1411FINALSAMPLEs and Key
... overall geometry is tetrahedral and the molecular geometry is trigonal pyramidal. The hybridization of the sulfur atom in the first structure is therefore sp3. However, the sulfur is not simply sp3 hybridized in the second structure, which has an “expanded octet” around the sulfur atom. Hybridizatio ...
... overall geometry is tetrahedral and the molecular geometry is trigonal pyramidal. The hybridization of the sulfur atom in the first structure is therefore sp3. However, the sulfur is not simply sp3 hybridized in the second structure, which has an “expanded octet” around the sulfur atom. Hybridizatio ...
Research on the Design of an Optical Information Storage Sensing
... An optical information sensor can be applied in many fields, such as optical storage and lightshows, as the quantities of the beam in the system can be detected, delivering macroscopic observation of the microscopic properties of a material [1]. Gold nanorods are one type of nanomaterial. They have ...
... An optical information sensor can be applied in many fields, such as optical storage and lightshows, as the quantities of the beam in the system can be detected, delivering macroscopic observation of the microscopic properties of a material [1]. Gold nanorods are one type of nanomaterial. They have ...
Quantum electrical transport in samples of limited
... The ability to make electrically conducting structures of ever smaller size by nanofabrication techniques ~the playground of mesoscopic physics! has brought with it entry into a wonderful new range of unexpected quantum phenomena. Interpretation of these phenomena requires full recognition of the wa ...
... The ability to make electrically conducting structures of ever smaller size by nanofabrication techniques ~the playground of mesoscopic physics! has brought with it entry into a wonderful new range of unexpected quantum phenomena. Interpretation of these phenomena requires full recognition of the wa ...
Inorganic and organic chemistry 2
... The substance that could carry out a reduction of vanadium must be on the right-hand side of the half-equations, so it cannot be iodine. Sulfur dioxide being oxidised to sulfate(VI) ions has a potential of −0.17 V. Adding this to each of the potentials for the vanadium half-equations gives +0.83 V ( ...
... The substance that could carry out a reduction of vanadium must be on the right-hand side of the half-equations, so it cannot be iodine. Sulfur dioxide being oxidised to sulfate(VI) ions has a potential of −0.17 V. Adding this to each of the potentials for the vanadium half-equations gives +0.83 V ( ...
Water-Mediated Dimerization of Ubiquitin Ions Captured by
... conserved in all eukaryotic cells in all species. Its function varies and is determined by a process called ubiquitination, in which the protein covalently attaches to a target protein, tagging it for a specific cellular process dependent on both the protein and the binding site.20,21 In aqueous solu ...
... conserved in all eukaryotic cells in all species. Its function varies and is determined by a process called ubiquitination, in which the protein covalently attaches to a target protein, tagging it for a specific cellular process dependent on both the protein and the binding site.20,21 In aqueous solu ...
3. Carbon nanostructures - Acclab h55.it.helsinki.fi
... - If you draw out the 4 bonds to equal length, then draw planes through the ends, you will get an ideal tetrahedron pyramid shape. The carbon bonds are very strong, typical energy/bond is of the order of 4 eV if they are to other carbon atoms. ...
... - If you draw out the 4 bonds to equal length, then draw planes through the ends, you will get an ideal tetrahedron pyramid shape. The carbon bonds are very strong, typical energy/bond is of the order of 4 eV if they are to other carbon atoms. ...
Fabrication and Application of Phase only Holograms for High
... leads to test sample destruction. Cooling of the samples with compressed air allowed them not to be destroyed at least for 60 seconds. 4. Beam shaping with binary phase only holograms Although the ability of ITO coatings to withstand high cw power levels is crucial for the envisioned applications it ...
... leads to test sample destruction. Cooling of the samples with compressed air allowed them not to be destroyed at least for 60 seconds. 4. Beam shaping with binary phase only holograms Although the ability of ITO coatings to withstand high cw power levels is crucial for the envisioned applications it ...
Ceramics for catalysis
... Once the reactant is bound to the surface, it can readily undergo reactions which take place only with difficulty in the gas or liquid phases. This may result from the close proximity of reactant molecules on the surface and/or the changes in bonding consequent upon chemisorption; both are essential ...
... Once the reactant is bound to the surface, it can readily undergo reactions which take place only with difficulty in the gas or liquid phases. This may result from the close proximity of reactant molecules on the surface and/or the changes in bonding consequent upon chemisorption; both are essential ...
Few-electron quantum dots
... dot, however, results in a considerable energy cost for adding an extra electron charge. Extra energy is therefore needed, and no current will flow until increasing the voltage provides this energy. This phenomenon is known as Coulomb blockade [10]. To see how this works in practice, we consider the ...
... dot, however, results in a considerable energy cost for adding an extra electron charge. Extra energy is therefore needed, and no current will flow until increasing the voltage provides this energy. This phenomenon is known as Coulomb blockade [10]. To see how this works in practice, we consider the ...
Fall 2012 Chem106 Final Review Name: Test 1 Materials Question
... a) Na2CO3(s) b) (NH4)3PO4(s) 3. (15pts) Which of the following reaction has a precipitate. Use the solubility rules and predict the products, if there is an insoluble product then write the total ionic equation and the net ionic equation for the reaction. a) KNO3(aq) + Na2SO4(aq) ...
... a) Na2CO3(s) b) (NH4)3PO4(s) 3. (15pts) Which of the following reaction has a precipitate. Use the solubility rules and predict the products, if there is an insoluble product then write the total ionic equation and the net ionic equation for the reaction. a) KNO3(aq) + Na2SO4(aq) ...
Chapter 5HW_Ans
... b) conversion factor: x X moles of C2H2; therefore 7 moles of O2 2molesC 2 H 2 as moles of C2H2 cancel as they are in both numerator and denominator 2molesC 2 H 2 c) conversion factor: x X moles of H2O; therefore 0.5 moles of 2molesH 2 O C2H2 as moles of H2O cancel as they are in both numerator and ...
... b) conversion factor: x X moles of C2H2; therefore 7 moles of O2 2molesC 2 H 2 as moles of C2H2 cancel as they are in both numerator and denominator 2molesC 2 H 2 c) conversion factor: x X moles of H2O; therefore 0.5 moles of 2molesH 2 O C2H2 as moles of H2O cancel as they are in both numerator and ...
Two-dimensional C/BN core/shell structures
... higher potential barrier than when it was surrounded by BN structure. Buffering BN layer reduces the confinement strength, thereby reducing the energy gap. However this reduction in energy gap is low, compared to that when r1 parameter of CS structure is increased. This again shows that the band edg ...
... higher potential barrier than when it was surrounded by BN structure. Buffering BN layer reduces the confinement strength, thereby reducing the energy gap. However this reduction in energy gap is low, compared to that when r1 parameter of CS structure is increased. This again shows that the band edg ...
LATTICE IMAGING IN TRANSMISSION ELECTRON MICROSCOPY
... The last decades are characterized by an evolution from macro- to micro- and more recently to nanotechnology. Examples are numerous, such as nanoparticles, nanotubes, quantum transistors, layered superconducting and magnetic materials, etc. Since many material properties are strongly connected to th ...
... The last decades are characterized by an evolution from macro- to micro- and more recently to nanotechnology. Examples are numerous, such as nanoparticles, nanotubes, quantum transistors, layered superconducting and magnetic materials, etc. Since many material properties are strongly connected to th ...
O usually has oxidation number of -2, except in peroxides where it is
... Give a little, alkali metals choose +1 (by giving up one electron). Hydrogen is above it all ...
... Give a little, alkali metals choose +1 (by giving up one electron). Hydrogen is above it all ...
Document
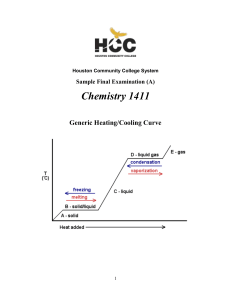
... during a process • if the final condition has a larger amount of internal energy than the initial condition, the change in the internal energy will be + • if the final condition has a smaller amount of internal energy than the initial condition, the change in the internal energy will be ─ ...
... during a process • if the final condition has a larger amount of internal energy than the initial condition, the change in the internal energy will be + • if the final condition has a smaller amount of internal energy than the initial condition, the change in the internal energy will be ─ ...
Experimental - AIP FTP Server
... both surfaces; the results favour the conclusion that the dissociation occurs on the S0 surface. This conclusion is further supported by comparison of the calculated and previously measured CO product vibrational quantum state distributions arising from photodissociation at 193.3 nm. The variational ...
... both surfaces; the results favour the conclusion that the dissociation occurs on the S0 surface. This conclusion is further supported by comparison of the calculated and previously measured CO product vibrational quantum state distributions arising from photodissociation at 193.3 nm. The variational ...
File - Mr Weng`s IB Chemistry
... • The mole is a fixed number of particles and refers to the amount, n, of substance. • Masses of atoms are compared on a scale relative to 12C and are expressed as relative atomic mass (Ar) and relative formula/molecular mass (Mr). • Molar mass (M) has the units g mol-1. • The empirical formula and ...
... • The mole is a fixed number of particles and refers to the amount, n, of substance. • Masses of atoms are compared on a scale relative to 12C and are expressed as relative atomic mass (Ar) and relative formula/molecular mass (Mr). • Molar mass (M) has the units g mol-1. • The empirical formula and ...
Theory of Ion Exchange
... (eqn [16]), the rate of exchange increases (t1/2 decreases) as the particle radius decreases, being proportional to 1/r20. For Rlm diffusion (eqn [17]), the rate increases less strongly as r0 is decreased (the proportionality is to 1/r0). In Rlm diffusion the exchange rate can also be increased by i ...
... (eqn [16]), the rate of exchange increases (t1/2 decreases) as the particle radius decreases, being proportional to 1/r20. For Rlm diffusion (eqn [17]), the rate increases less strongly as r0 is decreased (the proportionality is to 1/r0). In Rlm diffusion the exchange rate can also be increased by i ...
Arenes - Science Skool!
... arranged in a hexagon, and he suggested alternating double and single bonds between them. Each carbon atom has a hydrogen attached to it. Although the Kekulé structure was a good attempt in its time, there are serious problems with it. Problems with the chemistry Because of the three double bonds, y ...
... arranged in a hexagon, and he suggested alternating double and single bonds between them. Each carbon atom has a hydrogen attached to it. Although the Kekulé structure was a good attempt in its time, there are serious problems with it. Problems with the chemistry Because of the three double bonds, y ...
VI. ELECTRONIC SPECTROSCOPY The visible (V) and ultraviolet
... same light arrive to the eye by different routes, either exactly or very nearly in the same direction, the light becomes most intense when the difference of the two routes is any multiple of a certain length, and least intense in the intermediate state of the interfering portions; and this length is ...
... same light arrive to the eye by different routes, either exactly or very nearly in the same direction, the light becomes most intense when the difference of the two routes is any multiple of a certain length, and least intense in the intermediate state of the interfering portions; and this length is ...
1)A neutral atom has no overall charge, and ion is a
... c)The valence electrons experience a smaller nuclear force of attraction due to i)the increased distance of the electron from the nucleus(electrons are in a higher energy level), and ii)the shielding effect is more due to the number of filled orbitals in the core before the valence electron is reach ...
... c)The valence electrons experience a smaller nuclear force of attraction due to i)the increased distance of the electron from the nucleus(electrons are in a higher energy level), and ii)the shielding effect is more due to the number of filled orbitals in the core before the valence electron is reach ...
Helium-Neon Laser 1 Introduction
... HeNe and Argon lasers typically have very small bore diameters. It is therefore difficult to send an alignment laser beam through these tubes. In addition, the laser manufacturer makes your life most difficult by locating the external cavity mirrors so close to the tube, that there is no way to inse ...
... HeNe and Argon lasers typically have very small bore diameters. It is therefore difficult to send an alignment laser beam through these tubes. In addition, the laser manufacturer makes your life most difficult by locating the external cavity mirrors so close to the tube, that there is no way to inse ...
Chirality in amino acid over layers at metal crystal surfaces
... A face-centred cubic material has high symmetry, but a surface of such a crystal may or may not be mirror-symmetric, depending on surface orientation. A surface cut to an orientation that has no mirror symmetry is an example of an intrinsically chiral surface. As with all chiral objects, such surfac ...
... A face-centred cubic material has high symmetry, but a surface of such a crystal may or may not be mirror-symmetric, depending on surface orientation. A surface cut to an orientation that has no mirror symmetry is an example of an intrinsically chiral surface. As with all chiral objects, such surfac ...
Rutherford backscattering spectrometry
Rutherford backscattering spectrometry (RBS) is an analytical technique used in materials science. Sometimes referred to as high-energy ion scattering (HEIS) spectrometry, RBS is used to determine the structure and composition of materials by measuring the backscattering of a beam of high energy ions (typically protons or alpha particles) impinging on a sample.