Stockholm University
... structure of C-peptide at lower pH. Also at pH 3.2, C-peptide is predominantly in a random coil conformation and our results indicate that no major structural rearrangements are induced as a consequence of adding POPC or POPC/POPG 1 : 1, respectively (Figure 1(a)). The lack of structural induction i ...
... structure of C-peptide at lower pH. Also at pH 3.2, C-peptide is predominantly in a random coil conformation and our results indicate that no major structural rearrangements are induced as a consequence of adding POPC or POPC/POPG 1 : 1, respectively (Figure 1(a)). The lack of structural induction i ...
2014 - icnirp
... Overall, these results indicated that the threshold of vertigo correlated somewhat better with DB than with the peak dB/dt and that the most effective frequency range was below 1 Hz. It is a common experience from working with clinical MR imaging that vertigo sensations disappear when movement is sl ...
... Overall, these results indicated that the threshold of vertigo correlated somewhat better with DB than with the peak dB/dt and that the most effective frequency range was below 1 Hz. It is a common experience from working with clinical MR imaging that vertigo sensations disappear when movement is sl ...
Magnetite in human tissues - Geological and Planetary Sciences
... noise. Even though biological systems excel at non-linear amplification, non-linear effects cannot short-circuit the laws of thermodynamics, or we would be able to build perpetual motion machines. Thermal noise amplified by any system is still thermal noise; however, processes which average over lar ...
... noise. Even though biological systems excel at non-linear amplification, non-linear effects cannot short-circuit the laws of thermodynamics, or we would be able to build perpetual motion machines. Thermal noise amplified by any system is still thermal noise; however, processes which average over lar ...
GENERAL INTRODUCTION 1. Introduction
... the ELF range of 3–3000 Hz (IEEE, 1988). Ancillary ELF phenomena, such as transients, which have frequency components in excess of 3000 Hz have also been examined for their potential contribution to the possible hazard of ELF radiation. At frequencies above those of interest here, electromagnetic fi ...
... the ELF range of 3–3000 Hz (IEEE, 1988). Ancillary ELF phenomena, such as transients, which have frequency components in excess of 3000 Hz have also been examined for their potential contribution to the possible hazard of ELF radiation. At frequencies above those of interest here, electromagnetic fi ...
measurement and analysis of electromagnetic fields from trams
... provide clear information about these effects. Managing health issues in relation to the transportation systems is presently at an early stage of deployment. This clarification significantly changes the range of frequencies to be considered for ‘transportation system EMFs’, producing open-ended ques ...
... provide clear information about these effects. Managing health issues in relation to the transportation systems is presently at an early stage of deployment. This clarification significantly changes the range of frequencies to be considered for ‘transportation system EMFs’, producing open-ended ques ...
chapter 24 design of magnetic resonance systems
... Atomic nuclei containing odd numbers of nucleons (i.e., protons and neutrons) have magnetic moments. Hydrogen (1H) nuclei (protons) have the highest magnetic moment of any nuclei and are the most abundant nuclei in biological materials. To obtain high signal-to-noise ratios, hydrogen nuclei are typi ...
... Atomic nuclei containing odd numbers of nucleons (i.e., protons and neutrons) have magnetic moments. Hydrogen (1H) nuclei (protons) have the highest magnetic moment of any nuclei and are the most abundant nuclei in biological materials. To obtain high signal-to-noise ratios, hydrogen nuclei are typi ...
ABSTRACT - Harlem Children Society
... were synthesized to observe their imitative relevance for fabricating the structure and reactivity of metallocysteinate proteins that are involved in metal transport. These ligands were used to observe if the type of reaction that takes place between them and the metal cations, which are Zinc (Zn2+ ...
... were synthesized to observe their imitative relevance for fabricating the structure and reactivity of metallocysteinate proteins that are involved in metal transport. These ligands were used to observe if the type of reaction that takes place between them and the metal cations, which are Zinc (Zn2+ ...
Magnet Theory
... electromagnetic forces in the sub atomic level, we are not only surrounded by positive and negative electromagnetic forces but every cell in our bodies relies upon electromagnetism for its very existence. “All biological systems are electromagnetic. Magnetic and electric poles cannot be separated. E ...
... electromagnetic forces in the sub atomic level, we are not only surrounded by positive and negative electromagnetic forces but every cell in our bodies relies upon electromagnetism for its very existence. “All biological systems are electromagnetic. Magnetic and electric poles cannot be separated. E ...
Slide 1
... field, with a short square pulse of radio-frequency energy containing all the frequencies in a given range of interest. • The polarized magnets of the nuclei begin to spin together, creating a radio frequency (RF) that is observable. Because the signals decays over time, this time-dependent pattern ...
... field, with a short square pulse of radio-frequency energy containing all the frequencies in a given range of interest. • The polarized magnets of the nuclei begin to spin together, creating a radio frequency (RF) that is observable. Because the signals decays over time, this time-dependent pattern ...
Physics 228 Today: April 22, 2012 Ch. 43 Nuclear
... It is a good starting approximation that the mass of a nucleus is simply proportional to the number of nucleons in the nucleus, A. But the nucleons are bound into the nucleus, so the mass of the nucleus is less than the sum of the masses of the constituent nucleons. The mass of a nucleus is roughly ...
... It is a good starting approximation that the mass of a nucleus is simply proportional to the number of nucleons in the nucleus, A. But the nucleons are bound into the nucleus, so the mass of the nucleus is less than the sum of the masses of the constituent nucleons. The mass of a nucleus is roughly ...
Historical influence to MRI
... recorded into spectroscopic images, corresponding to their frequency values. Virtually overnight Nuclear Magnetic ...
... recorded into spectroscopic images, corresponding to their frequency values. Virtually overnight Nuclear Magnetic ...
abstract
... The main effect of the static magnetic field on the growth dynamics of the bacterium S. aureus is shown in Fig. 1. Each symbol is an average from 4 independent measurements performed previously. We found that the number of CFU increases with the time of exposure and decreases with the magnitude of t ...
... The main effect of the static magnetic field on the growth dynamics of the bacterium S. aureus is shown in Fig. 1. Each symbol is an average from 4 independent measurements performed previously. We found that the number of CFU increases with the time of exposure and decreases with the magnitude of t ...
05 shell model
... coordinates (r, θ, φ) is of the form Ψnlm = Rnl (r)Ylm (θ, φ). The energy eigenvalues will depend on the principle quantum number, n, and the orbital angular momentum, l, but are degenerate in the magnetic quantum number m. These energy levels come in ‘bunches’ called “shells” with a large energy ga ...
... coordinates (r, θ, φ) is of the form Ψnlm = Rnl (r)Ylm (θ, φ). The energy eigenvalues will depend on the principle quantum number, n, and the orbital angular momentum, l, but are degenerate in the magnetic quantum number m. These energy levels come in ‘bunches’ called “shells” with a large energy ga ...
VIII. NUCLEAR MAGNETIC RESONANCE (NMR) SPECTROSCOPY
... momentum of electrons in orbitals of atoms or molecules. Electrons in orbitals with l = 1, 2, 3, … have angular momentum and therefore produce a magnetic dipole moment. Nuclei with magnetic moments produce a paramagnetic effect, but this effect is only about a millionth as large as paramagnetism due ...
... momentum of electrons in orbitals of atoms or molecules. Electrons in orbitals with l = 1, 2, 3, … have angular momentum and therefore produce a magnetic dipole moment. Nuclei with magnetic moments produce a paramagnetic effect, but this effect is only about a millionth as large as paramagnetism due ...
NUCLEAR MAGNETIC RESONANCE (NMR)
... Nuclear Magnetic Resonance Spectroscopy Introduction to NMR Spectroscopy • Protons in different environments absorb at slightly different frequencies, so they are distinguishable by NMR. • The frequency at which a particular proton absorbs is determined by its electronic environment. • The size of ...
... Nuclear Magnetic Resonance Spectroscopy Introduction to NMR Spectroscopy • Protons in different environments absorb at slightly different frequencies, so they are distinguishable by NMR. • The frequency at which a particular proton absorbs is determined by its electronic environment. • The size of ...
Lodestones Magnetic Poles
... years • Magnetic poles wander! • Magnetic & geographic poles not the same. • Magnetic declination: 11.5° • What’s strange about this picture? à ...
... years • Magnetic poles wander! • Magnetic & geographic poles not the same. • Magnetic declination: 11.5° • What’s strange about this picture? à ...
A. Blocal = Bo (1 - ) - USC Upstate: Faculty
... rotating magnetic component in the x-y plane that will induce the magnetic moment to tip to a different angle b) apply 2nd magnetic field, B1, to detect c) part of electromagnetic spectrum d) frequency, = absorption e) this absorption referred to as resonance f) energy absorbed g) frequency of o ...
... rotating magnetic component in the x-y plane that will induce the magnetic moment to tip to a different angle b) apply 2nd magnetic field, B1, to detect c) part of electromagnetic spectrum d) frequency, = absorption e) this absorption referred to as resonance f) energy absorbed g) frequency of o ...
Chapter 3: Nuclear Magnetic Resonance Spectroscopy
... What information can you obtain from a proton NMR spectrum of a compound? -How many different kinds of hydrogen are present (-CH, -CH2, -CH3). -Environment of the hydrogen atoms. -Relative number of hydrogen atoms. -Which hydrogen atoms are 2- or 3-bond neighbors. What to get from today’s lecture: - ...
... What information can you obtain from a proton NMR spectrum of a compound? -How many different kinds of hydrogen are present (-CH, -CH2, -CH3). -Environment of the hydrogen atoms. -Relative number of hydrogen atoms. -Which hydrogen atoms are 2- or 3-bond neighbors. What to get from today’s lecture: - ...
NMR Lecture II - Structure Determination
... Introduction to NMR Spectroscopy • Protons in different environments absorb at slightly different frequencies, so they are distinguishable by NMR. • The frequency at which a particular proton absorbs is determined by its electronic environment. • The size of the magnetic field generated by the elect ...
... Introduction to NMR Spectroscopy • Protons in different environments absorb at slightly different frequencies, so they are distinguishable by NMR. • The frequency at which a particular proton absorbs is determined by its electronic environment. • The size of the magnetic field generated by the elect ...
NMR-Understanding.pps
... Nuclear magnetic resonance is concerned with the magnetic properties of certain nuclei. In this course we are concerned with the magnetic behaviour of 13C nuclei. About 1% of all carbon atoms are the 13C isotope; the rest (apart from tiny amounts of the radioactive 14C) is 12C. ...
... Nuclear magnetic resonance is concerned with the magnetic properties of certain nuclei. In this course we are concerned with the magnetic behaviour of 13C nuclei. About 1% of all carbon atoms are the 13C isotope; the rest (apart from tiny amounts of the radioactive 14C) is 12C. ...
NMR Slides 2.1
... nuclear charges in rapidly rotating molecules or groups • The field associated with the molecular magnetic moment causes relaxation • Relaxation is most effective for small, symmetric molecules and groups, e.g CH3 ...
... nuclear charges in rapidly rotating molecules or groups • The field associated with the molecular magnetic moment causes relaxation • Relaxation is most effective for small, symmetric molecules and groups, e.g CH3 ...
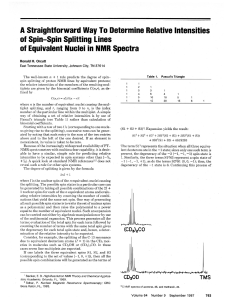
A straightforward way to determine relative intensities of spin
... where I is the nuclear spin of t h e n equivalent nuclei causing the splitting. The possible spin states in a particular case can he generated by taking all possible combinations of the 21 1nuclear spins for each of t h e n equivalent atoms and evaluating relative intensities by counting the number ...
... where I is the nuclear spin of t h e n equivalent nuclei causing the splitting. The possible spin states in a particular case can he generated by taking all possible combinations of the 21 1nuclear spins for each of t h e n equivalent atoms and evaluating relative intensities by counting the number ...
- Post Graduate Government College
... absorption of energy by a precessing nucleus and the resulting “flip” of its nuclear spin from a lower energy state to a higher energy state. • The precessing spins induce an oscillating magnetic field that is recorded as a signal by the instrument. – Signal: A recording in an NMR spectrum of a nucl ...
... absorption of energy by a precessing nucleus and the resulting “flip” of its nuclear spin from a lower energy state to a higher energy state. • The precessing spins induce an oscillating magnetic field that is recorded as a signal by the instrument. – Signal: A recording in an NMR spectrum of a nucl ...
for I = 1/2 nuclei - Instrumentation Engineer`s Site
... • He was the first to report the introduction of the MAS (magic angle sample spinning; MASS) technique that allowed him to achieve spectral resolution in solids sufficient to distinguish between chemical groups with either different chemical shifts or distinct Knight shifts. • In MASS, the sample is ...
... • He was the first to report the introduction of the MAS (magic angle sample spinning; MASS) technique that allowed him to achieve spectral resolution in solids sufficient to distinguish between chemical groups with either different chemical shifts or distinct Knight shifts. • In MASS, the sample is ...
Nuclear magnetic resonance
Nuclear magnetic resonance (NMR) is a physical phenomenon in which nuclei in a magnetic field absorb and re-emit electromagnetic radiation. This energy is at a specific resonance frequency which depends on the strength of the magnetic field and the magnetic properties of the isotope of the atoms; in practical applications, the frequency is similar to VHF and UHF television broadcasts (60–1000 MHz).NMR allows the observation of specific quantum mechanical magnetic properties of the atomic nucleus. Many scientific techniques exploit NMR phenomena to study molecular physics, crystals, and non-crystalline materials through NMR spectroscopy. NMR is also routinely used in advanced medical imaging techniques, such as in magnetic resonance imaging (MRI).All isotopes that contain an odd number of protons and/or of neutrons (see Isotope) have an intrinsic magnetic moment and angular momentum, in other words a nonzero spin, while all nuclides with even numbers of both have a total spin of zero. The most commonly studied nuclei are 1H and 13C, although nuclei from isotopes of many other elements (e.g. 2H, 6Li, 10B, 11B, 14N, 15N, 17O, 19F, 23Na, 29Si, 31P, 35Cl, 113Cd, 129Xe, 195Pt) have been studied by high-field NMR spectroscopy as well.A key feature of NMR is that the resonance frequency of a particular substance is directly proportional to the strength of the applied magnetic field. It is this feature that is exploited in imaging techniques; if a sample is placed in a non-uniform magnetic field then the resonance frequencies of the sample's nuclei depend on where in the field they are located. Since the resolution of the imaging technique depends on the magnitude of magnetic field gradient, many efforts are made to develop increased field strength, often using superconductors. The effectiveness of NMR can also be improved using hyperpolarization, and/or using two-dimensional, three-dimensional and higher-dimensional multi-frequency techniques.The principle of NMR usually involves two sequential steps:The alignment (polarization) of the magnetic nuclear spins in an applied, constant magnetic field B0.The perturbation of this alignment of the nuclear spins by employing an electro-magnetic, usually radio frequency (RF) pulse. The required perturbing frequency is dependent upon the static magnetic field (H0) and the nuclei of observation.The two fields are usually chosen to be perpendicular to each other as this maximizes the NMR signal strength. The resulting response by the total magnetization (M) of the nuclear spins is the phenomenon that is exploited in NMR spectroscopy and magnetic resonance imaging. Both use intense applied magnetic fields (H0) in order to achieve dispersion and very high stability to deliver spectral resolution, the details of which are described by chemical shifts, the Zeeman effect, and Knight shifts (in metals).NMR phenomena are also utilized in low-field NMR, NMR spectroscopy and MRI in the Earth's magnetic field (referred to as Earth's field NMR), and in several types of magnetometers.