Microstructure and humidity sensitive properties of MgFe2O4 ferrite
... environment. Basically, a ceramic sensor can detect humidity on the principle of measuring a change in the resistance by water vapor adsorption. The change in the resistance of the sensor can be explained by the fact that water molecules act as an electron-donating gas [1]. When water molecules are ...
... environment. Basically, a ceramic sensor can detect humidity on the principle of measuring a change in the resistance by water vapor adsorption. The change in the resistance of the sensor can be explained by the fact that water molecules act as an electron-donating gas [1]. When water molecules are ...
The interaction between colloids in polar mixtures above Tc
... same as the solid curve for τ = 0.008 except for one parameter. The effect of charge asymmetry is shown by the dasheddotted curve, where the total charge was kept constant, but σ L = 3σ R . Here the attraction is stronger, as in the classic Poisson-Boltzmann theory,32 since the electrostatic repulsi ...
... same as the solid curve for τ = 0.008 except for one parameter. The effect of charge asymmetry is shown by the dasheddotted curve, where the total charge was kept constant, but σ L = 3σ R . Here the attraction is stronger, as in the classic Poisson-Boltzmann theory,32 since the electrostatic repulsi ...
Slide 1 - scie
... The time taken for a compound to travel through the column to the detector. It is measured from the time the sample is injected to the time its peak shows maximum height. ...
... The time taken for a compound to travel through the column to the detector. It is measured from the time the sample is injected to the time its peak shows maximum height. ...
Soluble - HCC Learning Web
... Solubility Equilibria and the Solubility Product Solubility of salt Calculate the solubility of CaF2 in g/L (Ksp = 4.0 x 10-8) First, write the BALANCED REACTION: ...
... Solubility Equilibria and the Solubility Product Solubility of salt Calculate the solubility of CaF2 in g/L (Ksp = 4.0 x 10-8) First, write the BALANCED REACTION: ...
Redalyc.Chalcopyrite Leaching in Acidic Chloride Solution without
... To determine the effect of dissolved oxygen and its participation in the reactions that take place on the surface of the chalcopyrite, tests were performed in solutions with and without dissolved oxygen. Aerobic tests were carried out by introducing oxygen into the system to maintain a dissolved oxy ...
... To determine the effect of dissolved oxygen and its participation in the reactions that take place on the surface of the chalcopyrite, tests were performed in solutions with and without dissolved oxygen. Aerobic tests were carried out by introducing oxygen into the system to maintain a dissolved oxy ...
Ca(ii), Cd(ii), Cu(ii) and Pb(ii)
... bound. The oxygen atoms from crown ether moiety are ‘‘hard’’ donors having the affinity toward ‘‘hard’’ cations of I and II group of the periodic table of elements.73 The mass spectrum of L with AgNO3 was dominated by the [Ag(I)L]+ complex. Among other investigated metal ions only the silver ion for ...
... bound. The oxygen atoms from crown ether moiety are ‘‘hard’’ donors having the affinity toward ‘‘hard’’ cations of I and II group of the periodic table of elements.73 The mass spectrum of L with AgNO3 was dominated by the [Ag(I)L]+ complex. Among other investigated metal ions only the silver ion for ...
AP Matter Class Packet Unit 5
... Predict the products of and balance the following reactions: ___ HF (aq) + ___ LiOH (aq) → __________ (aq) + ___ HOH (l) ___ HCl (aq) + ___ Ca(OH)2 (aq) → __________(aq) + ___ HOH (l) ___ HClO3 (aq) + ___ Mg(OH)2 (aq) → __________(aq) + ___ HOH (l) ___ H2CO3 (aq) + ___ NaOH (aq) → __________(aq) + _ ...
... Predict the products of and balance the following reactions: ___ HF (aq) + ___ LiOH (aq) → __________ (aq) + ___ HOH (l) ___ HCl (aq) + ___ Ca(OH)2 (aq) → __________(aq) + ___ HOH (l) ___ HClO3 (aq) + ___ Mg(OH)2 (aq) → __________(aq) + ___ HOH (l) ___ H2CO3 (aq) + ___ NaOH (aq) → __________(aq) + _ ...
factors affecting strength of acids
... - A change of 1 unit in pH corresponds to a tenfold change in [H3O+] pH = 3.00 implies [H3O+] = 1.0 x 10-3 M = 0.0010 M pH = 2.00 implies [H3O+] = 1.0 x 10-2 M = 0.010 M which is tenfold - The pH meter and the litmus paper are used to determine pH values of solutions ...
... - A change of 1 unit in pH corresponds to a tenfold change in [H3O+] pH = 3.00 implies [H3O+] = 1.0 x 10-3 M = 0.0010 M pH = 2.00 implies [H3O+] = 1.0 x 10-2 M = 0.010 M which is tenfold - The pH meter and the litmus paper are used to determine pH values of solutions ...
Examples
... to both donate and accept hydrogen ions (not at the same time!!). Substances such as water are described as AMPHOTERIC. ...
... to both donate and accept hydrogen ions (not at the same time!!). Substances such as water are described as AMPHOTERIC. ...
chromapp
... The time taken for a compound to travel through the column to the detector. It is measured from the time the sample is injected to the time its peak shows maximum height. ...
... The time taken for a compound to travel through the column to the detector. It is measured from the time the sample is injected to the time its peak shows maximum height. ...
No Slide Title
... The time taken for a compound to travel through the column to the detector. It is measured from the time the sample is injected to the time its peak shows maximum height. ...
... The time taken for a compound to travel through the column to the detector. It is measured from the time the sample is injected to the time its peak shows maximum height. ...
Option A Materials - Cambridge Resources for the IB Diploma
... Because aluminium is more reactive than carbon, aluminium oxide cannot be reduced to aluminium by heating with carbon and electrolysis must be used. Alumina (aluminium oxide) is an ionic solid made up of Al3+ and O2− ions. In order to conduct electricity, the ions must be free to move. This requires ...
... Because aluminium is more reactive than carbon, aluminium oxide cannot be reduced to aluminium by heating with carbon and electrolysis must be used. Alumina (aluminium oxide) is an ionic solid made up of Al3+ and O2− ions. In order to conduct electricity, the ions must be free to move. This requires ...
structure and thermodynamics of lanthanide
... of a small degree of inner sphere formation, possibly two per cent8. Moreover, it seems that the nitrate ions function as bidentate ligands in these complexes. The thiocyanate complexes of both the actinides and lanthanides have been studied and the thermodynamic and spectral data for the monothiocy ...
... of a small degree of inner sphere formation, possibly two per cent8. Moreover, it seems that the nitrate ions function as bidentate ligands in these complexes. The thiocyanate complexes of both the actinides and lanthanides have been studied and the thermodynamic and spectral data for the monothiocy ...
Equilibrium Expression (Keq)
... (The is a unitless number and is unique to that reaction.) The only thing that can change the value of Keq is a change in temperature. ...
... (The is a unitless number and is unique to that reaction.) The only thing that can change the value of Keq is a change in temperature. ...
Evaluation of Interaction Effect of Sulfate and Chloride Ions on
... Fig. 4 shows the corrosion rates of the electrodes in Solution 1 to 4. The variation of the corrosion rates had same trends with the OCP measurement results. The corrosion rates of the electrodes in Solution 1 were lower than 0.0003 mm/a, suggesting nearly no corrosion occurred on the electrode. Whi ...
... Fig. 4 shows the corrosion rates of the electrodes in Solution 1 to 4. The variation of the corrosion rates had same trends with the OCP measurement results. The corrosion rates of the electrodes in Solution 1 were lower than 0.0003 mm/a, suggesting nearly no corrosion occurred on the electrode. Whi ...
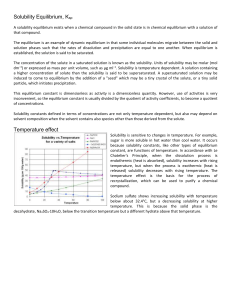
Solubility Equilibria
... The addition of the common ion will shift the solubility equilibrium backwards. This means that there is more solid salt in the solution and therefore the solubility is lower! ...
... The addition of the common ion will shift the solubility equilibrium backwards. This means that there is more solid salt in the solution and therefore the solubility is lower! ...
Uranyl Ion Complexes with Ammoniobenzoates as
... Curiously, instead of another chelating HL3 ligand, two water molecules complete the coordination sphere of U4. U2 and U3 are also bound to one water molecule each, all uranium atoms being thus in pentagonal bipyramidal environments. The average U–O bond lengths for oxo, hydroxo, bridging carboxylat ...
... Curiously, instead of another chelating HL3 ligand, two water molecules complete the coordination sphere of U4. U2 and U3 are also bound to one water molecule each, all uranium atoms being thus in pentagonal bipyramidal environments. The average U–O bond lengths for oxo, hydroxo, bridging carboxylat ...
Useful Rocks - Prairie Rose School Division No. 8
... • Evaporation of this water along with further geological changes led to deposits of rock salt in many countries including the UK. ...
... • Evaporation of this water along with further geological changes led to deposits of rock salt in many countries including the UK. ...
Useful Rocks - We can`t sign you in
... • Evaporation of this water along with further geological changes led to deposits of rock salt in many countries including the UK. ...
... • Evaporation of this water along with further geological changes led to deposits of rock salt in many countries including the UK. ...
Scaling of manganese compounds in kraft mills
... catalytically decomposed by several transition metal ions, viz. Mn2+ and Fe2+, which are easily oxidized in the presence of hydrogen peroxide forming radical species. These radicals may then reduce the metal ions back to the divalent stage while forming oxygen and water. It is, however, possible to ...
... catalytically decomposed by several transition metal ions, viz. Mn2+ and Fe2+, which are easily oxidized in the presence of hydrogen peroxide forming radical species. These radicals may then reduce the metal ions back to the divalent stage while forming oxygen and water. It is, however, possible to ...
CHEMISTRY
... maintain the correct pH compounds containing hydrogencarbonate ions, HCO3-(aq), can be added. i. Write the equations which show how hydrogencarbonate ions can control the pH within the region given. ...
... maintain the correct pH compounds containing hydrogencarbonate ions, HCO3-(aq), can be added. i. Write the equations which show how hydrogencarbonate ions can control the pH within the region given. ...
A simple model for semipermeable membrane: Donnan equilibrium
... By means of integral equations, a previous study of Donnan equilibrium has been carried out by Zhou and Stell [24, 25]. They used the method proposed by Henderson et al. [17], which can be described as follows: starting from a semipermeable spherical cavity [26], the planar membrane is obtained taki ...
... By means of integral equations, a previous study of Donnan equilibrium has been carried out by Zhou and Stell [24, 25]. They used the method proposed by Henderson et al. [17], which can be described as follows: starting from a semipermeable spherical cavity [26], the planar membrane is obtained taki ...
ksp - lozon.ca
... oversaturated sugar solutions, containing other substances such as citric acids. For oversatureated solutions, Qsp is greater than Ksp. When a seed crystal is provided or formed, a precipitate will form immediately due to equilibrium of requiring Qsp to approach Ksp. Sodium acetate trihydrate, NaCH3 ...
... oversaturated sugar solutions, containing other substances such as citric acids. For oversatureated solutions, Qsp is greater than Ksp. When a seed crystal is provided or formed, a precipitate will form immediately due to equilibrium of requiring Qsp to approach Ksp. Sodium acetate trihydrate, NaCH3 ...
electrochemical processing of regeneration solutions from ion
... published work developed removal of sulfate from water (Salnikov et al., 1992) with some approaches using electrolysis (Pisarska et al., 2005). However, the processes of electrolysis of acid and alkaline solutions of sodium sulfate, which are formed during regeneration of ion exchangers by ion-excha ...
... published work developed removal of sulfate from water (Salnikov et al., 1992) with some approaches using electrolysis (Pisarska et al., 2005). However, the processes of electrolysis of acid and alkaline solutions of sodium sulfate, which are formed during regeneration of ion exchangers by ion-excha ...
Mass spectrometry and iccd analysis of coupled and uncoupled mode in a gatling-gun like plasma source
... seven plasma jets arranged adjacent to one another, similar in shape to a Gatling machine gun, was recently developed to take advantage of the jet-to-jet coupling phenomenon and to generate atmospheric pressure cold plasmas with higher intensity and energy with respect to singular plasma jets [1]. T ...
... seven plasma jets arranged adjacent to one another, similar in shape to a Gatling machine gun, was recently developed to take advantage of the jet-to-jet coupling phenomenon and to generate atmospheric pressure cold plasmas with higher intensity and energy with respect to singular plasma jets [1]. T ...
Ionic compound

In chemistry, an ionic compound is a chemical compound in which ions are held together in a structure by electrostatic forces termed ionic bonds. The positively charged ions are called cations and the negatively charged ions are called anions. These can be simple ions such as the sodium (Na+) and chloride (Cl−) in sodium chloride, or polyatomic species such as the carbonate ion (CO32−) in calcium carbonate. Individual ions within an ionic compound usually have multiple nearest neighbours, so are not considered to be part of molecules, but instead part of a continuous three-dimensional network, usually in a crystalline structure.Ionic compounds typically have high melting and boiling points, and are hard and brittle. As solids they are almost always electrically insulating, but when melted or dissolved they become highly conductive, because the ions are mobilized.Ionic compounds without the acidic hydrogen ion (H+), or the basic ions hydroxide (OH−) or oxide (O2−), are also known as salts and can be formed by acid-base reactions. Ionic compounds containing hydrogen ions are classified as acids and compounds containing hydroxide or oxide ions are classified as bases.