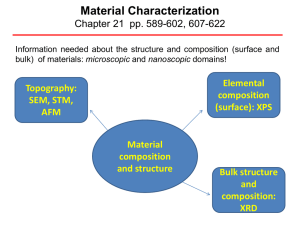
Material Characterization
... STM – Modes of Operation Constant Height Mode = By using a feedback loop the tip is vertically adjusted in such a way that the current always stays constant. As the current is proportional to the local density of states, the tip follows a contour of a constant density of states during scanning. A k ...
... STM – Modes of Operation Constant Height Mode = By using a feedback loop the tip is vertically adjusted in such a way that the current always stays constant. As the current is proportional to the local density of states, the tip follows a contour of a constant density of states during scanning. A k ...
ppt
... 2. To get analysis at micron scale, we need high energies (keV) focused on small area Raymond Castaing formulated the technique for microanalysis and built the first working unit by 1951. ...
... 2. To get analysis at micron scale, we need high energies (keV) focused on small area Raymond Castaing formulated the technique for microanalysis and built the first working unit by 1951. ...
Atomic Radii Answers File
... electron so the remaining ones are pulled in closer. When an atom gains an electron to form a negative ion, the nuclear charge has not changed. However, now the nucleus is attracting one more electron so they are not pulled as strongly as before. The negative ions will all be bigger than Ar and the ...
... electron so the remaining ones are pulled in closer. When an atom gains an electron to form a negative ion, the nuclear charge has not changed. However, now the nucleus is attracting one more electron so they are not pulled as strongly as before. The negative ions will all be bigger than Ar and the ...
2.2.3.- X-ray diffraction
... atomic spacing in crystals and, therefore, they could be diffracted [36]. This was immediately confirmed by Walter Friedrich and Paul Knipping [36]. In 1914 Darwin elaborated a Kinematic Theory of Diffraction, which assumed that xrays diffracted by each element in the volume of the material were ind ...
... atomic spacing in crystals and, therefore, they could be diffracted [36]. This was immediately confirmed by Walter Friedrich and Paul Knipping [36]. In 1914 Darwin elaborated a Kinematic Theory of Diffraction, which assumed that xrays diffracted by each element in the volume of the material were ind ...
AP Chemistry Study Guide – Chapter 7, Atomic Structure
... 6) Account for each of the following in terms of principles of atomic structure, including the number, properties, and arrangements of subatomic particles. (a) The second ionization energy of sodium is about three times greater than the second ionization energy of magnesium. (b) The difference betwe ...
... 6) Account for each of the following in terms of principles of atomic structure, including the number, properties, and arrangements of subatomic particles. (a) The second ionization energy of sodium is about three times greater than the second ionization energy of magnesium. (b) The difference betwe ...
Appendix I.
... constructive or destructive interference is experienced, depending whether the waves are in phase or in opposition. Constructive interference occurs if Bragg’s law of crystal diffraction is satisfied. The material identity is established as follows: (i) The wavelength, λ, of the incident radiation i ...
... constructive or destructive interference is experienced, depending whether the waves are in phase or in opposition. Constructive interference occurs if Bragg’s law of crystal diffraction is satisfied. The material identity is established as follows: (i) The wavelength, λ, of the incident radiation i ...
Section 4-2 What gives metals their distinctive properties?
... – Malleable – hammered or rolled into sheets – Ductile – can be drawn into wires ...
... – Malleable – hammered or rolled into sheets – Ductile – can be drawn into wires ...
Electron Diffraction
... revolutionary idea, since there was no evidence at the time that matter behaved like waves. In 1927, however, Clinton Davisson and Lester Germer discovered experimental proof of the wave-like properties of matterparticularly electrons (This discovery was quite by mistake!). They were studying electr ...
... revolutionary idea, since there was no evidence at the time that matter behaved like waves. In 1927, however, Clinton Davisson and Lester Germer discovered experimental proof of the wave-like properties of matterparticularly electrons (This discovery was quite by mistake!). They were studying electr ...
X-ray diffraction techniques X
... 1. Qualitatively, where the diffraction pattern is recorded and analysis of the spot positions gives information on the symmetry of the surface structure. In the presence of an adsorbate the qualitative analysis may reveal information about the size and rotational alignment of the adsorbate unit cel ...
... 1. Qualitatively, where the diffraction pattern is recorded and analysis of the spot positions gives information on the symmetry of the surface structure. In the presence of an adsorbate the qualitative analysis may reveal information about the size and rotational alignment of the adsorbate unit cel ...
electron diffraction - Department of Physics | Oregon State
... PROCEDURE: The procedure in this experiment is to determine from the diffraction rings and then to use the interference condition to find the corresponding wavelength. (This is similar to the way a diffraction grating is used for light – the wavelength can be calculated from the angular position o ...
... PROCEDURE: The procedure in this experiment is to determine from the diffraction rings and then to use the interference condition to find the corresponding wavelength. (This is similar to the way a diffraction grating is used for light – the wavelength can be calculated from the angular position o ...
ELECTRON DIFFRACTION
... that measured value for the wavelength of the electrons with the value expected from the deBroglie relationship. The diffraction rings are diffuse and hard to measure accurately. Therefore use calipers and average 3 separate measurements to get the best value for the ring diameters. When measuring, ...
... that measured value for the wavelength of the electrons with the value expected from the deBroglie relationship. The diffraction rings are diffuse and hard to measure accurately. Therefore use calipers and average 3 separate measurements to get the best value for the ring diameters. When measuring, ...
Moderne Methoden der Materialcharakterisierung
... – Tungsten – Lanthanum hexaboride (LaB6) – Others… ...
... – Tungsten – Lanthanum hexaboride (LaB6) – Others… ...
here - TCD Maths home - Trinity College Dublin
... In 1928, Max Knoll and a team of researchers at the Technological University of Berlin created a device that used two magnetic lenses to achieve higher magnifications, arguably the first electron microscope. The wave nature of electrons, which were considered charged matter particles, had not been f ...
... In 1928, Max Knoll and a team of researchers at the Technological University of Berlin created a device that used two magnetic lenses to achieve higher magnifications, arguably the first electron microscope. The wave nature of electrons, which were considered charged matter particles, had not been f ...
Physics 200 Class #1 Outline
... experiment at Bell Labs! They looked at crystal diffraction patterns using electrons. First, what is crystal diffraction? Light rays reflecting off the different faces of the crystal can interfere with each other. This is similar to thin film interference we spoke about. In the diagram below, we're ...
... experiment at Bell Labs! They looked at crystal diffraction patterns using electrons. First, what is crystal diffraction? Light rays reflecting off the different faces of the crystal can interfere with each other. This is similar to thin film interference we spoke about. In the diagram below, we're ...
EXPERIMENT Q-5 Electron Diffraction Abstract References Pre-Lab
... You should see a pair of circular rings concentric with the central spot. QUESTION 2: The Bragg law shows that diffracted electrons from a single crystal emerge at only one diffraction angle. This experiment uses a polycrystalline, or "powder" sample. Why do the diffracted electrons form a circle on ...
... You should see a pair of circular rings concentric with the central spot. QUESTION 2: The Bragg law shows that diffracted electrons from a single crystal emerge at only one diffraction angle. This experiment uses a polycrystalline, or "powder" sample. Why do the diffracted electrons form a circle on ...
X‐ray diffraction: Determining lattice constants of crystal structure
... the emitted x-rays have the following characteristic wavelengths: Kα = 71.08 pm and Kβ = 63.09 pm. These xrays are collimated into a beam by the collimating slit and the incident beam makes an angle θ with the surface of the crystal sample. The detector, a Geiger-Müller tube is mounted in such a way ...
... the emitted x-rays have the following characteristic wavelengths: Kα = 71.08 pm and Kβ = 63.09 pm. These xrays are collimated into a beam by the collimating slit and the incident beam makes an angle θ with the surface of the crystal sample. The detector, a Geiger-Müller tube is mounted in such a way ...
Secondary electrons
... -Chemical mapping of a polished sample surface. Accomplished by sweeping (rastering) the beam over the sample surface. Used for many of the same types of studies described above. Particularly useful for: -Location and distribution of phases -Quick location and identification of trace particles -Subt ...
... -Chemical mapping of a polished sample surface. Accomplished by sweeping (rastering) the beam over the sample surface. Used for many of the same types of studies described above. Particularly useful for: -Location and distribution of phases -Quick location and identification of trace particles -Subt ...
Reflection high-energy electron diffraction
Reflection high-energy electron diffraction (RHEED) is a technique used to characterize the surface of crystalline materials. RHEED systems gather information only from the surface layer of the sample, which distinguishes RHEED from other materials characterization methods that also rely on diffraction of high-energy electrons. Transmission electron microscopy, another common electron diffraction method samples the bulk of the sample due to the geometry of the system. Low-energy electron diffraction (LEED) is also surface sensitive, but LEED achieves surface sensitivity through the use of low energy electrons.