Chapter 2 Notes
... A. Physical Properties of Matter = things you can observe without changing a substance; Examples: color; measurements; texture B. Density = measure of the mass of an object divided by its volume; usually given in grams per cubic centimeter (g/cm ); Example: average density of liquid water is 1 g/cm ...
... A. Physical Properties of Matter = things you can observe without changing a substance; Examples: color; measurements; texture B. Density = measure of the mass of an object divided by its volume; usually given in grams per cubic centimeter (g/cm ); Example: average density of liquid water is 1 g/cm ...
Enhanced backward scattering by surface plasmons on silver film Applied Physics A
... ABSTRACT Surface plasmon and its potential application in nanotechnology have attracted a remarkable amount of attention recently due to their novel properties. In this work we present an angularly resolved surface plasmon scattering study on a primitive metal surface. Using a reversed attenuated to ...
... ABSTRACT Surface plasmon and its potential application in nanotechnology have attracted a remarkable amount of attention recently due to their novel properties. In this work we present an angularly resolved surface plasmon scattering study on a primitive metal surface. Using a reversed attenuated to ...
Worksheet 4 - Periodic Trends A number of physical and chemical
... When an electron is removed from an atom the repulsion between the remaining electrons decreases. The nuclear charge remains constant, so more energy is required to remove another electron from the positively charged ion. This means that, I1 < I2 < I3 < ..., for any given atom. Going down a group t ...
... When an electron is removed from an atom the repulsion between the remaining electrons decreases. The nuclear charge remains constant, so more energy is required to remove another electron from the positively charged ion. This means that, I1 < I2 < I3 < ..., for any given atom. Going down a group t ...
Basics of Material Sciences - E
... 3. Write about the vector model 4. Give a short note on Pauli’s exclusion Principle 8 Marks 1. Explain the properties of cathode rays with suitable experiment 2. Explain quantum numbers 3. Explain in detail about the Atomic Models ...
... 3. Write about the vector model 4. Give a short note on Pauli’s exclusion Principle 8 Marks 1. Explain the properties of cathode rays with suitable experiment 2. Explain quantum numbers 3. Explain in detail about the Atomic Models ...
Covalent Bonding - Effingham County Schools
... •As independent particles, most atoms are at relatively high potential energy. •Nature, however, favors arrangements in which potential energy is minimized. •This means that most atoms are less stable existing by themselves than when they are combined. •By bonding with each other, atoms decrease in ...
... •As independent particles, most atoms are at relatively high potential energy. •Nature, however, favors arrangements in which potential energy is minimized. •This means that most atoms are less stable existing by themselves than when they are combined. •By bonding with each other, atoms decrease in ...
Covalent Bonding - Effingham County Schools
... •As independent particles, most atoms are at relatively high potential energy. •Nature, however, favors arrangements in which potential energy is minimized. •This means that most atoms are less stable existing by themselves than when they are combined. •By bonding with each other, atoms decrease in ...
... •As independent particles, most atoms are at relatively high potential energy. •Nature, however, favors arrangements in which potential energy is minimized. •This means that most atoms are less stable existing by themselves than when they are combined. •By bonding with each other, atoms decrease in ...
CHEM 11 Practice Exam 2
... 8) Which of the following is a general trend from left to right in the periodic table of elements? A) atomic radius increases; ionization energy increases B) atomic radius increases; ionization energy decreases C) atomic radius decreases; ionization energy increases D) atomic radius decreases; ioni ...
... 8) Which of the following is a general trend from left to right in the periodic table of elements? A) atomic radius increases; ionization energy increases B) atomic radius increases; ionization energy decreases C) atomic radius decreases; ionization energy increases D) atomic radius decreases; ioni ...
Regents questions
... Arranging the elements by atomic weight leads to an order slightly different from that in a modern periodic table, where the arrangement is by atomic number. Why does this happen? ...
... Arranging the elements by atomic weight leads to an order slightly different from that in a modern periodic table, where the arrangement is by atomic number. Why does this happen? ...
Plasmonics
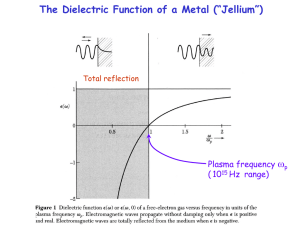
... at k=0. It is possible to provide the necessary momentum ħk by a grating, which transfers the k = 2/d (d = line spacing) . Thereby one can control the wavelength of surface plasmons. ...
... at k=0. It is possible to provide the necessary momentum ħk by a grating, which transfers the k = 2/d (d = line spacing) . Thereby one can control the wavelength of surface plasmons. ...
Lecture 30/31
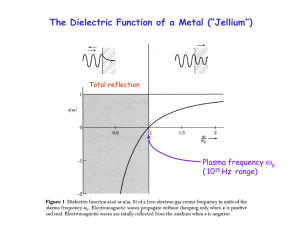
... at k=0. It is possible to provide the necessary momentum ħk by a grating, which transfers the k = 2/d (d = line spacing) . Thereby one can control the wavelength of surface plasmons. ...
... at k=0. It is possible to provide the necessary momentum ħk by a grating, which transfers the k = 2/d (d = line spacing) . Thereby one can control the wavelength of surface plasmons. ...
Care and Maintenance Sandstone / Quartzite
... Use only care and maintenance products that are specifically designed for natural stone. General purpose cleaners contain acids, ammonia, chelating agents and abrasives that can discolor, stain, dissolve, or dull the finish of the stone. The absorbency of unsealed sandstone and quartzite makes it su ...
... Use only care and maintenance products that are specifically designed for natural stone. General purpose cleaners contain acids, ammonia, chelating agents and abrasives that can discolor, stain, dissolve, or dull the finish of the stone. The absorbency of unsealed sandstone and quartzite makes it su ...
Chapter 5
... (Reprinted from Journal of Crystal Growth, Volume 271, T. Aoki, M. Takeguchi, P. Boieriu, R. Singh, C. Grein, Y. Chang, S. Sivananthan and David J. Smith, "Microstructural characterization of HgTe/HgCdTe superlattices" Pages 29-36, Copyright 2004, with permission from Elsevier. ) ...
... (Reprinted from Journal of Crystal Growth, Volume 271, T. Aoki, M. Takeguchi, P. Boieriu, R. Singh, C. Grein, Y. Chang, S. Sivananthan and David J. Smith, "Microstructural characterization of HgTe/HgCdTe superlattices" Pages 29-36, Copyright 2004, with permission from Elsevier. ) ...
Fall Exam 1
... 17. Which of the following statements about Thomson’s experiments with cathode rays is false? A. ...
... 17. Which of the following statements about Thomson’s experiments with cathode rays is false? A. ...
Eighth Grade Review - PAMS-Doyle
... A chemical equation represents the change that takes place in a chemical reaction • In a chemical equation, the chemical formulas of the reactants are written on the left; an arrow indicates a change to a new substance; and the chemical formulas of the products are written on the right ...
... A chemical equation represents the change that takes place in a chemical reaction • In a chemical equation, the chemical formulas of the reactants are written on the left; an arrow indicates a change to a new substance; and the chemical formulas of the products are written on the right ...
Advanced Characterization methods lectures
... IR spectra have similar selection rules as for optical spectroscopy – this means that many bands can not be measured with usual IR absorption. Raman spectroscopy (scattering) offers a way around this - since the interactions involve more than one photon, the selection rules are different than in sta ...
... IR spectra have similar selection rules as for optical spectroscopy – this means that many bands can not be measured with usual IR absorption. Raman spectroscopy (scattering) offers a way around this - since the interactions involve more than one photon, the selection rules are different than in sta ...
chapter5
... People can go up a ladder by going from rung to rung the way electrons in an atom can go from energy level to energy level. A person can never stand between the rungs of a ladder and electrons must always be at a specific energy level as well (and not between them). In order for a person to go from ...
... People can go up a ladder by going from rung to rung the way electrons in an atom can go from energy level to energy level. A person can never stand between the rungs of a ladder and electrons must always be at a specific energy level as well (and not between them). In order for a person to go from ...
Characterization of Nano Materials using Electron Microscopy
... evaporation. Conductive materials in current use for specimen coating include gold, gold/palladium alloy, platinum, osmium, iridium, tungsten, chromium and graphite [5]. Coating prevents the accumulation of static electric charge on the specimen during electron irradiation. An alternative to coating ...
... evaporation. Conductive materials in current use for specimen coating include gold, gold/palladium alloy, platinum, osmium, iridium, tungsten, chromium and graphite [5]. Coating prevents the accumulation of static electric charge on the specimen during electron irradiation. An alternative to coating ...
op_bessel1 - School of Physics
... Fraunhofer diffraction occurs when both the incident and diffracted waves are effectively plane. This occurs when the distance from the source to the aperture is large so that the aperture is assumed to be uniformly illuminated and the distance from the aperture plane to the observation plane is als ...
... Fraunhofer diffraction occurs when both the incident and diffracted waves are effectively plane. This occurs when the distance from the source to the aperture is large so that the aperture is assumed to be uniformly illuminated and the distance from the aperture plane to the observation plane is als ...
Fraunhofer Diffraction from a circular aperture
... Fraunhofer diffraction occurs when both the incident and diffracted waves are effectively plane. This occurs when the distance from the source to the aperture is large so that the aperture is assumed to be uniformly illuminated and the distance from the aperture plane to the observation plane is als ...
... Fraunhofer diffraction occurs when both the incident and diffracted waves are effectively plane. This occurs when the distance from the source to the aperture is large so that the aperture is assumed to be uniformly illuminated and the distance from the aperture plane to the observation plane is als ...
Ideal two-dimensional electron systems with a giant Rashba
... pair of spin-split surface states with negative effective masses (Fig. 3(b)). The resulting surface structure resembles dispersion of spin-orbit split surface states in surface alloys (see, e.g., [16]). Owing to noticeable anisotropy of the surface state dispersion of the upper state the CEC shown i ...
... pair of spin-split surface states with negative effective masses (Fig. 3(b)). The resulting surface structure resembles dispersion of spin-orbit split surface states in surface alloys (see, e.g., [16]). Owing to noticeable anisotropy of the surface state dispersion of the upper state the CEC shown i ...
Fang
... say that metals could be differentiated by their magnetic properties, so then we moved on to mechanical properties. o I said we could measure the elastic modulus, so he asked me what ratio do people ...
... say that metals could be differentiated by their magnetic properties, so then we moved on to mechanical properties. o I said we could measure the elastic modulus, so he asked me what ratio do people ...
Year 11 Chemistry Balancing Equations
... 17. Listed below are five atoms using symbols that are not the usual symbols of the elements. Use the letters V to Z to answer the following questions. ...
... 17. Listed below are five atoms using symbols that are not the usual symbols of the elements. Use the letters V to Z to answer the following questions. ...
Low-energy electron diffraction

Low-energy electron diffraction (LEED) is a technique for the determination of the surface structure of single-crystalline materials by bombardment with a collimated beam of low energy electrons (20–200 eV) and observation of diffracted electrons as spots on a fluorescent screen.LEED may be used in one of two ways: Qualitatively, where the diffraction pattern is recorded and analysis of the spot positions gives information on the symmetry of the surface structure. In the presence of an adsorbate the qualitative analysis may reveal information about the size and rotational alignment of the adsorbate unit cell with respect to the substrate unit cell. Quantitatively, where the intensities of diffracted beams are recorded as a function of incident electron beam energy to generate the so-called I-V curves. By comparison with theoretical curves, these may provide accurate information on atomic positions on the surface at hand.↑