Chapter 2 1
... Hence, even though it is called “Atomic Theory” because, of course, it could be wrong, no scientist thinks in any other terms than atoms nor that there is another explanation. Chapter 2 ...
... Hence, even though it is called “Atomic Theory” because, of course, it could be wrong, no scientist thinks in any other terms than atoms nor that there is another explanation. Chapter 2 ...
Question paper - Edexcel
... Answer ALL the questions in this section. You should aim to spend no more than 20 minutes on this section. For each question, select one answer from A to D and put a cross in the box . and then mark your new answer with a If you change your mind, put a line through the box cross . 1 In which of the ...
... Answer ALL the questions in this section. You should aim to spend no more than 20 minutes on this section. For each question, select one answer from A to D and put a cross in the box . and then mark your new answer with a If you change your mind, put a line through the box cross . 1 In which of the ...
Effect Of Convection For Gaseous Hydrochloride
... metals. It is collected as a dust. For the steel production, 6 millions ton/year in the Czech, 100 000 t of the dust /year is produced. It cannot be recycled as a iron feedstock because it contains not only 50% of Fe but also 5-15% Zn, mostly in form of zinc ferrite (franklinite) ZnFe2O4, and furthe ...
... metals. It is collected as a dust. For the steel production, 6 millions ton/year in the Czech, 100 000 t of the dust /year is produced. It cannot be recycled as a iron feedstock because it contains not only 50% of Fe but also 5-15% Zn, mostly in form of zinc ferrite (franklinite) ZnFe2O4, and furthe ...
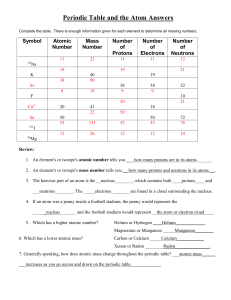
Periodic Table and the Atom Answers
... do this reaction with 125 grams of acetic acid and 275 grams of aluminum hydroxide. Two calculations are required. One determines the quantity of aluminum acetate that can be made with 125 grams of acetic acid and the other determines the quantity of aluminum acetate that can be made using 275 grams ...
... do this reaction with 125 grams of acetic acid and 275 grams of aluminum hydroxide. Two calculations are required. One determines the quantity of aluminum acetate that can be made with 125 grams of acetic acid and the other determines the quantity of aluminum acetate that can be made using 275 grams ...
Section 1e chemical formulae and chemical equations
... compounds to determine how much there is (in grams or in percentage) of each element in a compound. There are two common experimental methods which allow us to obtain this information. Which method is used depends on the type of compound. We will restrict ourselves to two types of compounds only and ...
... compounds to determine how much there is (in grams or in percentage) of each element in a compound. There are two common experimental methods which allow us to obtain this information. Which method is used depends on the type of compound. We will restrict ourselves to two types of compounds only and ...
Question 2
... c) What mass of CH4 must have reacted, if 2 mg HCl is liberated? d) Calculate the mass of both CH4 and Cl2 required to produce exactly 0.761 kg CCl4? ...
... c) What mass of CH4 must have reacted, if 2 mg HCl is liberated? d) Calculate the mass of both CH4 and Cl2 required to produce exactly 0.761 kg CCl4? ...
Date - PetyaPisanScienceAQ
... 2) Copper reacts slowly when heated to from an oxide layer. Copper is a bigger atom and therefore not as reactive with water and ...
... 2) Copper reacts slowly when heated to from an oxide layer. Copper is a bigger atom and therefore not as reactive with water and ...
Part 3 Answers Only for Questions, Exercises, and Problems in The
... 58. Kinetic energy increases. 60. The masses would be the same. Products of a chemical reaction have the same total mass as reactants. 62. Potential energy of the water above the dam changes into kinetic energy as it falls into the turbine. The turbine drives a generator in which kinetic energy is c ...
... 58. Kinetic energy increases. 60. The masses would be the same. Products of a chemical reaction have the same total mass as reactants. 62. Potential energy of the water above the dam changes into kinetic energy as it falls into the turbine. The turbine drives a generator in which kinetic energy is c ...
Answers to Selected Questions and Problems
... (d) main-group element; (e) relative atomic mass; (f) mass number; (g) isotope; (h) cation; (i) subatomic particle; (j) alkali metal; (k) periodic table Dalton used the laws of conservation of mass (Lavoisier) and definite proportions (Proust). They differ in their atomic masses and chemical propert ...
... (d) main-group element; (e) relative atomic mass; (f) mass number; (g) isotope; (h) cation; (i) subatomic particle; (j) alkali metal; (k) periodic table Dalton used the laws of conservation of mass (Lavoisier) and definite proportions (Proust). They differ in their atomic masses and chemical propert ...
Example 1: An experiment shows that 64g of
... compounds to determine how much there is (in grams or in percentage) of each element in a compound. There are two common experimental methods which allow us to obtain this information. Which method is used depends on the type of compound. We will restrict ourselves to two types of compounds only and ...
... compounds to determine how much there is (in grams or in percentage) of each element in a compound. There are two common experimental methods which allow us to obtain this information. Which method is used depends on the type of compound. We will restrict ourselves to two types of compounds only and ...
Example 1: An experiment shows that 64g of
... compounds to determine how much there is (in grams or in percentage) of each element in a compound. There are two common experimental methods which allow us to obtain this information. Which method is used depends on the type of compound. We will restrict ourselves to two types of compounds only and ...
... compounds to determine how much there is (in grams or in percentage) of each element in a compound. There are two common experimental methods which allow us to obtain this information. Which method is used depends on the type of compound. We will restrict ourselves to two types of compounds only and ...
Scientific Measurement
... _____11. I can answer questions about the Law of Conservation of Matter (Mass). ...
... _____11. I can answer questions about the Law of Conservation of Matter (Mass). ...
Balanced Chemical Equation
... VIA THE HALF REACTION METHOD • Redox reactions that take place in aqueous media often involve water, hydronium ions, and hydroxide ions as reactants or products. • Although these species are not oxidized or reduced, they do participate in the chemical change in other ways. • These reactions are diff ...
... VIA THE HALF REACTION METHOD • Redox reactions that take place in aqueous media often involve water, hydronium ions, and hydroxide ions as reactants or products. • Although these species are not oxidized or reduced, they do participate in the chemical change in other ways. • These reactions are diff ...
importance of chemistry in geothermal exploration and utilization
... calcium (Ca), magnesium (Mg), carbonate (total CO2), hydrogen sulphide (H2S), sulphate (SO4), chloride (Cl) and fluoride (F). The changes in chemistry due to exploitation are primarily caused by invention and mixing of other water types. Most often this is cold water but sometimes it may have undert ...
... calcium (Ca), magnesium (Mg), carbonate (total CO2), hydrogen sulphide (H2S), sulphate (SO4), chloride (Cl) and fluoride (F). The changes in chemistry due to exploitation are primarily caused by invention and mixing of other water types. Most often this is cold water but sometimes it may have undert ...
lect 7
... is, organisms take electrons from the organic carbon, run it through their metabolic cycles, and then dump the electron to an electron acceptor. When oxygen is not present, microorganisms must seek alternate electron acceptors. The energy gain for the organisms is the energy difference between reduc ...
... is, organisms take electrons from the organic carbon, run it through their metabolic cycles, and then dump the electron to an electron acceptor. When oxygen is not present, microorganisms must seek alternate electron acceptors. The energy gain for the organisms is the energy difference between reduc ...
Electrolysis answers - Barnard Castle School
... (if they do not get the correct answer we then look for working marks general principle is one mark lost for each mistake) giving the equation volume / 24000 = mass of gas / Mr (one mark) answer 0.0355 = 2 marks (without working) answers based on Mr of chlorine as 35.5 are worth a maximum of 2 marks ...
... (if they do not get the correct answer we then look for working marks general principle is one mark lost for each mistake) giving the equation volume / 24000 = mass of gas / Mr (one mark) answer 0.0355 = 2 marks (without working) answers based on Mr of chlorine as 35.5 are worth a maximum of 2 marks ...
Lab #4: Chemical Reactions
... hydrochloric acid (HCl) into a medium test tube. Add a small piece of magnesium metal (labeled "magnesium turnings") to the test tube. Record any evidence of chemical change in the Part C-2 Observation Data Table. If a gas is produced, hold your thumb over the mouth of the test tube for about one mi ...
... hydrochloric acid (HCl) into a medium test tube. Add a small piece of magnesium metal (labeled "magnesium turnings") to the test tube. Record any evidence of chemical change in the Part C-2 Observation Data Table. If a gas is produced, hold your thumb over the mouth of the test tube for about one mi ...
Name AP Chemistry Take Home Quiz – Due Thursday, 1/9/2014
... 12. Oxygen, which is 16 times as dense as hydrogen, diffuses: a. 1/16 times as fast. b. 1/4 times as fast. c. 4 times as fast. d. 16 times as fast e. equally as fast as hydrogen. 13. The density of an unknown gas is 4.20 grams per liter at 3.00 atmospheres pressure and 127 °C. What is the molecular ...
... 12. Oxygen, which is 16 times as dense as hydrogen, diffuses: a. 1/16 times as fast. b. 1/4 times as fast. c. 4 times as fast. d. 16 times as fast e. equally as fast as hydrogen. 13. The density of an unknown gas is 4.20 grams per liter at 3.00 atmospheres pressure and 127 °C. What is the molecular ...
Water splitting

Water splitting is the general term for a chemical reaction in which water is separated into oxygen and hydrogen. Efficient and economical water splitting would be a key technology component of a hydrogen economy. Various techniques for water splitting have been issued in water splitting patents in the United States. In photosynthesis, water splitting donates electrons to power the electron transport chain in photosystem II.