Basic Concepts
... condition of a thermodynamic system. Regardless of the path by which a system goes from one state to another — i.e., the sequence of intermediate states — the total change in any state variable will be the same. This means that the incremental changes in such variables are ww exact differentials. w. ...
... condition of a thermodynamic system. Regardless of the path by which a system goes from one state to another — i.e., the sequence of intermediate states — the total change in any state variable will be the same. This means that the incremental changes in such variables are ww exact differentials. w. ...
Thermodynamics Chapter 4
... Chapter Summary • Taking heat transfer to the system and work done by the system to be positive quantities, the conservation of mass and energy equations for steady-flow processes are expressed as ...
... Chapter Summary • Taking heat transfer to the system and work done by the system to be positive quantities, the conservation of mass and energy equations for steady-flow processes are expressed as ...
Boundless Study Slides
... • internal energy The sum of all energy present in the system, including kinetic and potential energy; equivalently, the energy needed to create a system, excluding the energy necessary to displace its surroundings. • internal energy The sum of all energy present in the system, including kinetic and ...
... • internal energy The sum of all energy present in the system, including kinetic and potential energy; equivalently, the energy needed to create a system, excluding the energy necessary to displace its surroundings. • internal energy The sum of all energy present in the system, including kinetic and ...
Equilibrium Statistical Mechanics
... constraint the final entropy, after the re-establisment of equilibrium, must be greater or equal to the initial i are such entropy. The new values of Em , Vm , Nm that the entropy is increased or remains the same. In summary: the entropy of an isolated system cannot decrease. I ...
... constraint the final entropy, after the re-establisment of equilibrium, must be greater or equal to the initial i are such entropy. The new values of Em , Vm , Nm that the entropy is increased or remains the same. In summary: the entropy of an isolated system cannot decrease. I ...
ch06A-2013
... component such as the throttling valve considered here often does not have much significance by itself. The significance of the entropy production of any component is normally determined through comparison with the entropy production values of other components combined with that component to form an ...
... component such as the throttling valve considered here often does not have much significance by itself. The significance of the entropy production of any component is normally determined through comparison with the entropy production values of other components combined with that component to form an ...
ee11042602mpt3.mov 110426ph423main3.mov Example of the
... While watching the video, David noted that this is a measurement of the heat capacity of a gas at fixed volume, and would naturally be done with a bomb calorimeter, much as the students describe. He would have a heating element, and measure how much power it outputs in a given time interval (to mea ...
... While watching the video, David noted that this is a measurement of the heat capacity of a gas at fixed volume, and would naturally be done with a bomb calorimeter, much as the students describe. He would have a heating element, and measure how much power it outputs in a given time interval (to mea ...
Maximum Entropy Closure of Balance Equations for Miniband
... as a closure to produce improved results for transport coefficients; see [13] and references cited therein. Compared to fluids, charges in semiconductors feel the effect of strong external fields, that appear in Boltzmann transport equations and, in the short mean free path and strong field limits, ...
... as a closure to produce improved results for transport coefficients; see [13] and references cited therein. Compared to fluids, charges in semiconductors feel the effect of strong external fields, that appear in Boltzmann transport equations and, in the short mean free path and strong field limits, ...
Chemical Thermodynamics
... reactions-the extent to which they occur. As we noted in the introduction, our experience tells us that physical and chemical processes have a directional character. For instance, sodium metal and chlorine gas combine readily to form sodium chloride, which we also know as table salt. We never find t ...
... reactions-the extent to which they occur. As we noted in the introduction, our experience tells us that physical and chemical processes have a directional character. For instance, sodium metal and chlorine gas combine readily to form sodium chloride, which we also know as table salt. We never find t ...
IV. Adiabatic Processes
... To gain some insights into the nature of vertical mixing in the atmosphere it is useful to consider the behavior of an air parcel of infinitesimal dimensions that is assumed to be: • thermally insulated from its environment, so that its temperature changes adiabatically as it rises or sinks • alway ...
... To gain some insights into the nature of vertical mixing in the atmosphere it is useful to consider the behavior of an air parcel of infinitesimal dimensions that is assumed to be: • thermally insulated from its environment, so that its temperature changes adiabatically as it rises or sinks • alway ...
Ecosystems as Self-organizing Holarchic Open Systems
... are far from equilibrium systems that maintains their local organization, through entropy export, at the expense of the larger system they are part of. He proposed that the investigation of living systems from a nonequilibrium perspective would help reconcile biological self-organization and thermod ...
... are far from equilibrium systems that maintains their local organization, through entropy export, at the expense of the larger system they are part of. He proposed that the investigation of living systems from a nonequilibrium perspective would help reconcile biological self-organization and thermod ...
\bf {The First Law of Thermodynamics for Closed Systems}\\
... Work is energy expended when a force acts through a displacement. Boundary work occurs because the mass of the substance contained within the system boundary causes a force, the pressure times the surface area, to act on the boundary surface and make it move. This is what happens when steam, the “ga ...
... Work is energy expended when a force acts through a displacement. Boundary work occurs because the mass of the substance contained within the system boundary causes a force, the pressure times the surface area, to act on the boundary surface and make it move. This is what happens when steam, the “ga ...
calcijlation of elastic properties from thermodynamic equation of
... Granato 1975a,b), implying that (dKT/dT)pis constant [as well as (dp/aT)p]. I11 this sanle limit, 6, in nlany materials is observationally constant (Anderson et a1 1968, Anderson & Goto 1989, Isaak et a1 1989). Since aKTappears constant as well as (dK,/aT),, 6, should also be constant by (7). We thu ...
... Granato 1975a,b), implying that (dKT/dT)pis constant [as well as (dp/aT)p]. I11 this sanle limit, 6, in nlany materials is observationally constant (Anderson et a1 1968, Anderson & Goto 1989, Isaak et a1 1989). Since aKTappears constant as well as (dK,/aT),, 6, should also be constant by (7). We thu ...
Multi-Physics Interactions for Coupled Thermo-Electro
... Figure 2: Complete form of MPID for fully coupled linear, reversible relationships ...... 12 Figure 3: Panel Form of MPID for linear, reversible relationships .................................. 13 Figure 4: An example of the isolated subset for coupled interactions .............................. 14 ...
... Figure 2: Complete form of MPID for fully coupled linear, reversible relationships ...... 12 Figure 3: Panel Form of MPID for linear, reversible relationships .................................. 13 Figure 4: An example of the isolated subset for coupled interactions .............................. 14 ...
basic thermodynamics
... energy supplied. Energy is said to be degraded each time when it flows through a finite temperature difference. That’s why the second law is some times called the law of degradation of energy and the energy is said to run down hill. ...
... energy supplied. Energy is said to be degraded each time when it flows through a finite temperature difference. That’s why the second law is some times called the law of degradation of energy and the energy is said to run down hill. ...
The Fourth Law of Black Hole Thermodynamics
... In the work of Bardeen, Carter and Hawking [1] it was established a remarkable mathematical analogy between the laws of thermodynamics and the laws of black hole mechanics derived from General Relativity. If one makes the formal replacements E → M , T → Cκ, and S → A/8πC (where C is a constant) in t ...
... In the work of Bardeen, Carter and Hawking [1] it was established a remarkable mathematical analogy between the laws of thermodynamics and the laws of black hole mechanics derived from General Relativity. If one makes the formal replacements E → M , T → Cκ, and S → A/8πC (where C is a constant) in t ...
book - University of Guelph Physics
... Thermodynamics is the study of macroscopic systems for which thermal effects are important. These systems are normally assumed to be at equilibrium, or at least, close to equilibrium. Systems at equilibrium are easier to study, both experimentally and theoretically, because their physical properties ...
... Thermodynamics is the study of macroscopic systems for which thermal effects are important. These systems are normally assumed to be at equilibrium, or at least, close to equilibrium. Systems at equilibrium are easier to study, both experimentally and theoretically, because their physical properties ...
First Law of Thermodynamics – Basic Concepts
... (1) The temperature of the system must be uniform and must be the same as the temperature of the surroundings (thermal equilibrium). (2) The mechanical properties must be uniform throughout the system (mechanical equilibrium). That is, no mechanical work is done by one part of the system on any othe ...
... (1) The temperature of the system must be uniform and must be the same as the temperature of the surroundings (thermal equilibrium). (2) The mechanical properties must be uniform throughout the system (mechanical equilibrium). That is, no mechanical work is done by one part of the system on any othe ...
Electromagnetism 电磁学
... well-developed before the beginning of the 20th century—classical mechanics, acoustics, optics, thermodynamics, and electromagnetism. Classical mechanics is concerned with bodies acted on by forces and bodies in motion and may be divided into statics (study of the forces on a body or bodies at rest) ...
... well-developed before the beginning of the 20th century—classical mechanics, acoustics, optics, thermodynamics, and electromagnetism. Classical mechanics is concerned with bodies acted on by forces and bodies in motion and may be divided into statics (study of the forces on a body or bodies at rest) ...
Chapter One :- Concepts and Definitions
... 1-Extensive Property :- The properties of a system whose value for the entire system is equal to the sum of their value for the individual parts of the system , these properties are related to the mass of the system. Like (volume, mass, all kinds of energy ). 2- Intensive Property :- the properties ...
... 1-Extensive Property :- The properties of a system whose value for the entire system is equal to the sum of their value for the individual parts of the system , these properties are related to the mass of the system. Like (volume, mass, all kinds of energy ). 2- Intensive Property :- the properties ...
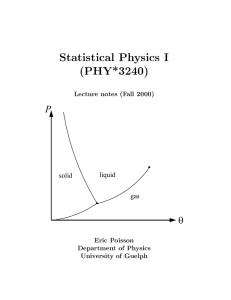
Phase changes
... possible paths to compute a vapour-region value from that reference point. Notice also that mean variables do not measure any local state but the weighted average according to the mass proportions of the phases, as seen in (6.6). When two-phase states of a system are represented in a thermodynamic d ...
... possible paths to compute a vapour-region value from that reference point. Notice also that mean variables do not measure any local state but the weighted average according to the mass proportions of the phases, as seen in (6.6). When two-phase states of a system are represented in a thermodynamic d ...
Thermodynamics and Thermochemistry for Engineers
... A variable describing a particular piece of matter is said to be extensive if its value depends on the quantity of the matter being described. For example, total heat capacity and mass are both extensive variables as opposed to intensive variables such as density, specific heat capacity, and tempera ...
... A variable describing a particular piece of matter is said to be extensive if its value depends on the quantity of the matter being described. For example, total heat capacity and mass are both extensive variables as opposed to intensive variables such as density, specific heat capacity, and tempera ...