Energy and Energy Resources
... energy into the form of energy needed to do the work. Not all energy conversions are useful for the work that you are trying to do – some is “wasted” energy ...
... energy into the form of energy needed to do the work. Not all energy conversions are useful for the work that you are trying to do – some is “wasted” energy ...
Chapter 11 Modern Atomic Theory
... – Can only be solved for simple systems, but approximated for others ...
... – Can only be solved for simple systems, but approximated for others ...
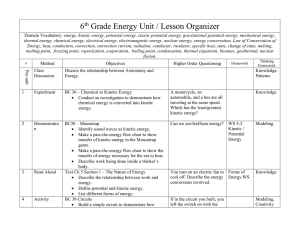
6th Grade Energy Unit / Lesson Organizer Domain Vocabulary
... Domain Vocabulary: energy, kinetic energy, potential energy, elastic potential energy, gravitational potential energy, mechanical energy, thermal energy, chemical energy, electrical energy, electromagnetic energy, nuclear energy, energy conservation, Law of Conservation of Energy, heat, conduction, ...
... Domain Vocabulary: energy, kinetic energy, potential energy, elastic potential energy, gravitational potential energy, mechanical energy, thermal energy, chemical energy, electrical energy, electromagnetic energy, nuclear energy, energy conservation, Law of Conservation of Energy, heat, conduction, ...
chapter 10: energy - Seattle Central College
... – To convert between calories and joules, we use the following: 1 cal ≡ 4.184 J ...
... – To convert between calories and joules, we use the following: 1 cal ≡ 4.184 J ...
The Nature of Heat
... • Mass is not the only factor that determines temperature change, what the substance is made of also affects its ability to absorb or lose heat – specific heat. – The number of calories needed to raise or lower the temperature of 1g of a substance 1°C. • Knowing the specific heat of a substance is u ...
... • Mass is not the only factor that determines temperature change, what the substance is made of also affects its ability to absorb or lose heat – specific heat. – The number of calories needed to raise or lower the temperature of 1g of a substance 1°C. • Knowing the specific heat of a substance is u ...
Mechanical Energy ME Example
... These perfect systems, although impossible to make, are called perpetual motion machines. This fictional machine transfers all PE into KE and back again never losing a drop of energy. This isn’t possible. Because some waste thermal energy (heat) always results when KE and PE transfer back and f ...
... These perfect systems, although impossible to make, are called perpetual motion machines. This fictional machine transfers all PE into KE and back again never losing a drop of energy. This isn’t possible. Because some waste thermal energy (heat) always results when KE and PE transfer back and f ...
Chapter 2 - Energy
... Objective • Students will define sources of energy and confirm whether or not those sources can be replaced once utilized. ...
... Objective • Students will define sources of energy and confirm whether or not those sources can be replaced once utilized. ...
Chapter 5: Work and Energy
... Work is done only when the force is parallel to the displacement. If the force is perpendicular to the displacement of an object, work is not done on the object. When the force on an object and the object’s displacement are in different directions, only the component of the force that is parallel to ...
... Work is done only when the force is parallel to the displacement. If the force is perpendicular to the displacement of an object, work is not done on the object. When the force on an object and the object’s displacement are in different directions, only the component of the force that is parallel to ...
NOTES SPS7 Energy Transformation
... Green plants convert the sun’s energy (electromagnetic) into starches and sugars (chemical energy) States of Energy The most common energy conversion is the conversion between potential and kinetic energy. All forms of energy can be in either of two states: Potential- energy of motion. Kin ...
... Green plants convert the sun’s energy (electromagnetic) into starches and sugars (chemical energy) States of Energy The most common energy conversion is the conversion between potential and kinetic energy. All forms of energy can be in either of two states: Potential- energy of motion. Kin ...
Thermochemistry
... constant, what is measured is really the change in internal energy, E, not H. For most reactions, the difference is very small. ...
... constant, what is measured is really the change in internal energy, E, not H. For most reactions, the difference is very small. ...
Let`s Convert Energy
... Energy is all around us, all of the time. It may, however, be known by different names depending on its source. Light, whether it comes from the sun or a light bulb, is radiant energy. Gravitational energy is the energy an object has due to its position above the ground. Food and fuel contain chemic ...
... Energy is all around us, all of the time. It may, however, be known by different names depending on its source. Light, whether it comes from the sun or a light bulb, is radiant energy. Gravitational energy is the energy an object has due to its position above the ground. Food and fuel contain chemic ...
Energy Conversions When energy is changed from one form to
... food down back into sugar. In our cells sugar is combined with oxygen to make water and carbon dioxide. The energy from “burning” sugar makes a chemical called ATP. ADP + P = ATP. ATP is the chemical which powers all living things. When your muscles move, they break apart ATP and convert the chemica ...
... food down back into sugar. In our cells sugar is combined with oxygen to make water and carbon dioxide. The energy from “burning” sugar makes a chemical called ATP. ADP + P = ATP. ATP is the chemical which powers all living things. When your muscles move, they break apart ATP and convert the chemica ...
Energy File
... Light – energy involving the production of visible light Elastic – energy involving a stretched object with the ability to return to its natural shape Electrical – energy involving the movement of electrons Chemical – energy involving chemical reactions Heat – energy involving the movement of the pa ...
... Light – energy involving the production of visible light Elastic – energy involving a stretched object with the ability to return to its natural shape Electrical – energy involving the movement of electrons Chemical – energy involving chemical reactions Heat – energy involving the movement of the pa ...
Name - SRHawks
... _position_____ and how _massive____ it is. 5. Energy has different _forms__________. II. Energy Transformations 1. One form of __energy__________ can be __transformed___into many other forms of energy. III. Law of Conservation of Energy 1. The law_______of conservation___ of energy states that the t ...
... _position_____ and how _massive____ it is. 5. Energy has different _forms__________. II. Energy Transformations 1. One form of __energy__________ can be __transformed___into many other forms of energy. III. Law of Conservation of Energy 1. The law_______of conservation___ of energy states that the t ...
15 Energy and Chemical Change
... 7. MAINIDEA Explain how energy changes from one form to another in an exothermic reaction. In an endothermic reaction. Chemical potential energy changes to heat in exothermic reactions, and the heat is released. In endothermic reactions, heat is absorbed and changed to chemical potential energy. ...
... 7. MAINIDEA Explain how energy changes from one form to another in an exothermic reaction. In an endothermic reaction. Chemical potential energy changes to heat in exothermic reactions, and the heat is released. In endothermic reactions, heat is absorbed and changed to chemical potential energy. ...
Energy
... Electrical energy is the movement of elections. Lightning and static electricity are examples of electrical energy that occur naturally. Science hasn't found a way to use natural forms of electrical energy, like lightning. Instead, we use different energy sources to create electrical energy by usin ...
... Electrical energy is the movement of elections. Lightning and static electricity are examples of electrical energy that occur naturally. Science hasn't found a way to use natural forms of electrical energy, like lightning. Instead, we use different energy sources to create electrical energy by usin ...
Kinetic and Potential Energy
... energy when we are rested or excited, and less energy when we are tired or bored. But that is only one kind of energy. Energy is working all around us. It powers cars and gives us light. Energy keeps us warm and creates sound. Without energy, we could not grow, move, or even stay alive! To understan ...
... energy when we are rested or excited, and less energy when we are tired or bored. But that is only one kind of energy. Energy is working all around us. It powers cars and gives us light. Energy keeps us warm and creates sound. Without energy, we could not grow, move, or even stay alive! To understan ...
What is energy?
... an object and Earth depends on the object’s weight and height. Elastic potential energy is energy stored in objects that are compressed or stretched. ...
... an object and Earth depends on the object’s weight and height. Elastic potential energy is energy stored in objects that are compressed or stretched. ...
Energy Test Study Guide
... 18. A cart is loaded with a brick and pulled at constant speed along an inclined plane to the height of a seat-top. If the mass o the loaded cart is 3.0 kg and the height of the seat top is 0.45 meters, then what is the potential energy of the loaded cart at t height of the seat-top? ...
... 18. A cart is loaded with a brick and pulled at constant speed along an inclined plane to the height of a seat-top. If the mass o the loaded cart is 3.0 kg and the height of the seat top is 0.45 meters, then what is the potential energy of the loaded cart at t height of the seat-top? ...
Introduction to Energy! - Epiphany Catholic School
... Is energy conserved? • A closed system is a group of objects that transfer energy only to one another. Energy is conserved in all closed systems. • The law of conservation of energy states that energy cannot be created or destroyed. It can only change forms. • All of the different forms of energy in ...
... Is energy conserved? • A closed system is a group of objects that transfer energy only to one another. Energy is conserved in all closed systems. • The law of conservation of energy states that energy cannot be created or destroyed. It can only change forms. • All of the different forms of energy in ...
What are Kinetic and Potential Energy?
... to the ball. As it flies over the net, the ball has kinetic energy. • Kinetic energy is the energy of motion. • All moving objects have kinetic energy. • Like all forms of energy, kinetic energy can be used to do work. For example, kinetic energy allows a hammer to do work on a nail. ...
... to the ball. As it flies over the net, the ball has kinetic energy. • Kinetic energy is the energy of motion. • All moving objects have kinetic energy. • Like all forms of energy, kinetic energy can be used to do work. For example, kinetic energy allows a hammer to do work on a nail. ...
Lewis Energy types
... Why are energy conversions important? • It is useful for every day life. – Ex. Wind used to cook a meal ...
... Why are energy conversions important? • It is useful for every day life. – Ex. Wind used to cook a meal ...
Matter and Energy - Empidonax-hommondii10-11
... in the fact that they are not very dense at all. That is the reason we can move our hands through the air with ...
... in the fact that they are not very dense at all. That is the reason we can move our hands through the air with ...
Negawatt power

Negawatt power is a theoretical unit of power representing an amount of energy (measured in watts) saved. The energy saved is a direct result of energy conservation or increased energy efficiency. The term was coined by the chief scientist of the Rocky Mountain Institute and environmentalist Amory Lovins in 1989, arguing that utility customers don’t want kilowatt-hours of electricity; they want energy services such as hot showers, cold beer, lit rooms, and spinning shafts, which can come more cheaply if electricity is used more efficiently. Lovins felt an international behavioral change was necessary in order to decrease countries' dependence on excessive amounts of energy. The concept of a negawatt could influence a behavioral change in consumers by encouraging them to think about the energy that they spend.A negawatt market can be thought of as a secondary market, in which electricity is allocated from one consumer to another consumer within the energy market. In this market, negawatts could be treated as a commodity. Commodities have the ability to be traded across time and space, which would allow negawatts to be incorporated in the international trading system. Roughly 10% of all U.S. electrical generating capacity is in place to meet the last 1% of demand and there is where the immediate efficiency opportunity exists.On March 15, 2011, the Federal Energy Regulatory Commission (FERC), the agency that regulates the U.S. electrical grid, approved a rule establishing the approach to compensation for demand response resources intended to benefit customers and help improve the operation and competitiveness of organized wholesale energy markets. This means that negawatts produced by reducing electrical use can demand the same market prices as real megawatts of generated electricity.The incentives for a negawatt market include receiving money, reduction of national energy dependency, and the local electricity deregulation within certain nations or states. As for the cost incentive, those who produce negawatts or simply conserve energy can earn money by selling the saved energy. The negawatt market could help nations or states obtain a deregulated electricity system by creating another market to purchase electricity from. The negawatt market also has two main drawbacks. Currently, there is no way to precisely measure the amount of energy saved in negawatts, and electricity providers may not want customers to use less energy due to the loss of profit.