. 3 along with Rayleigh-Jeans and Wien’s approximations for a blackbody of absolute temperature T = 6000◦ K. ...
radiation in medical imaging
... Ionising radiation is the radiation relating to diagnostic imaging, excluding Ultrasound and MRI which do not emit radiation. There are 4 types of ionising radiation: alpha, beta, Gamma and X-ray. Ionising radiation does not build up in your body.6 ...
... Ionising radiation is the radiation relating to diagnostic imaging, excluding Ultrasound and MRI which do not emit radiation. There are 4 types of ionising radiation: alpha, beta, Gamma and X-ray. Ionising radiation does not build up in your body.6 ...
Powerpoint handout on microclimate
... • Temperature of object being measured • Speed of measurement response ...
... • Temperature of object being measured • Speed of measurement response ...
Part III
... The attainment of a temperature of T = 0 K (“Absolute Zero”) is not possible. • The 3rd Law says that the entropy-temperature curves for a fixed external parameter, such as magnetic field, meet at T → 0. So, it is impossible to reach T = 0 in a finite number of steps. ...
... The attainment of a temperature of T = 0 K (“Absolute Zero”) is not possible. • The 3rd Law says that the entropy-temperature curves for a fixed external parameter, such as magnetic field, meet at T → 0. So, it is impossible to reach T = 0 in a finite number of steps. ...
The hydrogen line spectrum explained as Raman shift
... of the (shifted) frequencies is obviously expressed in the well known phenomenical Ritz-Rydberg formula for the hydrogen frequencies: ν = νH ( 1/n2 – 1/m2), for any n = 1, 2, 3,... , m = n +1 and goes from n + 1, n + 2, n + 3,... to ∞ . ν∞ is the limit frequency. For n = 1 we get the Lyman series, f ...
... of the (shifted) frequencies is obviously expressed in the well known phenomenical Ritz-Rydberg formula for the hydrogen frequencies: ν = νH ( 1/n2 – 1/m2), for any n = 1, 2, 3,... , m = n +1 and goes from n + 1, n + 2, n + 3,... to ∞ . ν∞ is the limit frequency. For n = 1 we get the Lyman series, f ...
Theoretical Physics T2 Quantum Mechanics
... quantized nature of energy. When Planck developed his formula for black body radiation he was forced to assume that the energy exchanged between a black body and it’s thermal (electromagnetic) radiation is not a continuous quantity but needs to be restricted to discrete values depending on the (angu ...
... quantized nature of energy. When Planck developed his formula for black body radiation he was forced to assume that the energy exchanged between a black body and it’s thermal (electromagnetic) radiation is not a continuous quantity but needs to be restricted to discrete values depending on the (angu ...
LAWS, RULES, PRINCIPLES, EFFECTS, PARADOXES, LIMITS,
... an energy density in empty space ofabout Cosmological redshift An effect where light emitted from a distant source appearsredshifted because of the expansion of space itself. Compare withthe Doppler effect. Coulomb's law The primary law for electrostatics, analogous to Newton's law ofuniversal gravi ...
... an energy density in empty space ofabout Cosmological redshift An effect where light emitted from a distant source appearsredshifted because of the expansion of space itself. Compare withthe Doppler effect. Coulomb's law The primary law for electrostatics, analogous to Newton's law ofuniversal gravi ...
Lecture 12
... A body may be in one of three states of static equilibrium: neutral, stable, and unstable: •Stable Equilibrium: A body is in stable equilibrium if it returns to its equilibrium position after it has been displaced slightly. •Unstable Equilibrium: A body is in unstable equilibrium if it does not ret ...
... A body may be in one of three states of static equilibrium: neutral, stable, and unstable: •Stable Equilibrium: A body is in stable equilibrium if it returns to its equilibrium position after it has been displaced slightly. •Unstable Equilibrium: A body is in unstable equilibrium if it does not ret ...
LAWS, RULES, PRINCIPLES, EFFECTS, PARADOXES, LIMITS,
... Waves emitted by a moving observer will be blueshifted(compressed) if approaching, redshifted (elongated) if receding.It occurs both in sound as well as electromagnetic phenomena,although it takes on different forms in each. Dulong-Petit law (P. Dulong, A.T. Petit; 1819) The molar heat capacity is a ...
... Waves emitted by a moving observer will be blueshifted(compressed) if approaching, redshifted (elongated) if receding.It occurs both in sound as well as electromagnetic phenomena,although it takes on different forms in each. Dulong-Petit law (P. Dulong, A.T. Petit; 1819) The molar heat capacity is a ...
Evidence for Photons.wxp
... Although Newton had first postulated that light was corpuscular in nature (i.e., that light consisted of small 'particles'), Young's double slit experiment had firmly established that light was a wave: it clearly exhibited interference phenomena. By the the early 1900's, the mathematical models used ...
... Although Newton had first postulated that light was corpuscular in nature (i.e., that light consisted of small 'particles'), Young's double slit experiment had firmly established that light was a wave: it clearly exhibited interference phenomena. By the the early 1900's, the mathematical models used ...
Spontaneous and Stimulated Transitions
... so the population of level i falls exponentially with time as electrons leave by spontaneous emission. The time in which the population falls to 1/e of its initial value is called the natural lifetime of level i, τi , where τi = 1/Ai . The magnitude of this lifetime is determined by the actual proba ...
... so the population of level i falls exponentially with time as electrons leave by spontaneous emission. The time in which the population falls to 1/e of its initial value is called the natural lifetime of level i, τi , where τi = 1/Ai . The magnitude of this lifetime is determined by the actual proba ...
Chapter 7: ELECTRONS IN ATOMS AND
... threshold frequency (ν0) • When ν<ν0 no electrons are emitted • When ν>ν0 electrons are emitted, more e– emitted with greater intensity of light ν < ν0 ...
... threshold frequency (ν0) • When ν<ν0 no electrons are emitted • When ν>ν0 electrons are emitted, more e– emitted with greater intensity of light ν < ν0 ...
5-1-light-quantized-energy
... O A piece of iron appears dark grey at room temperature, glows red when heated sufficiently, and turns orange, then bluish in color at even higher temperatures. ...
... O A piece of iron appears dark grey at room temperature, glows red when heated sufficiently, and turns orange, then bluish in color at even higher temperatures. ...
Hot gases: The transition from the line spectra to
... different energy level separations, different masses, and different total number of atoms lead to the same universal Planck black-body radiation energy distribution, even if the range of photon frequency for the separate processes overlap. Moreover, due to the finite lifetime of the excited levels o ...
... different energy level separations, different masses, and different total number of atoms lead to the same universal Planck black-body radiation energy distribution, even if the range of photon frequency for the separate processes overlap. Moreover, due to the finite lifetime of the excited levels o ...
P5.5.2.4 The Wien`s displacement law
... law: λmax = 967 nm. This deviation is caused by spectrometer sensitivity (grey curve), which is not constant for all wavelengths. If one determines spectrometer sensitivity on the basis of filament temperature then the measured spectra can be corrected by this factor. Measured spectrometer sensitivi ...
... law: λmax = 967 nm. This deviation is caused by spectrometer sensitivity (grey curve), which is not constant for all wavelengths. If one determines spectrometer sensitivity on the basis of filament temperature then the measured spectra can be corrected by this factor. Measured spectrometer sensitivi ...
Atomic quantum and nuclear
... and proton have the same momentum, they cannot have the same speed because of the difference in their masses. For the same reason, remembering that KE = p2/2m, they cannot have the same kinetic energy. Because the kinetic energy is the only type of energy an isolated particle can have, and we have a ...
... and proton have the same momentum, they cannot have the same speed because of the difference in their masses. For the same reason, remembering that KE = p2/2m, they cannot have the same kinetic energy. Because the kinetic energy is the only type of energy an isolated particle can have, and we have a ...
Planck`s radiation law, the light quantum, and the prehistory of
... does not depend on the position with which one sides. The reason is the following: While the question whether Planck himself implied a discontinuity is surely of historical interest it is of less importance in the teaching of physics. For physics education it is more relevant whether black-body radi ...
... does not depend on the position with which one sides. The reason is the following: While the question whether Planck himself implied a discontinuity is surely of historical interest it is of less importance in the teaching of physics. For physics education it is more relevant whether black-body radi ...
Lecture 23
... a single photon with energy E2-E1. The light has a frequency ω = (E2-E1)/ħ. For all this to happen, the system must be in the ψ2 state initially. 2. A photon is absorbed by a quantum system when it makes a transition from a quantum state of energy E1 to a state of energy E2, with E2>E1. The absorpti ...
... a single photon with energy E2-E1. The light has a frequency ω = (E2-E1)/ħ. For all this to happen, the system must be in the ψ2 state initially. 2. A photon is absorbed by a quantum system when it makes a transition from a quantum state of energy E1 to a state of energy E2, with E2>E1. The absorpti ...
Learning material
... is another constant of nature called Boltzmann’s constant. It has the units of energy per degree so we can think of it as a conversion factor from temperature to energy units. We’ll meet it again when we study thermodynamics. k =1.38x10-23 JK-1 ...
... is another constant of nature called Boltzmann’s constant. It has the units of energy per degree so we can think of it as a conversion factor from temperature to energy units. We’ll meet it again when we study thermodynamics. k =1.38x10-23 JK-1 ...
QPexam2012 - QMplus - Queen Mary University of London
... Show explicitly that the approximated formula valid at large λ obtained in part (ii) is incompatible with Stefan-Boltzmann’s law. [7 marks] (iv) Assume, as Planck did, that the energies of the oscillators are quantised and distributed with Boltzmann’s probability distribution factor e−E/(kT ) , wher ...
... Show explicitly that the approximated formula valid at large λ obtained in part (ii) is incompatible with Stefan-Boltzmann’s law. [7 marks] (iv) Assume, as Planck did, that the energies of the oscillators are quantised and distributed with Boltzmann’s probability distribution factor e−E/(kT ) , wher ...
Chemistry Websites of key interest Electron Arrangements Aufbau
... contributions to science in the field of electromagnetism. One hertz simply means "one per second"; 100 Hz means "one hundred per second", and so on. The unit may be applied to any periodic event – for example, a clock might be said to tick ...
... contributions to science in the field of electromagnetism. One hertz simply means "one per second"; 100 Hz means "one hundred per second", and so on. The unit may be applied to any periodic event – for example, a clock might be said to tick ...
The Physics BIG Crossword
... 21. The type of wave where the amplitude is perpendicular to the direction of wave motion. 24. His experiment scattered electrons with x-ray photons causing the photon wavelength to shift. 25. His principle states every point on a wavefront can be treated as a new wave source. 26. Constant obtained ...
... 21. The type of wave where the amplitude is perpendicular to the direction of wave motion. 24. His experiment scattered electrons with x-ray photons causing the photon wavelength to shift. 25. His principle states every point on a wavefront can be treated as a new wave source. 26. Constant obtained ...
On an Improvement of the Planck radiation Energy Distribution
... It is obvious that the products of integer numbers by discrete energy levels produce discrete levels for the energy distribution. More relevant to the present paper is the derivation of the Planck radiation energy distribution, eq. (13.20), which is carried out by A&F in section 13.6, that they call ...
... It is obvious that the products of integer numbers by discrete energy levels produce discrete levels for the energy distribution. More relevant to the present paper is the derivation of the Planck radiation energy distribution, eq. (13.20), which is carried out by A&F in section 13.6, that they call ...
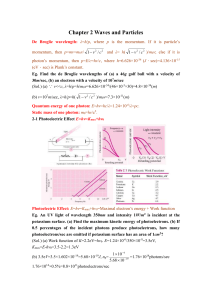
Chapter 2 Waves and Particles De Broglie wavelength: λ=h/p, where
... Eg. Blue light of wavelength 300nm and intensity 0.5W/m2 is directed at a material with work function of 2.7eV. (1) Find the maximum kinetic energy of photoelectrons. (2) If the quantum efficiency is 1%, how many photoelectrons are emitted per second if the semiconductor surface has an area of 2cm2 ...
... Eg. Blue light of wavelength 300nm and intensity 0.5W/m2 is directed at a material with work function of 2.7eV. (1) Find the maximum kinetic energy of photoelectrons. (2) If the quantum efficiency is 1%, how many photoelectrons are emitted per second if the semiconductor surface has an area of 2cm2 ...
Scientific Evaluation of Aulterra`s cell phone neutralizer
... superimposed fields. The first report of such an interaction demonstrated that a secondary crossbeam of visible light could neutralize the biological effects (induction of glutamate-pyruvate-transaminase activity by pretreatment of the substrate alanine) produced by a primary beam of visible light ...
... superimposed fields. The first report of such an interaction demonstrated that a secondary crossbeam of visible light could neutralize the biological effects (induction of glutamate-pyruvate-transaminase activity by pretreatment of the substrate alanine) produced by a primary beam of visible light ...