Influence of Ionic Mobile Phase Additives with Low Charge
... Ionic suppression technique is based on the pH adjustment of the mobile phase which results in a non-ionized analyte. However, this adjustment of pH is only suitable for single compounds or simple mixtures of bases where the pKa's ofthe compounds are close together4• ...
... Ionic suppression technique is based on the pH adjustment of the mobile phase which results in a non-ionized analyte. However, this adjustment of pH is only suitable for single compounds or simple mixtures of bases where the pKa's ofthe compounds are close together4• ...
Effect of N-donor ancillary ligands on structural and magnetic
... interactions between metal atoms and organic ligand moieties.2 Oxalate ions have been proven to be good connectors to bind to metal joints and play a key role in the design of new functional coordination networks. They have presented highly versatile binding modes (Scheme 1) and also serve as good h ...
... interactions between metal atoms and organic ligand moieties.2 Oxalate ions have been proven to be good connectors to bind to metal joints and play a key role in the design of new functional coordination networks. They have presented highly versatile binding modes (Scheme 1) and also serve as good h ...
Evidence for a Post-Translational Modification, Aspartyl Aldehyde, in
... both subunits have been shown to interact with the oxygenevolving site (reviewed in ref 3). Extrinsic polypeptides also associate with the oxygen-evolving complex and are important for active-site stabilization and inorganic cofactor retention.4 Biochemical evidence suggests that there are three car ...
... both subunits have been shown to interact with the oxygenevolving site (reviewed in ref 3). Extrinsic polypeptides also associate with the oxygen-evolving complex and are important for active-site stabilization and inorganic cofactor retention.4 Biochemical evidence suggests that there are three car ...
Small Dopant Effect on Static Grain Growth and Flow Stress in
... and the static grain growth in cation-doped 3Y-TZP. The diffusivity in cation-doped TZP, which is related to the accommodation process for the flow stress and the grain growth behavior, can be evaluated from the activation energy as a function of dopant cation’s ionic size. In our previous report, it ...
... and the static grain growth in cation-doped 3Y-TZP. The diffusivity in cation-doped TZP, which is related to the accommodation process for the flow stress and the grain growth behavior, can be evaluated from the activation energy as a function of dopant cation’s ionic size. In our previous report, it ...
Synthesis and Characterization of Tetradentate Complexes Type
... Amino oxime metal complexes have been known since a long time (1). The different coordination modes of oxime and oximato sp ecies indicated a versatile electronic distribution within the ligand. This, in turn suggested that the chemistry of metal-bonded oximes should be rich. The inspection of data ...
... Amino oxime metal complexes have been known since a long time (1). The different coordination modes of oxime and oximato sp ecies indicated a versatile electronic distribution within the ligand. This, in turn suggested that the chemistry of metal-bonded oximes should be rich. The inspection of data ...
Philicities, Fugalities, and Equilibrium Constants
... relationships between rate and equilibrium constants. Thus, strong bases are generally considered to be good nucleophiles and poor nucleofuges. Exceptions to this rule have long been known, and the ability of iodide ions to catalyze nucleophilic substitutions, because they are good nucleophiles as w ...
... relationships between rate and equilibrium constants. Thus, strong bases are generally considered to be good nucleophiles and poor nucleofuges. Exceptions to this rule have long been known, and the ability of iodide ions to catalyze nucleophilic substitutions, because they are good nucleophiles as w ...
cmc by uv 2
... mimetics), and pharmacy (1). Surfactants are amphiphilic materials containing both apolar long-chain hydrocarbon “tail” and polar, usually ionic, “head” groups. In polar solvents, for example water, this dual character of the amphiphile leads to self-association or micellization: the surfactant mole ...
... mimetics), and pharmacy (1). Surfactants are amphiphilic materials containing both apolar long-chain hydrocarbon “tail” and polar, usually ionic, “head” groups. In polar solvents, for example water, this dual character of the amphiphile leads to self-association or micellization: the surfactant mole ...
міністерство освіти і науки україни
... the shell. These elements are metals. When metal atoms bond, a metallic bond occurs. When an atom has a nearly full electron shell, it will try to find electrons from another atom so that it can fill its outer shell. These elements are usually described as nonmetals. The bond between two nonmetal at ...
... the shell. These elements are metals. When metal atoms bond, a metallic bond occurs. When an atom has a nearly full electron shell, it will try to find electrons from another atom so that it can fill its outer shell. These elements are usually described as nonmetals. The bond between two nonmetal at ...
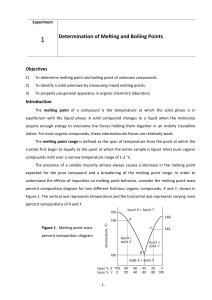
Making Sense of Boiling Points and Melting Points
... Among the molecular covalent compounds (most organic compounds come under this category), it is possible, to some extent, to rationalize the trends in these physical constants based on noncovalent interactions and symmetry, which constitute the contents of this article. If molecules have only paired ...
... Among the molecular covalent compounds (most organic compounds come under this category), it is possible, to some extent, to rationalize the trends in these physical constants based on noncovalent interactions and symmetry, which constitute the contents of this article. If molecules have only paired ...
Alkyl Chain Length Dependence of the Dynamics and Structure in
... though minor, are an indication that the chain length does have some impact on the structure of the ionic regions, resulting in changes in the intermolecular interactions between the SeCN− vibrational probe and its environment. Infrared PSPP experiments were used to measure the vibrational lifetimes ...
... though minor, are an indication that the chain length does have some impact on the structure of the ionic regions, resulting in changes in the intermolecular interactions between the SeCN− vibrational probe and its environment. Infrared PSPP experiments were used to measure the vibrational lifetimes ...
Carbon–carbon bond cleavage in the photoionization of ethanol and
... long field-free flight tube. The ions were detected by a chevron microchannel plate 共MCP兲 detector. After amplification of the signal by a fast preamplifier, the mass spectrum was recorded by a digital oscilloscope and an ion counter. The VUV laser beam was generated either by frequency tripling of ...
... long field-free flight tube. The ions were detected by a chevron microchannel plate 共MCP兲 detector. After amplification of the signal by a fast preamplifier, the mass spectrum was recorded by a digital oscilloscope and an ion counter. The VUV laser beam was generated either by frequency tripling of ...
Ch. 14 Study Guide
... 20. If an equilibrium cannot be established, then the solution is unsaturated. 21. Supersaturated solutions hold more solute than is theoretically possible. 22. Generally, higher temperatures result in higher solubility of solids. 23. Henry’s Law states that if we push a gas hard enough (with extern ...
... 20. If an equilibrium cannot be established, then the solution is unsaturated. 21. Supersaturated solutions hold more solute than is theoretically possible. 22. Generally, higher temperatures result in higher solubility of solids. 23. Henry’s Law states that if we push a gas hard enough (with extern ...
Pass Assured`s Pharmacy Technician Training Program
... Milliequivalents (mEq) A milliequivalent is equal to the number of univalent counter ions (H+ or OH) which will be needed to react with one molecule of the substance When an atom has a valence of one ( e.g. Na+, K+, CI, etc.) a mEq of that ion is equal to the atomic weight of the atom in milligrams. ...
... Milliequivalents (mEq) A milliequivalent is equal to the number of univalent counter ions (H+ or OH) which will be needed to react with one molecule of the substance When an atom has a valence of one ( e.g. Na+, K+, CI, etc.) a mEq of that ion is equal to the atomic weight of the atom in milligrams. ...
Exam 1 Goals
... c. Important chemical features of EDTA i. 7 different acid-base forms ii. 6 different pK values iii. The most basic form (Y4-) primarily reacts with metal ion iv. The fractional composition () of Y4- is strongly pH dependent v. EDTA binds tightly to multiple metal ions a) Kf is modulated by pH 1. m ...
... c. Important chemical features of EDTA i. 7 different acid-base forms ii. 6 different pK values iii. The most basic form (Y4-) primarily reacts with metal ion iv. The fractional composition () of Y4- is strongly pH dependent v. EDTA binds tightly to multiple metal ions a) Kf is modulated by pH 1. m ...
... atomic mass and as a tool for determination of operation of a magnetic sector mass spectrometer; molecular structure (see also section 2.10). recognize and interpret a simple mass spectrum of an atomic and a molecular sample, explaining the peak height ratios for the mass spectra of Cl2 and Br2, and ...
1 AM SYLLABUS (2016) CHEMISTRY AM 06 SYLLABUS
... atomic mass and as a tool for determination of operation of a magnetic sector mass spectrometer; molecular structure (see also section 2.10). recognize and interpret a simple mass spectrum of an atomic and a molecular sample, explaining the peak height ratios for the mass spectra of Cl2 and Br2, and ...
... atomic mass and as a tool for determination of operation of a magnetic sector mass spectrometer; molecular structure (see also section 2.10). recognize and interpret a simple mass spectrum of an atomic and a molecular sample, explaining the peak height ratios for the mass spectra of Cl2 and Br2, and ...
Solutions - WordPress.com
... universal solvent • The term aqueous or (aq) describes a solution in which the solvent is water • Not all solutes and solvents are liquid! ▫ Brass (zinc and nickel) ▫ Seawater (salt and other substances in water) ▫ Air (Many different gasses in Nitrogen) ...
... universal solvent • The term aqueous or (aq) describes a solution in which the solvent is water • Not all solutes and solvents are liquid! ▫ Brass (zinc and nickel) ▫ Seawater (salt and other substances in water) ▫ Air (Many different gasses in Nitrogen) ...
1 AM SYLLABUS (2015) CHEMISTRY AM 06 SYLLABUS
... draw the electronic configuration of isolated atoms of elements H to Kr using 1s, 2s, 2p etc notation and electrons- in-boxes notation and applying the ‘building– up’ (aufbau) principle. Questions on interpretation of atomic spectra will not be set although students are expected to explain how trans ...
... draw the electronic configuration of isolated atoms of elements H to Kr using 1s, 2s, 2p etc notation and electrons- in-boxes notation and applying the ‘building– up’ (aufbau) principle. Questions on interpretation of atomic spectra will not be set although students are expected to explain how trans ...
190 - Chimica
... (H2O)–bpethy–(H2O)–Cu, of length 17.59 Å. Taking into accout these interactions nets II and III together can be described as an unique tridimensional five-connected array, interpenetrated by the four-connected net I. Interpenetration phenomena involving different types of nets are quite rare; we hav ...
... (H2O)–bpethy–(H2O)–Cu, of length 17.59 Å. Taking into accout these interactions nets II and III together can be described as an unique tridimensional five-connected array, interpenetrated by the four-connected net I. Interpenetration phenomena involving different types of nets are quite rare; we hav ...
Chapter 6 One-Electron Reduction Potentials of Aqueous Co2+
... the same aqueous nanodrop, whereas Cr+, Mn+, Fe+, Co+, Ni+, and Cu+ do not change their oxidation states and instead form a metal chloride (MCl) “precipitate,” consistent with the propensity of these metal ions to be oxidized in solution.32 Cluster measurements can also be extrapolated to infinite s ...
... the same aqueous nanodrop, whereas Cr+, Mn+, Fe+, Co+, Ni+, and Cu+ do not change their oxidation states and instead form a metal chloride (MCl) “precipitate,” consistent with the propensity of these metal ions to be oxidized in solution.32 Cluster measurements can also be extrapolated to infinite s ...
Ksp Problem Sets 1 and 2
... 4. The solubility product constant for lead (II) chloride is 1.7 x 10 . How many moles of lead nitrate can dissolve in 1.00 L of a 0.1 M solution of sodium chloride? (Hint: One of the ions found in PbCl2, the chloride ion, is already present in the solution. That can be taken to be its equilibrium ...
... 4. The solubility product constant for lead (II) chloride is 1.7 x 10 . How many moles of lead nitrate can dissolve in 1.00 L of a 0.1 M solution of sodium chloride? (Hint: One of the ions found in PbCl2, the chloride ion, is already present in the solution. That can be taken to be its equilibrium ...
4 Types of Chemical Reactions and Solution Stoichiometry
... blood and other body fluids. In addition to the common tests for sugar, cholesterol, and iron, analyses for specific chemical markers allow detection of many diseases before more obvious symptoms occur. Aqueous chemistry is also important in our environment. In recent years contamination of the grou ...
... blood and other body fluids. In addition to the common tests for sugar, cholesterol, and iron, analyses for specific chemical markers allow detection of many diseases before more obvious symptoms occur. Aqueous chemistry is also important in our environment. In recent years contamination of the grou ...
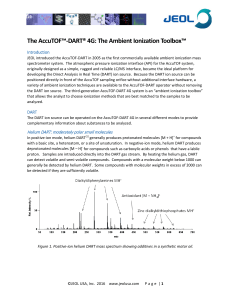
The Ambient Ionization Toolbox
... JEOL introduced the AccuTOF-DART in 2005 as the first commercially available ambient ionization mass spectrometer system. The atmospheric pressure ionization interface (API) for the AccuTOF system, originally designed as a simple, rugged and reliable LC/MS interface, became the ideal platform for de ...
... JEOL introduced the AccuTOF-DART in 2005 as the first commercially available ambient ionization mass spectrometer system. The atmospheric pressure ionization interface (API) for the AccuTOF system, originally designed as a simple, rugged and reliable LC/MS interface, became the ideal platform for de ...
Ionic compound

In chemistry, an ionic compound is a chemical compound in which ions are held together in a structure by electrostatic forces termed ionic bonds. The positively charged ions are called cations and the negatively charged ions are called anions. These can be simple ions such as the sodium (Na+) and chloride (Cl−) in sodium chloride, or polyatomic species such as the carbonate ion (CO32−) in calcium carbonate. Individual ions within an ionic compound usually have multiple nearest neighbours, so are not considered to be part of molecules, but instead part of a continuous three-dimensional network, usually in a crystalline structure.Ionic compounds typically have high melting and boiling points, and are hard and brittle. As solids they are almost always electrically insulating, but when melted or dissolved they become highly conductive, because the ions are mobilized.Ionic compounds without the acidic hydrogen ion (H+), or the basic ions hydroxide (OH−) or oxide (O2−), are also known as salts and can be formed by acid-base reactions. Ionic compounds containing hydrogen ions are classified as acids and compounds containing hydroxide or oxide ions are classified as bases.