آهن و TIBC
... can dissociate and react with low-molecular weight compounds such fructose, ascorbic acid, citric acid, amino acids to form ferric complexes soluble in neutral pH of intestine fluid. A protein DMT1 (divalent metal transporter 1), which transports all kinds of divalent metals, then transports the iro ...
... can dissociate and react with low-molecular weight compounds such fructose, ascorbic acid, citric acid, amino acids to form ferric complexes soluble in neutral pH of intestine fluid. A protein DMT1 (divalent metal transporter 1), which transports all kinds of divalent metals, then transports the iro ...
Chapter 3
... ● The oxygen acts as the hydrogen acceptor in the final steps of energy production catalyzed by the flavoproteins and cytochromes. ● Because the use of oxygen generates two toxic molecules, hydrogen peroxide (H2O2) and the free radical superoxide (O2), bacteria require two enzymes to utilize ...
... ● The oxygen acts as the hydrogen acceptor in the final steps of energy production catalyzed by the flavoproteins and cytochromes. ● Because the use of oxygen generates two toxic molecules, hydrogen peroxide (H2O2) and the free radical superoxide (O2), bacteria require two enzymes to utilize ...
understanding bacterial iron transport
... Fax : (44) 18 65 27 51 82 E-mail : [email protected] / [email protected] We present preliminary results of molecular dynamics (MD) simulations on a nanosecond timescale in a fully solvated lipid bilayer to probe the mechanism of the FepA bacterial iron transporter protein. Gram negative bacteria t ...
... Fax : (44) 18 65 27 51 82 E-mail : [email protected] / [email protected] We present preliminary results of molecular dynamics (MD) simulations on a nanosecond timescale in a fully solvated lipid bilayer to probe the mechanism of the FepA bacterial iron transporter protein. Gram negative bacteria t ...
Expression of the Vacuolar Iron Transporter
... investigated the function of a small family of five membrane proteins that have amino acid sequence homology to AtVIT1 and consequently also to yeast CCC1p (1-3). These Vacuolar-Iron-Transporterlike (VTL) genes are found both in mono‐ and dicotyledon plants, as well as in the green alga Chamydomonas ...
... investigated the function of a small family of five membrane proteins that have amino acid sequence homology to AtVIT1 and consequently also to yeast CCC1p (1-3). These Vacuolar-Iron-Transporterlike (VTL) genes are found both in mono‐ and dicotyledon plants, as well as in the green alga Chamydomonas ...
Silke Alt
... Aminocoumarin antibiotics like clorobiocin and novobiocin produced by different Streptomyces strains are potent inhibitors of DNA gyrase. Although novobiocin has been licensed for clinical use in human infections with Gram-positive bacteria such as methicillin-resistant Staphylococcus aureus strains ...
... Aminocoumarin antibiotics like clorobiocin and novobiocin produced by different Streptomyces strains are potent inhibitors of DNA gyrase. Although novobiocin has been licensed for clinical use in human infections with Gram-positive bacteria such as methicillin-resistant Staphylococcus aureus strains ...
File
... Total iron absorption can increase to 3-4 mg/day in pregnant woman Iron in meat is absorbed as such (hemoglobin and myoglobin) Nonheme iron in foods is reduced by ferroreductase to ferrous form A peptide hepcidin produced by liver cells-key central regulator of the system ...
... Total iron absorption can increase to 3-4 mg/day in pregnant woman Iron in meat is absorbed as such (hemoglobin and myoglobin) Nonheme iron in foods is reduced by ferroreductase to ferrous form A peptide hepcidin produced by liver cells-key central regulator of the system ...
The Trace Minerals
... • Constipation, nausea, vomiting, diarrhea • In the U.S. accidental consumption of supplements containing iron is leading cause of poisoning deaths in children under age 6 • Ingestion of as little as 2200mg as been shown to be fatal • The FDA requires warning label on iron supplements that contain 3 ...
... • Constipation, nausea, vomiting, diarrhea • In the U.S. accidental consumption of supplements containing iron is leading cause of poisoning deaths in children under age 6 • Ingestion of as little as 2200mg as been shown to be fatal • The FDA requires warning label on iron supplements that contain 3 ...
Fe-Max Iron Tonic Phytosynergist
... an increased need for iron.3 Women in their childbearing years may also need additional iron so they can make up the blood lost each month in the menstrual flow.1 Some athletes may also require additional iron. Iron supplementation depends on the individual, as not everyone needs extra iron.3 Vitami ...
... an increased need for iron.3 Women in their childbearing years may also need additional iron so they can make up the blood lost each month in the menstrual flow.1 Some athletes may also require additional iron. Iron supplementation depends on the individual, as not everyone needs extra iron.3 Vitami ...
So, you want to know about siderophore synthesis
... Although it has neither hydroxamate nor cathechol groups, Yersiniabactin follows some of the same synthesis pathways, using a nonribosomal peptide sythetase that has clear homologies with, for example, vibriobactin sythetase ...
... Although it has neither hydroxamate nor cathechol groups, Yersiniabactin follows some of the same synthesis pathways, using a nonribosomal peptide sythetase that has clear homologies with, for example, vibriobactin sythetase ...
Diet Recommendations for Hemochromatosis
... Avoid raw shellfish if iron levels are elevated Shellfish can contain a bacterium called Vibrio vulnificus, which can be fatal to people with high body iron levels. Take care when walking barefoot on beaches where contaminated shells may be present. Tea or coffee with meals can reduce the absorption ...
... Avoid raw shellfish if iron levels are elevated Shellfish can contain a bacterium called Vibrio vulnificus, which can be fatal to people with high body iron levels. Take care when walking barefoot on beaches where contaminated shells may be present. Tea or coffee with meals can reduce the absorption ...
arsenic removal by controlled biological iron oxidation reactions
... sulfate from aqueous streams. These technologies find their origin in the exploration of microorganisms involved in the global sulfur cycle. Currently, several sulfur cycle biotechnologies are applied successfully at full-scale. The sulfur cycle is closely linked with the iron cycle, and also the la ...
... sulfate from aqueous streams. These technologies find their origin in the exploration of microorganisms involved in the global sulfur cycle. Currently, several sulfur cycle biotechnologies are applied successfully at full-scale. The sulfur cycle is closely linked with the iron cycle, and also the la ...
Table 3.1 The distribution of body iron. Table 3.2 Iron absorption
... Increased demands (see also Table 3.3) Prematurity Growth Pregnancy Erythropoietin therapy Malabsorption Gluten-induced enteropathy, gastrectomy, autoimmune gastritis Poor diet A major factor in many developing countries but rarely the sole cause in developed countries ...
... Increased demands (see also Table 3.3) Prematurity Growth Pregnancy Erythropoietin therapy Malabsorption Gluten-induced enteropathy, gastrectomy, autoimmune gastritis Poor diet A major factor in many developing countries but rarely the sole cause in developed countries ...
Some application of d block metal in biology
... Inside the sphere, Fe is stored in the Fe(III) oxidation state. It is incorporated in the mineral ferrihydrite, [FeO(OH)]8[FeO(H2PO4)], which is attached to the inner wall of the sphere. (b) To release Fe when the body needs it, the Fe must be changed from the Fe(III) to the Fe(II) oxidation state. ...
... Inside the sphere, Fe is stored in the Fe(III) oxidation state. It is incorporated in the mineral ferrihydrite, [FeO(OH)]8[FeO(H2PO4)], which is attached to the inner wall of the sphere. (b) To release Fe when the body needs it, the Fe must be changed from the Fe(III) to the Fe(II) oxidation state. ...
Iron - University of Minnesota Duluth
... • In Fe2O3 each iron atom has an oxidation number (Ox#) equal to +3 meaning the iron has 26 p+ and only 23 e-. • After smelting, each iron has gained 3 electrons so that #p+ = #e+and iron’s final ...
... • In Fe2O3 each iron atom has an oxidation number (Ox#) equal to +3 meaning the iron has 26 p+ and only 23 e-. • After smelting, each iron has gained 3 electrons so that #p+ = #e+and iron’s final ...
Physiology
... about 65% of which is in the form of Hb. About 4% is in the form of myoglobin, 1% is in the form of the various heme compounds that promote intracellular oxidation, 0.1% is combined with the protein transferrin in the blood plasma, and 15-30% is stored mainly in the reticuloendothelial system and ...
... about 65% of which is in the form of Hb. About 4% is in the form of myoglobin, 1% is in the form of the various heme compounds that promote intracellular oxidation, 0.1% is combined with the protein transferrin in the blood plasma, and 15-30% is stored mainly in the reticuloendothelial system and ...
An Experimental Study into the Clogging of Leachate Collection
... chelation, adsorption, modification of chemical environment around the cell, or by acting as nucleation center for metal precipitation. • Iron and Mn-reducing bacteria ...
... chelation, adsorption, modification of chemical environment around the cell, or by acting as nucleation center for metal precipitation. • Iron and Mn-reducing bacteria ...
File - Andrea Sigrist
... Absorption: Heme iron is more readily absorbed. For example, 95% of the iron in steak is absorbed whereas only 57% of the iron from fortified bread is absorbed. Non-heme iron must first be reduced to the ferrous state. This process can be aided by consuming food rich in vitamin C with non-heme iro ...
... Absorption: Heme iron is more readily absorbed. For example, 95% of the iron in steak is absorbed whereas only 57% of the iron from fortified bread is absorbed. Non-heme iron must first be reduced to the ferrous state. This process can be aided by consuming food rich in vitamin C with non-heme iro ...
Metals in Redox Biology - University of Nebraska–Lincoln
... molecular chaperones. -This interaction is enhanced by HscB, which can bind to both IscU and HscA, leading to a strong enhancement of the intrinsic HscA ATPase activity. -HscA binds to a conserved stretch of amino acids (LPPVK) in IscU. The LPPVK motif is located near a highly conserved Cys (Cys106) ...
... molecular chaperones. -This interaction is enhanced by HscB, which can bind to both IscU and HscA, leading to a strong enhancement of the intrinsic HscA ATPase activity. -HscA binds to a conserved stretch of amino acids (LPPVK) in IscU. The LPPVK motif is located near a highly conserved Cys (Cys106) ...
Lecture 20.The d-Block Elements.VII
... Manganese(IV) oxide is the starting point for making most other manganese compounds. Potassium permanganate, KMnO4, is an important oxidizing agent that is used in both analytical and organic chemistry laboratories, and in water treatment. ...
... Manganese(IV) oxide is the starting point for making most other manganese compounds. Potassium permanganate, KMnO4, is an important oxidizing agent that is used in both analytical and organic chemistry laboratories, and in water treatment. ...
Media:Iron_Broccoli
... Maintained in EE compartments Cycled to membrane for uptake activity Trafficked to vacuole for degradation ...
... Maintained in EE compartments Cycled to membrane for uptake activity Trafficked to vacuole for degradation ...
Structure of an iron-transport protein revealed
... Molecular Biology. Based on these results the researchers were able to explain why DMT1 binds the divalent metal ions iron and manganese (Fe2+ and Mn2+), but not calcium (Ca2+) – in spite of the latter being several orders of magnitude more abundant. Moleclar basis for selective ion transport Struct ...
... Molecular Biology. Based on these results the researchers were able to explain why DMT1 binds the divalent metal ions iron and manganese (Fe2+ and Mn2+), but not calcium (Ca2+) – in spite of the latter being several orders of magnitude more abundant. Moleclar basis for selective ion transport Struct ...
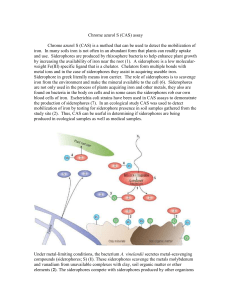
Chrome azurol S (CAS) assay
... To test mobilization of iron, chrome azurol S (CAS) media can be used. On CAS, iron mobilization is done via the production of complexing acids or siderophores. Since siderophore production increases availability of iron, measuring the production of these gives us a measure of iron mobilization abil ...
... To test mobilization of iron, chrome azurol S (CAS) media can be used. On CAS, iron mobilization is done via the production of complexing acids or siderophores. Since siderophore production increases availability of iron, measuring the production of these gives us a measure of iron mobilization abil ...
Power Point Presentation
... • The photo-oxidized ligand continues to bind Fe(III) • Iron bound by these ligands may be more available for uptake, as stability is reduced from original siderophoreReferences ...
... • The photo-oxidized ligand continues to bind Fe(III) • Iron bound by these ligands may be more available for uptake, as stability is reduced from original siderophoreReferences ...
Siderophore

Siderophores (Greek: ""iron carrier"") are small, high-affinity iron chelating compounds secreted by microorganisms such as bacteria, fungi and grasses. Siderophores are amongst the strongest soluble Fe3+ binding agents known.