Tunneling through a Barrier
... and its slope must be continuous at the edges of the barrier. The conditions for continuity enable us to connect the wavefunctions in the three zones and hence to obtain relations between the coefficients that appear in the solutions of the Schrödinger equation. ...
... and its slope must be continuous at the edges of the barrier. The conditions for continuity enable us to connect the wavefunctions in the three zones and hence to obtain relations between the coefficients that appear in the solutions of the Schrödinger equation. ...
The absorption spectra of very small CdS or ZnS particles (1
... small as can be recognized from the onset of the absorption well below 510 nm . All three fluorescence bands mentioned above are present , the first one appearing at 480 nm , i . e . at the onset of the absorption . This band is attributed to the fluorescence from the exciton state. The strong band ...
... small as can be recognized from the onset of the absorption well below 510 nm . All three fluorescence bands mentioned above are present , the first one appearing at 480 nm , i . e . at the onset of the absorption . This band is attributed to the fluorescence from the exciton state. The strong band ...
June 2010 Regents Exam Part C Questions
... (2) absorb energy as they move to higher energy states (3) release energy as they move to lower energy states (4) release energy as they move to higher energy states Q8 An atom of which element has the greatest attraction for electrons in a chemical bond? ( note Q8-10 are on Bonding ; see items ...
... (2) absorb energy as they move to higher energy states (3) release energy as they move to lower energy states (4) release energy as they move to higher energy states Q8 An atom of which element has the greatest attraction for electrons in a chemical bond? ( note Q8-10 are on Bonding ; see items ...
Environmental Analysis - Evergreen State College Archives
... American Society for Testing and Materials (ASTM). These methods are specific for use with certain analytes in certain types of samples. They commonly define both the sample handling requirements and the analytical techniques that are to be used. Some methods require calibration. This involves makin ...
... American Society for Testing and Materials (ASTM). These methods are specific for use with certain analytes in certain types of samples. They commonly define both the sample handling requirements and the analytical techniques that are to be used. Some methods require calibration. This involves makin ...
Volatility of Organic Aerosol: Evaporation of Ammonium Sulfate
... product of the activity coefficient and the molar fraction of the given compound in the particle. For aqueous solutions of single organic compounds directly measurement-based activity models9 and models based on group contribution methods, like UNIFAC,10 are available. The latter can also be applied f ...
... product of the activity coefficient and the molar fraction of the given compound in the particle. For aqueous solutions of single organic compounds directly measurement-based activity models9 and models based on group contribution methods, like UNIFAC,10 are available. The latter can also be applied f ...
CHEM-212 Eggshell Lab - Winona State University
... This reaction can be used to determine the mass percentage of CaCO3 in an eggshell sample. This can be accomplished in two ways. The portion of the eggshell that is not CaCO3 does not react with acid and remains as a solid. This solid can be filtered from the solution and weighed to give the mass of ...
... This reaction can be used to determine the mass percentage of CaCO3 in an eggshell sample. This can be accomplished in two ways. The portion of the eggshell that is not CaCO3 does not react with acid and remains as a solid. This solid can be filtered from the solution and weighed to give the mass of ...
Literacy - TECC Science
... Read the following passage and then answer the questions that follow. Nanoparticles Nanoparticles are very small particles that are between 1 and 100 nm in size. They can also be described as being ultra-fine particles. Nanotechnology is the use of nanoparticles to help improve people’s lives. Nanop ...
... Read the following passage and then answer the questions that follow. Nanoparticles Nanoparticles are very small particles that are between 1 and 100 nm in size. They can also be described as being ultra-fine particles. Nanotechnology is the use of nanoparticles to help improve people’s lives. Nanop ...
syllabus - WordPress.com
... In a colloidal sol, the dispersed phase is a solid and the dispersion medium is a liquid. In a gel, it is opposite. In emulsion, both the dispersed phase and dispersion medium are liquids. Explain the term: Peptization. Peptization may be defined as the process of converting a precipitate into collo ...
... In a colloidal sol, the dispersed phase is a solid and the dispersion medium is a liquid. In a gel, it is opposite. In emulsion, both the dispersed phase and dispersion medium are liquids. Explain the term: Peptization. Peptization may be defined as the process of converting a precipitate into collo ...
homework assignment 2 - the Petersen Home Page
... 1. A 15.40-g sample of a finely-divided mixture of only Fe2S3 and FeS was reacted with excess H2 at elevated temperatures. If the weight percent of Fe2S3 in this mixture is 57.4%, then calculate the total mass in grams of Fe that can be produced. Assume the only other product of these reactions is H ...
... 1. A 15.40-g sample of a finely-divided mixture of only Fe2S3 and FeS was reacted with excess H2 at elevated temperatures. If the weight percent of Fe2S3 in this mixture is 57.4%, then calculate the total mass in grams of Fe that can be produced. Assume the only other product of these reactions is H ...
Figure 4 - University of Wisconsin–Madison
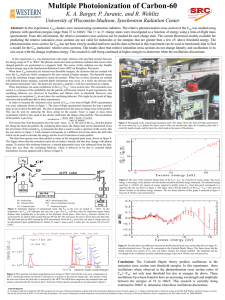
... predicting the relative ionization cross sections are not valid for molecules or for energies that are greater then a few eV above threshold energy. The photoionization cross section for C60 has not been closely studied until now and the data collected in this experiment can be used as benchmark dat ...
... predicting the relative ionization cross sections are not valid for molecules or for energies that are greater then a few eV above threshold energy. The photoionization cross section for C60 has not been closely studied until now and the data collected in this experiment can be used as benchmark dat ...
Pulsed-laser ablation of Mg in liquids: surfactant
... and temperature due to adiabatic expansion and creation of a shock wave at the plasma–liquid interface [22,23]. However, it is known that excited electrons will also transfer energy to the metal lattice by typical electron–phonon coupling. The electron-electron and electron-phonon relaxation times a ...
... and temperature due to adiabatic expansion and creation of a shock wave at the plasma–liquid interface [22,23]. However, it is known that excited electrons will also transfer energy to the metal lattice by typical electron–phonon coupling. The electron-electron and electron-phonon relaxation times a ...
maxwell distribution of gas molecule velocities
... Outline the procedure for finding the maximum of a function like the Maxwell distribution. Let MATHCAD help you do it. Although doing the operations by hand is not too difficult there are many places where error can occur. A modern tool like MATHCAD helps you to do things like this more accurately a ...
... Outline the procedure for finding the maximum of a function like the Maxwell distribution. Let MATHCAD help you do it. Although doing the operations by hand is not too difficult there are many places where error can occur. A modern tool like MATHCAD helps you to do things like this more accurately a ...
Chapter 2
... • Energy can not be created or destroyed (The law of conservation of energy) • Energy is either used to do work on the molecules, moving, them out of the crystal lattice at the melting point, or moving them farther from each other into gas state at the boiling point. ...
... • Energy can not be created or destroyed (The law of conservation of energy) • Energy is either used to do work on the molecules, moving, them out of the crystal lattice at the melting point, or moving them farther from each other into gas state at the boiling point. ...
Cellular Automata and Lattice Gases
... that can perform the local cell updates can execute the same global programs [Gershenfeld et al., 2010]. Alternatively, since CAs can model physical systems, and physical systems can compute, CAs can model physical systems that compute [Margolus, 1984]. Consider the billiard-ball CA in Figure 10.4 ( ...
... that can perform the local cell updates can execute the same global programs [Gershenfeld et al., 2010]. Alternatively, since CAs can model physical systems, and physical systems can compute, CAs can model physical systems that compute [Margolus, 1984]. Consider the billiard-ball CA in Figure 10.4 ( ...
18 - cloudfront.net
... 11.5 second. She runs at a speed of 8.70 m/s. Another sprinter takes 15.0 seconds to cover the same distance. She runs at a speed of 6.67 m/s. Both 8.70 m/s and 6.67 m/s express rates of travel. A rate is a measure of speed of any change that occurs within an interval of time. The interval of time m ...
... 11.5 second. She runs at a speed of 8.70 m/s. Another sprinter takes 15.0 seconds to cover the same distance. She runs at a speed of 6.67 m/s. Both 8.70 m/s and 6.67 m/s express rates of travel. A rate is a measure of speed of any change that occurs within an interval of time. The interval of time m ...
〈1181〉 SCANNING ELECTRON MICROSCOPY
... The size of the final aperture controls the beam diameter and, accordingly, the image resolution and total current at a specimen. Small apertures are required for high resolution and large apertures provide high current for optimal X-ray emission intensity. In many systems, the objective aperture ca ...
... The size of the final aperture controls the beam diameter and, accordingly, the image resolution and total current at a specimen. Small apertures are required for high resolution and large apertures provide high current for optimal X-ray emission intensity. In many systems, the objective aperture ca ...
Metathesis Problems (and Some Solutions) Identified Through
... molecular solutions even at the same molarity or molality – Ionic compounds dissociate into individual ions • i = number of ions resulting from solvation of one formula unit • Multiply i in any colligative formula if the solute is ionic ∆Tf = iKfcm ∆P = iP°AXB ∆Tb = iKbcm π = iMRT • i is only accura ...
... molecular solutions even at the same molarity or molality – Ionic compounds dissociate into individual ions • i = number of ions resulting from solvation of one formula unit • Multiply i in any colligative formula if the solute is ionic ∆Tf = iKfcm ∆P = iP°AXB ∆Tb = iKbcm π = iMRT • i is only accura ...
key to sample questions test 2
... Which of the diagrams above corresponds to a molecule with a triple bond? __D____ Which of the diagrams above corresponds molecule with no net bonds? ___C_____ Which of the diagrams above could correspond to a N2 molecule? ____D____ Which of the diagrams above corresponds to a paramagnetic molecule? ...
... Which of the diagrams above corresponds to a molecule with a triple bond? __D____ Which of the diagrams above corresponds molecule with no net bonds? ___C_____ Which of the diagrams above could correspond to a N2 molecule? ____D____ Which of the diagrams above corresponds to a paramagnetic molecule? ...
I 1 Chemical Reaction Cross Sections I and Rate Constants
... sense to indicate that two particles come close enough together to interact, i.e. they approach to a distance at which the forces between them are different from zero. Thus contact between the hard cores is not a necessary condition for a collision of A with B if the interaction is given by eqn. (23 ...
... sense to indicate that two particles come close enough together to interact, i.e. they approach to a distance at which the forces between them are different from zero. Thus contact between the hard cores is not a necessary condition for a collision of A with B if the interaction is given by eqn. (23 ...
Effects of antioxidants for the degradation of flame
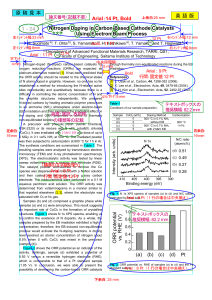
... 6 MGy in 0.1 vol% NH3 at 500 °C. The irradiated powder was then subjected to carbonization at 800 °C for 1 h in Ar. The synthesis conditions are summarized in Table 1. The resulting samples were analyzed by transmission electron microscopy (TEM) and X-ray photoelectron spectroscopy (XPS). The electr ...
... 6 MGy in 0.1 vol% NH3 at 500 °C. The irradiated powder was then subjected to carbonization at 800 °C for 1 h in Ar. The synthesis conditions are summarized in Table 1. The resulting samples were analyzed by transmission electron microscopy (TEM) and X-ray photoelectron spectroscopy (XPS). The electr ...
Chapter 2 What Is Matter
... If a mixture contains large particles dispersed in a liquid or a gas that will settle out, it is classified as a suspension. If the particles of a suspension are not stirred constantly, they will settle out. Particles of a suspension can also be filtered out. The particles are large enough to be ca ...
... If a mixture contains large particles dispersed in a liquid or a gas that will settle out, it is classified as a suspension. If the particles of a suspension are not stirred constantly, they will settle out. Particles of a suspension can also be filtered out. The particles are large enough to be ca ...
PowerPoint Chapter 14 - Preparatory Chemistry
... Liquids: Condensation, Evaporation, and Dynamic ...
... Liquids: Condensation, Evaporation, and Dynamic ...
II. Classification of Matter
... Law vs. theory Scientific (natural) _____________: a general statement based on the observed behavior of matter to which no exceptions are known. __________________: a broad generalization that explains a body of facts or phenomena. Quantitative vs. qualitative data Quantitative: numerical (________ ...
... Law vs. theory Scientific (natural) _____________: a general statement based on the observed behavior of matter to which no exceptions are known. __________________: a broad generalization that explains a body of facts or phenomena. Quantitative vs. qualitative data Quantitative: numerical (________ ...
CHEMONE Directions: Select the letter of the best
... 34. Which of the following statements about liquid is true? a. Droplet formation occurs because of the higher stability associated with increased surface area. b. Substances that can form hydrogen bonds will display low melting points than predicted from periodic trends. c. London dispersion forces ...
... 34. Which of the following statements about liquid is true? a. Droplet formation occurs because of the higher stability associated with increased surface area. b. Substances that can form hydrogen bonds will display low melting points than predicted from periodic trends. c. London dispersion forces ...
Evaluation of toxicity of nanoclays and graphene oxide: in vivo A
... Abstract © The Royal Society of Chemistry 2016.We report here the successful use of a protozoan model organism P. caudatum to investigate the toxicity of clay nanoparticles (montmorillonite, halloysite, kaolin, and bentonite), silica nanospheres and graphene oxide nanoflakes. The distribution of nan ...
... Abstract © The Royal Society of Chemistry 2016.We report here the successful use of a protozoan model organism P. caudatum to investigate the toxicity of clay nanoparticles (montmorillonite, halloysite, kaolin, and bentonite), silica nanospheres and graphene oxide nanoflakes. The distribution of nan ...
Particle-size distribution
The particle-size distribution (PSD) of a powder, or granular material, or particles dispersed in fluid, is a list of values or a mathematical function that defines the relative amount, typically by mass, of particles present according to size. PSD is also known as grain size distribution.