Unit B Chemistry Unit study guide
... Names, placement and characteristics of Families- Alkali metals, alkali earth metals, transition metals, halogens, noble gases as well as metals vs nonmetals Why are lanthanides and actinides on bottom? What are the only two liquids? Where are the gasses? Which element is in a group of its own? Whic ...
... Names, placement and characteristics of Families- Alkali metals, alkali earth metals, transition metals, halogens, noble gases as well as metals vs nonmetals Why are lanthanides and actinides on bottom? What are the only two liquids? Where are the gasses? Which element is in a group of its own? Whic ...
(the products). Mass is conserved in a chemical reaction
... • Some chemical reactions are slow, some are fast. • For example a car rusting is slow; a matchstick burning is fast. • The reaction rate of a chemical reaction is the amount of reactant consumed per unit time or the amount of product formed per unit time. • It is the “speed” of the reaction. ...
... • Some chemical reactions are slow, some are fast. • For example a car rusting is slow; a matchstick burning is fast. • The reaction rate of a chemical reaction is the amount of reactant consumed per unit time or the amount of product formed per unit time. • It is the “speed” of the reaction. ...
Irreversible Changes
... Most changes that cannot be reversed are chemical reactions where a new material is formed, and it is not possible or extremely difficult to recover the original materials. Children will experience such changes all the time in their everyday life and in the science activities they do in school, but ...
... Most changes that cannot be reversed are chemical reactions where a new material is formed, and it is not possible or extremely difficult to recover the original materials. Children will experience such changes all the time in their everyday life and in the science activities they do in school, but ...
4 hon chem classifying matter b
... decomposed into simpler substances only by chemical means Element – substances that cannot be ...
... decomposed into simpler substances only by chemical means Element – substances that cannot be ...
13.2 Chemical Formulas
... H2O, tells us that a water molecule is made of the elements hydrogen (H) and oxygen (O) and that it takes two atoms of hydrogen and one atom of oxygen to build the molecule. For sodium nitrate, NaNO3, the chemical formula tells us there are three elements in the compound: sodium (Na), nitrogen (N), ...
... H2O, tells us that a water molecule is made of the elements hydrogen (H) and oxygen (O) and that it takes two atoms of hydrogen and one atom of oxygen to build the molecule. For sodium nitrate, NaNO3, the chemical formula tells us there are three elements in the compound: sodium (Na), nitrogen (N), ...
Role of Chemistry in Everyday Life
... series of chemical reactions using enzymes, to change complex chemicals in food to end products that can be absorbed by the body's cells. The caffeine in the coffee and tea we drink affects our mood and keeps us awake. These are all chemical through a chemical change, which is why cooked food often ...
... series of chemical reactions using enzymes, to change complex chemicals in food to end products that can be absorbed by the body's cells. The caffeine in the coffee and tea we drink affects our mood and keeps us awake. These are all chemical through a chemical change, which is why cooked food often ...
File
... Chemical symbols are used in writing chemical formulas, in which the symbols represent the atoms of the elements present in a compound. ...
... Chemical symbols are used in writing chemical formulas, in which the symbols represent the atoms of the elements present in a compound. ...
Notes for Matter Packet- Balancing equations (PDF
... products – New pure substance/s that are made from reactants in a Chemical Reaction ...
... products – New pure substance/s that are made from reactants in a Chemical Reaction ...
1-OCTANESULFONIC ACID SODIUM SALT ANHYDROUS HPLC
... 4.FIRST AID MEASURES: Eyes: rinse with plenty of water as precaution Skin:. Rinse with plenty of soap & water Ingestion: Do not induce vomiting without medical advice. Rinse with water. Inhalation: Move to fresh air. Physician: Treat symptomatically and supportively. 5. FIRE FRIGHTING MEASURES: Info ...
... 4.FIRST AID MEASURES: Eyes: rinse with plenty of water as precaution Skin:. Rinse with plenty of soap & water Ingestion: Do not induce vomiting without medical advice. Rinse with water. Inhalation: Move to fresh air. Physician: Treat symptomatically and supportively. 5. FIRE FRIGHTING MEASURES: Info ...
MSDS - Dudley Chemical Corporation
... pressure-demand, MSHA/NIOSH approved or equivalent, and full protective gear. During a fire, irritating and highly toxic gases may be generated by thermal decomposition or combustion. Vapors may be heavier than air. They can spread along the ground and collect in low or confined areas. Runoff from f ...
... pressure-demand, MSHA/NIOSH approved or equivalent, and full protective gear. During a fire, irritating and highly toxic gases may be generated by thermal decomposition or combustion. Vapors may be heavier than air. They can spread along the ground and collect in low or confined areas. Runoff from f ...
Unit 3C Standards for Quiz
... Unit 2C Standards Quiz on Monday, November 24. It will be similar to the last exam but there will be at least three questions per standard. Remember that since no calculators are allowed on the standards exam that we will be modeling this in this assessment of progress. Atomic and Molecular Structur ...
... Unit 2C Standards Quiz on Monday, November 24. It will be similar to the last exam but there will be at least three questions per standard. Remember that since no calculators are allowed on the standards exam that we will be modeling this in this assessment of progress. Atomic and Molecular Structur ...
CHAPTER 1 Practice Exercises 1.1 12.3 g Cd 1.3 26.9814 u 1.5
... A chemical reaction is a process whereby one or more chemical species is/are transformed into different chemical species. This generally involves the making and/or breaking of chemical bonds. A product is the species formed in a chemical reaction. ...
... A chemical reaction is a process whereby one or more chemical species is/are transformed into different chemical species. This generally involves the making and/or breaking of chemical bonds. A product is the species formed in a chemical reaction. ...
Science24-UnitA-Section3.4
... In Chapter 1 you studied chemical formulas. When chemical formulas are used to represent substances in a reaction, you get a chemical equation. Chemical equations are used to represent all reactions. In mathematics you work with equations. What you do to one side of the equation must be done to the ...
... In Chapter 1 you studied chemical formulas. When chemical formulas are used to represent substances in a reaction, you get a chemical equation. Chemical equations are used to represent all reactions. In mathematics you work with equations. What you do to one side of the equation must be done to the ...
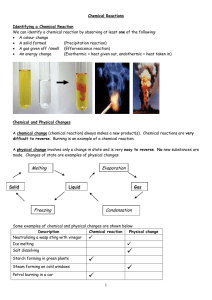
Physical and Chemical Changes
... Example: Hand warmers are an exothermic reaction. Example: Lighting a match is an exothermic reaction. Exothermic reaction ...
... Example: Hand warmers are an exothermic reaction. Example: Lighting a match is an exothermic reaction. Exothermic reaction ...
Discover Chemical Changes - gk-12
... combine to form iron oxide, the red flaky compound we all know as rust. Rotting: a common word for decomposition. which on a chemical level, really means the same as in everyday language, only applied to the molecules that make up something. When something decomposes, the molecules that make up that ...
... combine to form iron oxide, the red flaky compound we all know as rust. Rotting: a common word for decomposition. which on a chemical level, really means the same as in everyday language, only applied to the molecules that make up something. When something decomposes, the molecules that make up that ...
Name: Date: AP Chemistry/Chemistry 145 Summer Assignment
... 16. Limestone, coral, and seashells are composed primarily of solid calcium carbonate. The test for the identification of a carbonate is to use a few drops of aqueous hydrochloric acid. Solid ...
... 16. Limestone, coral, and seashells are composed primarily of solid calcium carbonate. The test for the identification of a carbonate is to use a few drops of aqueous hydrochloric acid. Solid ...
MATTER QUIZ: What to Study From: PHASE CHANGES
... What phase of matter has the highest density? __________________________________ What phase of matter has the most energy? ____________________________________ Does the amount of heat change during a phase change? ____________ temp? _______ Which phase of matter has a definite shape and volume? ____ ...
... What phase of matter has the highest density? __________________________________ What phase of matter has the most energy? ____________________________________ Does the amount of heat change during a phase change? ____________ temp? _______ Which phase of matter has a definite shape and volume? ____ ...
Document
... A. the amount of product formed by a chemical reaction. B. whether or not a specific chemical reaction is possible. C. the coefficients needed to balance a chemical equation. ...
... A. the amount of product formed by a chemical reaction. B. whether or not a specific chemical reaction is possible. C. the coefficients needed to balance a chemical equation. ...
Stoichiometry and Balanced Reactions Chemical Accounting
... • Balanced reaction must have the correct identities of reactants and products and must follow the law of conservation of matter • Reactions are balanced by changing coefficients for each species… NEVER change subscripts in formulas • Typically indicate physical states of reactants and products (s, ...
... • Balanced reaction must have the correct identities of reactants and products and must follow the law of conservation of matter • Reactions are balanced by changing coefficients for each species… NEVER change subscripts in formulas • Typically indicate physical states of reactants and products (s, ...
Chemistry Standard Outline
... SC6a. Compare and contrast atomic/molecular motion in solids, liquids, gases, and plasmas. SC6c. Analyzing (both conceptually and quantitatively) flow of energy during change of state (phase) SC7. Students will characterize the properties that describe solutions and the nature of acids and bases. SC ...
... SC6a. Compare and contrast atomic/molecular motion in solids, liquids, gases, and plasmas. SC6c. Analyzing (both conceptually and quantitatively) flow of energy during change of state (phase) SC7. Students will characterize the properties that describe solutions and the nature of acids and bases. SC ...
Chemical Reactions
... The reactants are separated from each other by a plus sign and the products are separated from each other by a plus sign. There should be an arrow in the middle. Examples: When sodium is mixed with water, a purple alkaline solution of sodium hydroxide is produced and hydrogen gas is evolved. Sodium ...
... The reactants are separated from each other by a plus sign and the products are separated from each other by a plus sign. There should be an arrow in the middle. Examples: When sodium is mixed with water, a purple alkaline solution of sodium hydroxide is produced and hydrogen gas is evolved. Sodium ...
Fundamentals of Chemistry
... – how much reactant is consumed and how much product is formed – coefficients must be consistent with the Law of Conservation of Mass; atoms are neither created nor destroyed in a chemical reaction. – i.e. chemical equation must be balanced ...
... – how much reactant is consumed and how much product is formed – coefficients must be consistent with the Law of Conservation of Mass; atoms are neither created nor destroyed in a chemical reaction. – i.e. chemical equation must be balanced ...
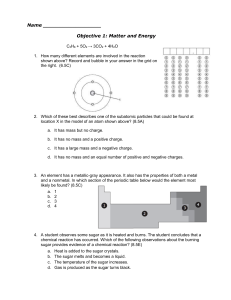
Name Objective 1: Matter and Energy C3H8 + 5O2 → 3CO2 + 4H2O
... b. NO2 and KCl c. 2Li2S and Be4Cl2 d. 2CO and CO2 17. All of the following are indicators of a chemical change except — (8.5E) a. formation of a gas b. change in temperature c. change in the state of matter d. formation of a precipitate 18. Why is the compound CaH10P4K3O4 an inorganic compound? (7.6 ...
... b. NO2 and KCl c. 2Li2S and Be4Cl2 d. 2CO and CO2 17. All of the following are indicators of a chemical change except — (8.5E) a. formation of a gas b. change in temperature c. change in the state of matter d. formation of a precipitate 18. Why is the compound CaH10P4K3O4 an inorganic compound? (7.6 ...
Chemical industry

The chemical industry comprises the companies that produce industrial chemicals. Central to the modern world economy, it converts raw materials (oil, natural gas, air, water, metals, and minerals) into more than 70,000 different products.