Calculations with Chemical Formulas and Equations
... sense since propane is the material that you must purchase in order to cook your food. b. Since the chemical reaction only requires propane and oxygen, if the grill will not light with ample propane present, then the limiting reactant must be oxygen. c. Once again, here is a case where you have adeq ...
... sense since propane is the material that you must purchase in order to cook your food. b. Since the chemical reaction only requires propane and oxygen, if the grill will not light with ample propane present, then the limiting reactant must be oxygen. c. Once again, here is a case where you have adeq ...
AP Chemistry - Jackson County School System
... 82. Calculate the molarity of a solution that contains 0.0345 mol NH4Cl in exactly 400 ml of solution? 83. Calculate the molarity of a solution that contains 20.0grams of sodium hydroxide in 200ml? 84. How many grams of solute are present in 50.0 ml of 0.360 M sodium chloride? 85. The compound adren ...
... 82. Calculate the molarity of a solution that contains 0.0345 mol NH4Cl in exactly 400 ml of solution? 83. Calculate the molarity of a solution that contains 20.0grams of sodium hydroxide in 200ml? 84. How many grams of solute are present in 50.0 ml of 0.360 M sodium chloride? 85. The compound adren ...
Chemistry 212 Name:
... Na2S(aq) + 2 HCl(aq) → 2 NaCl(aq) + H2S(g) 5. Discuss the halogens. (5 points) Each halogen is obtained by oxidation of the halide ion to the halogen in a molten salt, except fluorine. None of the halogens is particularly abundant in nature, however all are easily accessible in concentrated forms re ...
... Na2S(aq) + 2 HCl(aq) → 2 NaCl(aq) + H2S(g) 5. Discuss the halogens. (5 points) Each halogen is obtained by oxidation of the halide ion to the halogen in a molten salt, except fluorine. None of the halogens is particularly abundant in nature, however all are easily accessible in concentrated forms re ...
Reactions and Equations
... molecule there is. The coefficient applies to all atoms in the molecule. – Ex: There are two water molecules – Ex: There are 4 hydrogen atoms and 2 oxygen atoms ...
... molecule there is. The coefficient applies to all atoms in the molecule. – Ex: There are two water molecules – Ex: There are 4 hydrogen atoms and 2 oxygen atoms ...
Chapter 2 - OrgSites.com
... 8. Answer the following for the element carbon: a. Atomic number ________________ b. Atomic mass __________________ c. Number of protons ______________ d. Number of electrons ____________ 9. What are isotopes (not a baseball team)? 10. Give 2 examples of stable carbon isotopes. a. b. Unit 1 Study Gu ...
... 8. Answer the following for the element carbon: a. Atomic number ________________ b. Atomic mass __________________ c. Number of protons ______________ d. Number of electrons ____________ 9. What are isotopes (not a baseball team)? 10. Give 2 examples of stable carbon isotopes. a. b. Unit 1 Study Gu ...
Experiment 11 – Comparison of the Energy Content of Fuels
... carbon based fuels, such as coal or gasoline. Gasoline is a type of fuel composed almost completely of liquid hydrocarbons. What’s a hydrocarbon? A hydrocarbon is a compound that is composed only of hydrogen and carbon. Gasoline is one example, since it’s a mixture of liquid hydrocarbons. The sizes ...
... carbon based fuels, such as coal or gasoline. Gasoline is a type of fuel composed almost completely of liquid hydrocarbons. What’s a hydrocarbon? A hydrocarbon is a compound that is composed only of hydrogen and carbon. Gasoline is one example, since it’s a mixture of liquid hydrocarbons. The sizes ...
HONG KONG DIPLOMA OF SECONDARY EDUCATION
... Caesium, Cs, is the chemical element with atomic number 55. It is a soft, silverygold metal with melting point of 28C. It is the least abundant metal among the metals (excluding Francium) in the same group of the Periodic Table. Caesium reacts explosively in cold water and the product formed, caes ...
... Caesium, Cs, is the chemical element with atomic number 55. It is a soft, silverygold metal with melting point of 28C. It is the least abundant metal among the metals (excluding Francium) in the same group of the Periodic Table. Caesium reacts explosively in cold water and the product formed, caes ...
quant6stoichiom
... Carbon dioxide that is produced by astronauts can be removed with lithium hydroxide. The reaction produces lithium carbonate and water. An astronaut produces an average of 1.00 x 103 g carbon dioxide per day. How much lithium hydroxide should engineers put on the spaceship per astronaut, per day? - ...
... Carbon dioxide that is produced by astronauts can be removed with lithium hydroxide. The reaction produces lithium carbonate and water. An astronaut produces an average of 1.00 x 103 g carbon dioxide per day. How much lithium hydroxide should engineers put on the spaceship per astronaut, per day? - ...
Single Replacement Reactions
... with excess water and inform the instructor. Wear safety goggles and closed toed shoes throughout the entirety of the lab procedure.*** b. Label five test tubes - each with the name of one of the metals (zinc, aluminum, copper, iron and magnesium) if this has not been done already. c. Following your ...
... with excess water and inform the instructor. Wear safety goggles and closed toed shoes throughout the entirety of the lab procedure.*** b. Label five test tubes - each with the name of one of the metals (zinc, aluminum, copper, iron and magnesium) if this has not been done already. c. Following your ...
The Atomic Theory
... Law of Multiple Proportions: If two elements combine to form more than one compound the different weights of one which combine with the same weight of the other are in the ration of small whole numbers. Law of Reciprocal Proportions: The weights of two (or more) substances which react separately wit ...
... Law of Multiple Proportions: If two elements combine to form more than one compound the different weights of one which combine with the same weight of the other are in the ration of small whole numbers. Law of Reciprocal Proportions: The weights of two (or more) substances which react separately wit ...
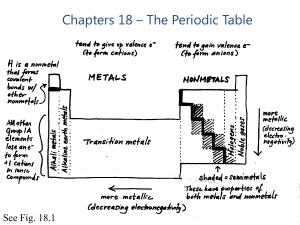
Chapters 18 – The Periodic Table
... element behaves like a true metal Common behavior: reaction with metal to become 2- ion in ionic compound; for most metals, most common minerals are oxides or sulfides Covalent bonds with other NMs; series of covalent hydrides (H2X) All but O have d orbitals available, so more than an octet is ...
... element behaves like a true metal Common behavior: reaction with metal to become 2- ion in ionic compound; for most metals, most common minerals are oxides or sulfides Covalent bonds with other NMs; series of covalent hydrides (H2X) All but O have d orbitals available, so more than an octet is ...
Test Booklet
... 22 Using the solubility graph provided, a student performs an experiment to find the solubility of a substance. The student finds the amount of substance needed to make a saturated solution in 100 g of water at different temperatures. The student’s data are shown in the table below the graph. ...
... 22 Using the solubility graph provided, a student performs an experiment to find the solubility of a substance. The student finds the amount of substance needed to make a saturated solution in 100 g of water at different temperatures. The student’s data are shown in the table below the graph. ...
Unit 2
... a) Solid zinc metal reacts with sulfuric acid to produce hydrogen gas and a solution of zinc sulfate. ...
... a) Solid zinc metal reacts with sulfuric acid to produce hydrogen gas and a solution of zinc sulfate. ...
Unit 2
... a) Solid zinc metal reacts with sulfuric acid to produce hydrogen gas and a solution of zinc sulfate. ...
... a) Solid zinc metal reacts with sulfuric acid to produce hydrogen gas and a solution of zinc sulfate. ...
Water splitting

Water splitting is the general term for a chemical reaction in which water is separated into oxygen and hydrogen. Efficient and economical water splitting would be a key technology component of a hydrogen economy. Various techniques for water splitting have been issued in water splitting patents in the United States. In photosynthesis, water splitting donates electrons to power the electron transport chain in photosystem II.