Post Lab Questions
... In general most missed work is due on the next Friday. For any planned absence work that is due during the absence must be turned in before your departure. Laboratories, quizzes, and exams cannot be made up unless prior arrangements in writing have been made. If you make prior arrangements and miss ...
... In general most missed work is due on the next Friday. For any planned absence work that is due during the absence must be turned in before your departure. Laboratories, quizzes, and exams cannot be made up unless prior arrangements in writing have been made. If you make prior arrangements and miss ...
industry: applying chemical reactions
... their useful life is over. Will they be recycled? Or will they simply be left in a landfill somewhere, where their metals can leach into that community’s drinking water? I realize that not all of these scenarios are likely to happen. However, we must be willing to accept the consequences should some ...
... their useful life is over. Will they be recycled? Or will they simply be left in a landfill somewhere, where their metals can leach into that community’s drinking water? I realize that not all of these scenarios are likely to happen. However, we must be willing to accept the consequences should some ...
Student Solutions Manual Errata
... bonds are the attractions of oppositely charged ions to one another. We can think of the ions (or the spheres in the diagram) as being separate from, but strongly attracted to, one another. Covalent bonding occurs when two atoms are mutually attracted to a pair (or pairs) of electrons. Because the a ...
... bonds are the attractions of oppositely charged ions to one another. We can think of the ions (or the spheres in the diagram) as being separate from, but strongly attracted to, one another. Covalent bonding occurs when two atoms are mutually attracted to a pair (or pairs) of electrons. Because the a ...
Chemistry 133 Problem Set Introduction
... 1.81 Antifreeze contains the compound ethylene glycol. This compound not only lowers the freezing point of water but also increases the boiling point of water. The density of ethylene glycol is 9.35 lb/gal, and the density of water is 62.5 lb/ft3. (a) Is the density of water greater than the density ...
... 1.81 Antifreeze contains the compound ethylene glycol. This compound not only lowers the freezing point of water but also increases the boiling point of water. The density of ethylene glycol is 9.35 lb/gal, and the density of water is 62.5 lb/ft3. (a) Is the density of water greater than the density ...
PREPARMACY PHYSICAL CHEMISTRY THERMOCHEMISTRY
... chemical reactions. Thus, Heat of reaction may be defined as the amount of heat absorbed or evolved in a reaction when the number of moles of reactants as represented by the balanced chemical equation change completely into the products. For example, the heat change for the reaction of one mole of c ...
... chemical reactions. Thus, Heat of reaction may be defined as the amount of heat absorbed or evolved in a reaction when the number of moles of reactants as represented by the balanced chemical equation change completely into the products. For example, the heat change for the reaction of one mole of c ...
Section 4.9 Oxidation–Reduction Reactions
... If the actual yield for the previous problem was 10.5 g, calculate the percent yield. The theoretical yield that we calculated was 13.6 g. If the actual yield is 3.16 g then percent yield is ...
... If the actual yield for the previous problem was 10.5 g, calculate the percent yield. The theoretical yield that we calculated was 13.6 g. If the actual yield is 3.16 g then percent yield is ...
Magnesium based ternary metal hydrides containing alkali and
... K. Yvon, B. Bertheville / Journal of Alloys and Compounds xxx (2006) xxx–xxx ...
... K. Yvon, B. Bertheville / Journal of Alloys and Compounds xxx (2006) xxx–xxx ...
Post Lab Questions
... In general most missed work is due on the next Friday. For any planned absence work that is due during the absence must be turned in before your departure. Laboratories, quizzes, and exams cannot be made up unless prior arrangements in writing have been made. If you make prior arrangements and miss ...
... In general most missed work is due on the next Friday. For any planned absence work that is due during the absence must be turned in before your departure. Laboratories, quizzes, and exams cannot be made up unless prior arrangements in writing have been made. If you make prior arrangements and miss ...
Chapter 19
... In situations such as the formation of ammonia, where two atoms share electrons, how is it possible to say that one atom lost electrons and was oxidized, while the other atom gained electrons and was reduced? To answer this, you need to know which atom attracts electrons more strongly, or, in other ...
... In situations such as the formation of ammonia, where two atoms share electrons, how is it possible to say that one atom lost electrons and was oxidized, while the other atom gained electrons and was reduced? To answer this, you need to know which atom attracts electrons more strongly, or, in other ...
The Mole - Humble ISD
... What if we had 50 moles of hydrogen, how much oxygen would we need and how much water produced? 25 mol O2, 50 mol H2O ...
... What if we had 50 moles of hydrogen, how much oxygen would we need and how much water produced? 25 mol O2, 50 mol H2O ...
aq - Valencia College
... a substance reported in grams per moles. a. For hydrates, the water of hydration must be included. 2) The molar mass for FeSO4·H2O is shown by the following calculation. Amount of atoms/molecules multiplied by ...
... a substance reported in grams per moles. a. For hydrates, the water of hydration must be included. 2) The molar mass for FeSO4·H2O is shown by the following calculation. Amount of atoms/molecules multiplied by ...
Chapter 9 Stoichiometry
... 7. Calculate the amount in moles of a product produced, given the amounts in moles of two reactants, one of which is in excess. 8. Calculate the mass in grams of a product produced, given the mass in grams of two reactants, one of which is in excess. 9. Distinguish between theoretical yield, actual ...
... 7. Calculate the amount in moles of a product produced, given the amounts in moles of two reactants, one of which is in excess. 8. Calculate the mass in grams of a product produced, given the mass in grams of two reactants, one of which is in excess. 9. Distinguish between theoretical yield, actual ...
Praktikum in Allgemeiner Chemie für Biologen und Pharmazeuten
... can be removed quickly from the reaction vessel. An apparatus fixed on a stand has to be mounted so high that the electric heater must stand on a socket (e.g. “Labor-Boy” in order to make contact. In case of overheating the socket can be lowered or removed instantly. The Bunsen burner is a versatile ...
... can be removed quickly from the reaction vessel. An apparatus fixed on a stand has to be mounted so high that the electric heater must stand on a socket (e.g. “Labor-Boy” in order to make contact. In case of overheating the socket can be lowered or removed instantly. The Bunsen burner is a versatile ...
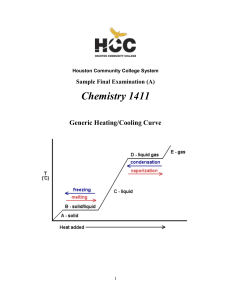
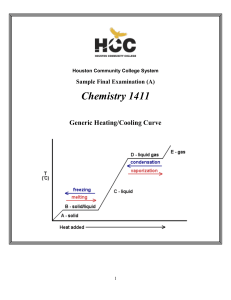
CHEM-1411 Final Practice Exam
... sulfur atom in the first structure is therefore sp3. However, the sulfur is not simply sp3 hybridized in the second structure, which has an “expanded octet” around the sulfur atom. Hybridizations that allow more than an octet of electrons around an atom are sp3d (10 electrons) and sp3d2 (12 electron ...
... sulfur atom in the first structure is therefore sp3. However, the sulfur is not simply sp3 hybridized in the second structure, which has an “expanded octet” around the sulfur atom. Hybridizations that allow more than an octet of electrons around an atom are sp3d (10 electrons) and sp3d2 (12 electron ...
CHEM 1411 Exam #2 - HCC Learning Web
... PART II- ( 5 points each) Please show all your work. 21. Consider the electronic transition from n = 2 to n = 5 in the hydrogen atom. a. Is light absorbed or emitted? b. Calculate the energy, the frequency and wavelength (in nm) of the photon involved. ...
... PART II- ( 5 points each) Please show all your work. 21. Consider the electronic transition from n = 2 to n = 5 in the hydrogen atom. a. Is light absorbed or emitted? b. Calculate the energy, the frequency and wavelength (in nm) of the photon involved. ...
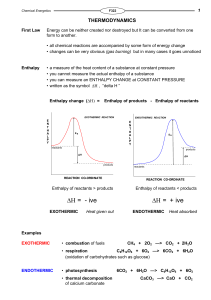
Enthalpy change
... • applying Hess’s Law enables one to calculate enthalpy changes from other data • used for calculating changes which can’t be measured directly - Lattice Enthalpy • used for calculating ...
... • applying Hess’s Law enables one to calculate enthalpy changes from other data • used for calculating changes which can’t be measured directly - Lattice Enthalpy • used for calculating ...
Water splitting

Water splitting is the general term for a chemical reaction in which water is separated into oxygen and hydrogen. Efficient and economical water splitting would be a key technology component of a hydrogen economy. Various techniques for water splitting have been issued in water splitting patents in the United States. In photosynthesis, water splitting donates electrons to power the electron transport chain in photosystem II.