CP Chemistry Practice Mid
... 16. What is the molecular geometry, or shape, of a molecule of water a. linear b. bent c. tringonal planar d. trigonal pyramidal 17. How many grams of calcium carbonate would be needed to produce 44.8 liters of carbon dioxide gas measured at STP? CaCO3 + 2 HCl CaCl2 + H2O + CO2 a. 50 b. 100 c. 111 ...
... 16. What is the molecular geometry, or shape, of a molecule of water a. linear b. bent c. tringonal planar d. trigonal pyramidal 17. How many grams of calcium carbonate would be needed to produce 44.8 liters of carbon dioxide gas measured at STP? CaCO3 + 2 HCl CaCl2 + H2O + CO2 a. 50 b. 100 c. 111 ...
Chemistry I – Fall 2004
... (B) NH4Cl (C) CCl4 (D) CO2 14. Covalent bonds are most likely to be found in the compound represented by the formula (A) NaCl (B) KBr (C) CH4 (D) HI E) CaF2 15. A pure substance melts at 113 °C and does not conduct electricity in either the solid or liquid state. What conclusions can be drawn concer ...
... (B) NH4Cl (C) CCl4 (D) CO2 14. Covalent bonds are most likely to be found in the compound represented by the formula (A) NaCl (B) KBr (C) CH4 (D) HI E) CaF2 15. A pure substance melts at 113 °C and does not conduct electricity in either the solid or liquid state. What conclusions can be drawn concer ...
South Pasadena • AP Chemistry
... 8. If some of the gas bubbles from the reaction had escaped out the bottom of the tube, how would this have affected your molar volume at STP? Explain. ...
... 8. If some of the gas bubbles from the reaction had escaped out the bottom of the tube, how would this have affected your molar volume at STP? Explain. ...
Exam Review
... 16. What volume of 0.250 M H3PO4 is required to neutralize 30.00 cm3 of a 0.0500 M Ba(OH)2 solution? 17. What mass of Ca(OH)2 would be required to completely neutralize 50.0 cm3 of 0.125 M HCl? 18. What mass of Mg(OH)2 would be required to completely neutralize 70.0 cm3 of 0.175 M HNO3? *19. Hydrazi ...
... 16. What volume of 0.250 M H3PO4 is required to neutralize 30.00 cm3 of a 0.0500 M Ba(OH)2 solution? 17. What mass of Ca(OH)2 would be required to completely neutralize 50.0 cm3 of 0.125 M HCl? 18. What mass of Mg(OH)2 would be required to completely neutralize 70.0 cm3 of 0.175 M HNO3? *19. Hydrazi ...
ConcepTest On Simple Redox Reactions
... Comment to Instructor: Correct answer is 3. HCl. Since the oxidation number of H is decreasing from +1 to 0, it is undergoing reduction. Zn is being oxidized, and HCl is the “agent” that is causing the Zn to be oxidized. #4 indicates that the student is thinking that the Zn+2in ZnCl2 is undergoing r ...
... Comment to Instructor: Correct answer is 3. HCl. Since the oxidation number of H is decreasing from +1 to 0, it is undergoing reduction. Zn is being oxidized, and HCl is the “agent” that is causing the Zn to be oxidized. #4 indicates that the student is thinking that the Zn+2in ZnCl2 is undergoing r ...
Chemistry Midterm Review 2006
... b) Air has a total pressure of 20.6 atm and contains carbon monoxide, oxygen, and nitrogen. If air is made up of 0.6 atm of carbon monoxide, 12.6 atm of oxygen, what would be the partial pressure of nitrogen? c) If a sample of gas occupies 15.9 L at 34 C, what will its volume be at 27 C if the press ...
... b) Air has a total pressure of 20.6 atm and contains carbon monoxide, oxygen, and nitrogen. If air is made up of 0.6 atm of carbon monoxide, 12.6 atm of oxygen, what would be the partial pressure of nitrogen? c) If a sample of gas occupies 15.9 L at 34 C, what will its volume be at 27 C if the press ...
APES Lesson 23B (2014-15) - Matter, Chemistry - science-b
... storage. This energy may be released as kinetic energy, or energy of motion, when the object falls and is accelerated by the force of gravity. When water evaporates and rises into a cloud, it gains considerable potential energy. When that water falls to the ground and runs downstream in a river, its ...
... storage. This energy may be released as kinetic energy, or energy of motion, when the object falls and is accelerated by the force of gravity. When water evaporates and rises into a cloud, it gains considerable potential energy. When that water falls to the ground and runs downstream in a river, its ...
Precipitation and Redox Reactions
... Oxidation and Reduction (Redox) Early chemists saw “oxidation” reactions only as the combination of a material with oxygen to produce an oxide. For example, when gasoline burns in air, it oxidizes and forms oxides of carbon and hydrogen (oxides are compounds containing Oxygen, duh) 2 C8H18 + 25 O2 ...
... Oxidation and Reduction (Redox) Early chemists saw “oxidation” reactions only as the combination of a material with oxygen to produce an oxide. For example, when gasoline burns in air, it oxidizes and forms oxides of carbon and hydrogen (oxides are compounds containing Oxygen, duh) 2 C8H18 + 25 O2 ...
Pre- AP & NET IONIC EQUATIONS
... Anhydrides taught with synthesis and decomposition reactions Calculation of oxidation numbers Oxidation, reduction, oxidizing agents, and reducing agents. Net ionic equations for single and double replacement (taught with solutions) Solubility rules ...
... Anhydrides taught with synthesis and decomposition reactions Calculation of oxidation numbers Oxidation, reduction, oxidizing agents, and reducing agents. Net ionic equations for single and double replacement (taught with solutions) Solubility rules ...
chemistry i - surrattchemistry
... 3. A biochemist is performing an experiment to determine the effects of Chemical X on the growth of bacteria. Which tube is the experimental control? a. Test tube 1 b. Test tube 2 c. Test tube 3 d. Test tube 4 Objective 2.01: Analyze the historical development of the current atomic theory. 4. Which ...
... 3. A biochemist is performing an experiment to determine the effects of Chemical X on the growth of bacteria. Which tube is the experimental control? a. Test tube 1 b. Test tube 2 c. Test tube 3 d. Test tube 4 Objective 2.01: Analyze the historical development of the current atomic theory. 4. Which ...
Chemistry Definitions
... Ligand: The ions of d-block elements have low energy unfilled orbitals. These orbitals can a pair of electrons from some species, known as ligands. Also can be defined as a molecule or ion which has a spare pair of electrons which it can donate to a transition element via a dative bond. Dative bond ...
... Ligand: The ions of d-block elements have low energy unfilled orbitals. These orbitals can a pair of electrons from some species, known as ligands. Also can be defined as a molecule or ion which has a spare pair of electrons which it can donate to a transition element via a dative bond. Dative bond ...
Type of Chemical Reactions Lab
... If it is not, tell your teacher 3. The BTB indicator will turn blue in a base and yellow in an acid. The formation of oxygen is accompanied by the formation of acid. The formation of hydrogen is accompanied by the formation of base. iii. Use the other clamp to support a 9 volt battery. Attach an all ...
... If it is not, tell your teacher 3. The BTB indicator will turn blue in a base and yellow in an acid. The formation of oxygen is accompanied by the formation of acid. The formation of hydrogen is accompanied by the formation of base. iii. Use the other clamp to support a 9 volt battery. Attach an all ...
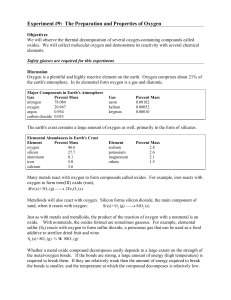
Experiment # 9 Properties of Oxygen
... Just as with metals and metalloids, the product of the reaction of oxygen with a nonmetal is an oxide. With nonmetals, the oxides formed are sometimes gaseous. For example, elemental sulfur (S8) reacts with oxygen to form sulfur dioxide, a poisonous gas that can be used as a food additive to sterili ...
... Just as with metals and metalloids, the product of the reaction of oxygen with a nonmetal is an oxide. With nonmetals, the oxides formed are sometimes gaseous. For example, elemental sulfur (S8) reacts with oxygen to form sulfur dioxide, a poisonous gas that can be used as a food additive to sterili ...
Per 3 - Old Saybrook Public Schools
... may have been added and removed from each chapter, so use your notes as your primary source of reference. ü I am available Tuesday after school until 3:15. I am here every morning by 7:05 and stay until 3:15. So, if you have questions or the times above do not work for you, just ask and we can ...
... may have been added and removed from each chapter, so use your notes as your primary source of reference. ü I am available Tuesday after school until 3:15. I am here every morning by 7:05 and stay until 3:15. So, if you have questions or the times above do not work for you, just ask and we can ...
Per 5 - Old Saybrook Public Schools
... may have been added and removed from each chapter, so use your notes as your primary source of reference. ü I am available Tuesday after school until 3:15. I am here every morning by 7:05 and stay until 3:15. So, if you have questions or the times above do not work for you, just ask and we can ...
... may have been added and removed from each chapter, so use your notes as your primary source of reference. ü I am available Tuesday after school until 3:15. I am here every morning by 7:05 and stay until 3:15. So, if you have questions or the times above do not work for you, just ask and we can ...
AP Chemistry Summer Assignment
... (on the reactant side) then it may displace only the metal in the compound. The metal (element) doing the displacing must be more active than the "metal" in the compound. To determine activity you must use the activity (electromotive) series. If the element is a nonmetal (on the reactant side) ...
... (on the reactant side) then it may displace only the metal in the compound. The metal (element) doing the displacing must be more active than the "metal" in the compound. To determine activity you must use the activity (electromotive) series. If the element is a nonmetal (on the reactant side) ...
Scientific Principles: Chemical Properties
... • Are pure substances consisting of one type of atom • Cannot be broken down or changed into another substance • Combine with other elements to form compounds ...
... • Are pure substances consisting of one type of atom • Cannot be broken down or changed into another substance • Combine with other elements to form compounds ...
Grade 11 Chemistry Exam Review
... a) oxygen is an odourless gas b) a water solution of copper(II) sulfate is blue c) 203.9 g of sugar can be dissolved in 100 g of water at 20ºC d) mercury is a liquid at room temperature ...
... a) oxygen is an odourless gas b) a water solution of copper(II) sulfate is blue c) 203.9 g of sugar can be dissolved in 100 g of water at 20ºC d) mercury is a liquid at room temperature ...
the optimization of proton exchange membrane hydrogen fuel cells
... There are four main components to a hydrogen fuel cell: anode, cathode, electrolyte, and catalyst. The placement of each of these components are illustrated in Figure 2. In order for a PEMFC to produce energy, it must undergo an electrochemical process called an oxidation reduction reaction. An exte ...
... There are four main components to a hydrogen fuel cell: anode, cathode, electrolyte, and catalyst. The placement of each of these components are illustrated in Figure 2. In order for a PEMFC to produce energy, it must undergo an electrochemical process called an oxidation reduction reaction. An exte ...
Water splitting

Water splitting is the general term for a chemical reaction in which water is separated into oxygen and hydrogen. Efficient and economical water splitting would be a key technology component of a hydrogen economy. Various techniques for water splitting have been issued in water splitting patents in the United States. In photosynthesis, water splitting donates electrons to power the electron transport chain in photosystem II.