by John Mu
... ones using special catalysts The catalyst used is called a zeolite If it is not decomposed entirely the Hydrocarbons are further decomposed by steam cracking − A mixture of alkanes with steam is passed through very hot metal tubes. ...
... ones using special catalysts The catalyst used is called a zeolite If it is not decomposed entirely the Hydrocarbons are further decomposed by steam cracking − A mixture of alkanes with steam is passed through very hot metal tubes. ...
Class XI Chemistry Practics Paper
... Q2 Why 4s orbital is filled earlier to 3d orbital? Q3.Locate the position of element with atomic number 33. Q4. Define Charles law. Q5.What is the entropy change when a liquid vaporizes? Q6.What is the conjugate acid of NH3? Q7.Which out of the two- lithium or sodium forms nitrides? Q8.What effect d ...
... Q2 Why 4s orbital is filled earlier to 3d orbital? Q3.Locate the position of element with atomic number 33. Q4. Define Charles law. Q5.What is the entropy change when a liquid vaporizes? Q6.What is the conjugate acid of NH3? Q7.Which out of the two- lithium or sodium forms nitrides? Q8.What effect d ...
Standard - Santee Education Complex
... oxidize (rust and tarnish) readily and form positive ions (cations). They are excellent conductors of both heat and electricity. The metals can be broken down into several groups. Transition metals (also called the transition elements) are known for their ability to refract light as a result of thei ...
... oxidize (rust and tarnish) readily and form positive ions (cations). They are excellent conductors of both heat and electricity. The metals can be broken down into several groups. Transition metals (also called the transition elements) are known for their ability to refract light as a result of thei ...
Lab Manual
... The ideal gas law can be derived from the kinetic theory of gases and relies on the assumptions that (1) the gas consists of a large number of molecules, which are in random motion and obey Newton's laws of motion; (2) the volume of the molecules is negligibly small compared to the volume occupied b ...
... The ideal gas law can be derived from the kinetic theory of gases and relies on the assumptions that (1) the gas consists of a large number of molecules, which are in random motion and obey Newton's laws of motion; (2) the volume of the molecules is negligibly small compared to the volume occupied b ...
RedOx notes:
... Continue with elements picking their preferred charges (work from outside columns to the inner “valley of confusion”) until there is only one left; if the element is last to choose it must have the charge that makes everything else sum to zero. If you don’t choose first you might not get your first ...
... Continue with elements picking their preferred charges (work from outside columns to the inner “valley of confusion”) until there is only one left; if the element is last to choose it must have the charge that makes everything else sum to zero. If you don’t choose first you might not get your first ...
Chemistry in Society Homework Booklet
... By-products are usually created in industrial chemical processes. (a) Explain what is meant by a by-product. (b) How can by-products be (i) profit making? (ii) profit losing? ...
... By-products are usually created in industrial chemical processes. (a) Explain what is meant by a by-product. (b) How can by-products be (i) profit making? (ii) profit losing? ...
Photosynthesis in Hydrogen-Dominated Atmospheres
... to generate a list of nearly [54] all plausible carbon-based chemicals of a defined size. For this work, we chose molecules of up to 9 non-hydrogen atoms, made of C, N, O, S in oxidation states −2, 0, 2 or 4, and P in oxidation state +5. Rings of 4 or more atoms were allowed. This resulted in a set ...
... to generate a list of nearly [54] all plausible carbon-based chemicals of a defined size. For this work, we chose molecules of up to 9 non-hydrogen atoms, made of C, N, O, S in oxidation states −2, 0, 2 or 4, and P in oxidation state +5. Rings of 4 or more atoms were allowed. This resulted in a set ...
Spring 2013 Semester Exam Study Guide (Bonding, Nomenclature
... a. CHO c. C3H4O3 b. CH2O d. C2H4O2 ____ 85. Of the following molecular formulas for hydrocarbons, which is an empirical formula? a. CH4 c. C3H6 b. C2H2 d. C4H10 ____ 86. A compound's empirical formula is N2O5. If the formula mass is 108 amu, what is the molecular formula? a. N2O5 c. NO3 b. N4O10 d. ...
... a. CHO c. C3H4O3 b. CH2O d. C2H4O2 ____ 85. Of the following molecular formulas for hydrocarbons, which is an empirical formula? a. CH4 c. C3H6 b. C2H2 d. C4H10 ____ 86. A compound's empirical formula is N2O5. If the formula mass is 108 amu, what is the molecular formula? a. N2O5 c. NO3 b. N4O10 d. ...
Answers to NHSCE 2002 Part A Page 1
... 6. The Pan-Canadian protocol is not very clear about the depth to which organic chemistry should be studied at high school, and different provinces appear to have interpreted this aspect of the protocol in different ways. It is, however, quite clear that students should be able to recognize some of ...
... 6. The Pan-Canadian protocol is not very clear about the depth to which organic chemistry should be studied at high school, and different provinces appear to have interpreted this aspect of the protocol in different ways. It is, however, quite clear that students should be able to recognize some of ...
MC84 - Southchemistry.com
... When the substances in the equation above are at equilibrium at pressure P and temperature T, the equilibrium can be shifted to favor the products by (A) increasing the pressure by means of a moving piston at constant T (B) increasing the pressure by adding an inert gas such as nitrogen (C) decreasi ...
... When the substances in the equation above are at equilibrium at pressure P and temperature T, the equilibrium can be shifted to favor the products by (A) increasing the pressure by means of a moving piston at constant T (B) increasing the pressure by adding an inert gas such as nitrogen (C) decreasi ...
Skill Practice 1
... 2. For which substance, A or B, does the freezing point decrease as the pressure is increased? 3. One of the substances behaves more like most other substances. Which substance and what property allows you to tell? 4. Assuming that the temperature scales for both phase diagrams are the same, which c ...
... 2. For which substance, A or B, does the freezing point decrease as the pressure is increased? 3. One of the substances behaves more like most other substances. Which substance and what property allows you to tell? 4. Assuming that the temperature scales for both phase diagrams are the same, which c ...
Chemistry Entrance Material for Grade 11 to 12
... [-A-] The remaining liquid continues to become cooler because the molecules continue to evaporate all the time. [-B-] The liquid continues to cool down. [-C-] The liquid may cool down initially, but then it will stay at the same temperature because the system has reached equilibrium. [-D-] The remai ...
... [-A-] The remaining liquid continues to become cooler because the molecules continue to evaporate all the time. [-B-] The liquid continues to cool down. [-C-] The liquid may cool down initially, but then it will stay at the same temperature because the system has reached equilibrium. [-D-] The remai ...
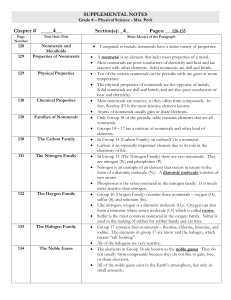
Ch. 11-12 Supplements
... 3) a. Write the balanced equation for the reaction of aqueous sulfuric acid, H2SO4, and solid aluminum hydroxide, Al(OH)3 forming liquid water and aqueous aluminum sulfate, Al2(SO4)3. If 30.0 grams of sulfuric acid and 25.0 grams of aluminum hydroxide react… b. How many grams of each product will b ...
... 3) a. Write the balanced equation for the reaction of aqueous sulfuric acid, H2SO4, and solid aluminum hydroxide, Al(OH)3 forming liquid water and aqueous aluminum sulfate, Al2(SO4)3. If 30.0 grams of sulfuric acid and 25.0 grams of aluminum hydroxide react… b. How many grams of each product will b ...
George Facer`s A level Chemistry
... The reaction involves nucleophilic attack on the carbon atom joined to the halogen by the lone pair of electrons on the oxygen in water. Since water is not as good a nucleophile as hydroxide ions, the reaction is much slower than that with aqueous potassium hydroxide. The halide ion formed then reac ...
... The reaction involves nucleophilic attack on the carbon atom joined to the halogen by the lone pair of electrons on the oxygen in water. Since water is not as good a nucleophile as hydroxide ions, the reaction is much slower than that with aqueous potassium hydroxide. The halide ion formed then reac ...
Name - TeacherWeb
... The elements in Group 18 are known as the noble gases. They do not usually form compounds because they do not like to gain, lose, or share electrons. All of the noble gases exist in the Earth’s atmosphere, but only in small amounts. ...
... The elements in Group 18 are known as the noble gases. They do not usually form compounds because they do not like to gain, lose, or share electrons. All of the noble gases exist in the Earth’s atmosphere, but only in small amounts. ...
www.xtremepapers.net
... Permission to reproduce items where third-party owned material protected by copyright is included has been sought and cleared where possible. Every reasonable effort has been made by the publisher (UCLES) to trace copyright holders, but if any items requiring clearance have unwittingly been included ...
... Permission to reproduce items where third-party owned material protected by copyright is included has been sought and cleared where possible. Every reasonable effort has been made by the publisher (UCLES) to trace copyright holders, but if any items requiring clearance have unwittingly been included ...
Balancing a Chemical Equation
... formulas for the products on the balance an equation by changing right with a yields sign (→) in the subscripts in a chemical formula. between. If two or more reactants or Each substance only has one products are involved, separate their ...
... formulas for the products on the balance an equation by changing right with a yields sign (→) in the subscripts in a chemical formula. between. If two or more reactants or Each substance only has one products are involved, separate their ...
Heat Capacity (C)
... temperature. Equality of temperature is a necessary and sufficient condition for thermal equilibrium. The Zeroth Law thus defines a property (temperature) and describes its behavior ...
... temperature. Equality of temperature is a necessary and sufficient condition for thermal equilibrium. The Zeroth Law thus defines a property (temperature) and describes its behavior ...
Water splitting

Water splitting is the general term for a chemical reaction in which water is separated into oxygen and hydrogen. Efficient and economical water splitting would be a key technology component of a hydrogen economy. Various techniques for water splitting have been issued in water splitting patents in the United States. In photosynthesis, water splitting donates electrons to power the electron transport chain in photosystem II.