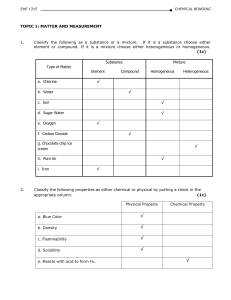
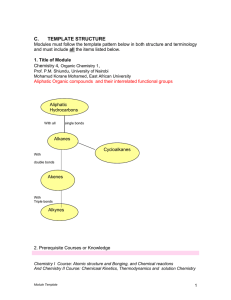
Chemical Bonding and Molecular Geometry
... H2, contains a covalent bond between its two hydrogen atoms. Figure 7.4 illustrates why this bond is formed. Starting on the far right, we have two separate hydrogen atoms with a particular potential energy, indicated by the red line. Along the x-axis is the distance between the two atoms. As the tw ...
... H2, contains a covalent bond between its two hydrogen atoms. Figure 7.4 illustrates why this bond is formed. Starting on the far right, we have two separate hydrogen atoms with a particular potential energy, indicated by the red line. Along the x-axis is the distance between the two atoms. As the tw ...
jyvaskla2 - School of Chemistry
... The topological atom The partitioning in real space of molecules into atoms by using these interatomic surfaces is a fundamental, quantum mechanically rigorous, method. It allows properties to be calculated for proper open systems, where exchange, e.g. with charge may occur between atoms. The proper ...
... The topological atom The partitioning in real space of molecules into atoms by using these interatomic surfaces is a fundamental, quantum mechanically rigorous, method. It allows properties to be calculated for proper open systems, where exchange, e.g. with charge may occur between atoms. The proper ...
Inorganometallic Chemistry
... elements, i.e. main groups 13, 14, except C, 15, except N and Se and Te from 16) replaces the carbon atom in the metal-carbon bond, then they really form a metalnonmetal bonding and, for the main part, p-block – d-block and f-block element bonding. Such compounds are the subject of a new field of st ...
... elements, i.e. main groups 13, 14, except C, 15, except N and Se and Te from 16) replaces the carbon atom in the metal-carbon bond, then they really form a metalnonmetal bonding and, for the main part, p-block – d-block and f-block element bonding. Such compounds are the subject of a new field of st ...
Correlation between Crystal Structure and Mobility in Organic Field
... Received March 23, 2004; E-mail: [email protected]; [email protected] ...
... Received March 23, 2004; E-mail: [email protected]; [email protected] ...
FREE Sample Here
... http://testbankwizard.eu/Test-Bank-for-Campbell-Biology-with-MasteringBiology-9th-Edition-by-Reec e 24) What is the maximum number of electrons in a single 2 p orbital of an atom? A) 1 B) 2 C) 3 D) 4 E) 5 Answer: B Topic: Concept 2.2 Skill: Knowledge/Comprehension 25) The organic molecules in living ...
... http://testbankwizard.eu/Test-Bank-for-Campbell-Biology-with-MasteringBiology-9th-Edition-by-Reec e 24) What is the maximum number of electrons in a single 2 p orbital of an atom? A) 1 B) 2 C) 3 D) 4 E) 5 Answer: B Topic: Concept 2.2 Skill: Knowledge/Comprehension 25) The organic molecules in living ...
jyvaskla2 - School of Chemistry
... The topological atom The partitioning in real space of molecules into atoms by using these interatomic surfaces is a fundamental, quantum mechanically rigorous, method. It allows properties to be calculated for proper open systems, where exchange, e.g. with charge may occur between atoms. The proper ...
... The topological atom The partitioning in real space of molecules into atoms by using these interatomic surfaces is a fundamental, quantum mechanically rigorous, method. It allows properties to be calculated for proper open systems, where exchange, e.g. with charge may occur between atoms. The proper ...
FREE Sample Here
... Full file at http://emailtestbank.com/ Test-Bank-for-Campbell-Biology-with-MasteringBiology-9th-Edition-by-Reece 24) What is the maximum number of electrons in a single 2 p orbital of an atom? A) 1 B) 2 C) 3 D) 4 E) 5 Answer: B Topic: Concept 2.2 Skill: Knowledge/Comprehension 25) The organic molec ...
... Full file at http://emailtestbank.com/ Test-Bank-for-Campbell-Biology-with-MasteringBiology-9th-Edition-by-Reece 24) What is the maximum number of electrons in a single 2 p orbital of an atom? A) 1 B) 2 C) 3 D) 4 E) 5 Answer: B Topic: Concept 2.2 Skill: Knowledge/Comprehension 25) The organic molec ...
FREE Sample Here
... 37) In comparing covalent bonds and ionic bonds, which of the following would you expect? A) An atom can form covalent bonds with multiple partner atoms, but only a single ionic bond with a single partner atom. B) Covalent bonds and ionic bonds occupy opposite ends of a continuous spectrum, from nea ...
... 37) In comparing covalent bonds and ionic bonds, which of the following would you expect? A) An atom can form covalent bonds with multiple partner atoms, but only a single ionic bond with a single partner atom. B) Covalent bonds and ionic bonds occupy opposite ends of a continuous spectrum, from nea ...
FREE Sample Here
... Full file at http://testbank360.eu/test-bank-campbell-biology-with-masteringbiology-9th-edition-reece Campbell's Biology, 9e (Reece et al.) Chapter 2 The Chemical Context of Life This chapter presents basic chemical principles for understanding the chemical context of living organisms, from atomic s ...
... Full file at http://testbank360.eu/test-bank-campbell-biology-with-masteringbiology-9th-edition-reece Campbell's Biology, 9e (Reece et al.) Chapter 2 The Chemical Context of Life This chapter presents basic chemical principles for understanding the chemical context of living organisms, from atomic s ...
DEPARTMENT OF CHEMISTRY, CFS, IIUM
... variety of matter is recognized is called a property. A characteristic that depends upon the amount of matter in the sample is called an extensive property. A characteristic that does not depend upon the amount of matter is called an intensive property. A characteristic that can be observed without ...
... variety of matter is recognized is called a property. A characteristic that depends upon the amount of matter in the sample is called an extensive property. A characteristic that does not depend upon the amount of matter is called an intensive property. A characteristic that can be observed without ...
Syntheses of Soluble, -Stacking Tetracene Derivatives π
... and alkyl/alkoxy-substituted tetracene molecules that are soluble in common organic solvents and have significant π-stacking motifs in crystal lattices. Fluorine has been applied extensively to tune the electronic and structural properties of organic semiconducting materials because of its strong el ...
... and alkyl/alkoxy-substituted tetracene molecules that are soluble in common organic solvents and have significant π-stacking motifs in crystal lattices. Fluorine has been applied extensively to tune the electronic and structural properties of organic semiconducting materials because of its strong el ...
P. Mignon, J. Steyaert, R. Loris, P. Geerlings, and S. Loverix, J. Biol
... Table I summarizes the crystallographic details of the structures that were used as a starting point for the computational study. In each mutant structure, the 3⬘-GMP substrate is bound in a catalytic productive mode, as is the case for the wild type structure (8). Apart from direct interactions inv ...
... Table I summarizes the crystallographic details of the structures that were used as a starting point for the computational study. In each mutant structure, the 3⬘-GMP substrate is bound in a catalytic productive mode, as is the case for the wild type structure (8). Apart from direct interactions inv ...
2.6 M - Thierry Karsenti
... atoms in the cyclic structure is other than carbon. Heterocyclic componds may be aliphatic or aromatic 15. Isomers: These are different compounds that have the same molecular formula. Isomers are further subdivided into: (a) structural isomers, (b) geometrical isomers and (c) stereoisomers(optical i ...
... atoms in the cyclic structure is other than carbon. Heterocyclic componds may be aliphatic or aromatic 15. Isomers: These are different compounds that have the same molecular formula. Isomers are further subdivided into: (a) structural isomers, (b) geometrical isomers and (c) stereoisomers(optical i ...
chm 205 - National Open University of Nigeria
... Many elements exist in more than one form. These forms are called allotropes, and the phenomenon is called allotropy. The two common allotropic forms of carbon, viz., diamond and graphite are well-known. These are, in fact, giant macromolecules consisting of C atoms linked by a network of covalent b ...
... Many elements exist in more than one form. These forms are called allotropes, and the phenomenon is called allotropy. The two common allotropic forms of carbon, viz., diamond and graphite are well-known. These are, in fact, giant macromolecules consisting of C atoms linked by a network of covalent b ...
CfE Advanced Higher Chemistry Unit 2: Organic
... mathematical functions for the two 1s orbitals that come together to form this molecule. A molecular orbital is a mathematical function describing the wave-like behaviour of an electron in a molecule. This function can be used to calculate chemical and physical properties such as the probability of ...
... mathematical functions for the two 1s orbitals that come together to form this molecule. A molecular orbital is a mathematical function describing the wave-like behaviour of an electron in a molecule. This function can be used to calculate chemical and physical properties such as the probability of ...
chemical bonding and molecular structure
... the inner electrons) and the outer shell that could accommodate a maximum of eight electrons. He, further assumed that these eight electrons occupy the corners of a cube which surround the ‘Kernel’. Thus the single outer shell electron of sodium would occupy one corner of the cube, while in the case ...
... the inner electrons) and the outer shell that could accommodate a maximum of eight electrons. He, further assumed that these eight electrons occupy the corners of a cube which surround the ‘Kernel’. Thus the single outer shell electron of sodium would occupy one corner of the cube, while in the case ...
Groups 2 and 7
... The name astatine comes from the Greek word for unstable. Astatine exists in nature in only very tiny amounts. It is estimated that only 30 grams of astatine exist on Earth at any one time. This is because it is radioactive, and its most stable isotope (210At) has a half-life of only 8 hours. It was ...
... The name astatine comes from the Greek word for unstable. Astatine exists in nature in only very tiny amounts. It is estimated that only 30 grams of astatine exist on Earth at any one time. This is because it is radioactive, and its most stable isotope (210At) has a half-life of only 8 hours. It was ...
Adsorption at Solid Surfaces
... Chemisorption of Hydrogen and Halogens Hydrogen (H2 ) In the H2 molecule, the valence electrons are all involved in the H-H -bond and there are no additional electrons which may interact with the substrate atoms. Consequently, chemisorption of hydrogen on metals is almost invariably a dissociative ...
... Chemisorption of Hydrogen and Halogens Hydrogen (H2 ) In the H2 molecule, the valence electrons are all involved in the H-H -bond and there are no additional electrons which may interact with the substrate atoms. Consequently, chemisorption of hydrogen on metals is almost invariably a dissociative ...
Adsorption at Solid Surfaces
... Chemisorption of Hydrogen and Halogens Hydrogen (H2 ) In the H2 molecule, the valence electrons are all involved in the H-H -bond and there are no additional electrons which may interact with the substrate atoms. Consequently, chemisorption of hydrogen on metals is almost invariably a dissociative ...
... Chemisorption of Hydrogen and Halogens Hydrogen (H2 ) In the H2 molecule, the valence electrons are all involved in the H-H -bond and there are no additional electrons which may interact with the substrate atoms. Consequently, chemisorption of hydrogen on metals is almost invariably a dissociative ...
Module 2 Alcohols, halogenoalkanes and analysis
... Throughout the centuries, chemists have synthesised new substances and investigated their properties in the search for more useful materials. In the recent past, organic chemists have developed a broad range of original and exciting materials, such as pharmaceuticals, refrigerants, solvents and plas ...
... Throughout the centuries, chemists have synthesised new substances and investigated their properties in the search for more useful materials. In the recent past, organic chemists have developed a broad range of original and exciting materials, such as pharmaceuticals, refrigerants, solvents and plas ...
chemistry -- questions -
... __ 23. An atom's atomic number is best described as the number of a) protons it contains. b) neutrons it contains. c) electrons in the outermost shell. d) protons and neutrons it contains. e) protons and electrons it contains. __ 24. An atom's atomic mass is best described as the mass of a) the pro ...
... __ 23. An atom's atomic number is best described as the number of a) protons it contains. b) neutrons it contains. c) electrons in the outermost shell. d) protons and neutrons it contains. e) protons and electrons it contains. __ 24. An atom's atomic mass is best described as the mass of a) the pro ...
H - Deans Community High School
... The reaction mixture stays colourless as the iodine molecules are converted back to iodide molecules by the thiosulphate ions. Once all the thiosulphate ions have been used, a blue black colour appears suddenly as iodine reacts with starch. ...
... The reaction mixture stays colourless as the iodine molecules are converted back to iodide molecules by the thiosulphate ions. Once all the thiosulphate ions have been used, a blue black colour appears suddenly as iodine reacts with starch. ...
Structural basis for bending of organic crystals{
... wherein the packing patterns are similar in three dimensions, will often have highly cross-linked structures (naphthalene, benzoic acid, D-glucose). These crystals would break rather than bend when subjected to a mechanical stress. In contrast, metals, which are isotropic but do not have cross-linki ...
... wherein the packing patterns are similar in three dimensions, will often have highly cross-linked structures (naphthalene, benzoic acid, D-glucose). These crystals would break rather than bend when subjected to a mechanical stress. In contrast, metals, which are isotropic but do not have cross-linki ...
Alkanes Chapter 1.1
... • A substitution reaction is one in which an atom or group of atoms in a molecule is replaced by another atom or group of atoms • Alkanes react in a specific type of substitution reaction called halogenation where a hydrogen atom is replaced by a halogen atom (Cl, Br, etc.) to form an alkyl halide c ...
... • A substitution reaction is one in which an atom or group of atoms in a molecule is replaced by another atom or group of atoms • Alkanes react in a specific type of substitution reaction called halogenation where a hydrogen atom is replaced by a halogen atom (Cl, Br, etc.) to form an alkyl halide c ...
Stabilization of Quinapril by Incorporating Hydrogen Bonding
... mol) was dissolved in a mixture of 125 ml water and 125 ml dichloromethane. The pH of the solution was adjusted between 7.5 and 8.5 by addition of aqueous ammonia. Reaction mixture was stirred for 30 min, the organic phase separated and washed with 50 ml water. The organic phase was evaporated under ...
... mol) was dissolved in a mixture of 125 ml water and 125 ml dichloromethane. The pH of the solution was adjusted between 7.5 and 8.5 by addition of aqueous ammonia. Reaction mixture was stirred for 30 min, the organic phase separated and washed with 50 ml water. The organic phase was evaporated under ...
Halogen bond

Halogen bonding (XB) is the non-covalent interaction that occurs between a halogen atom (Lewis acid) and a Lewis base. Although halogens are involved in other types of bonding (e.g. covalent), halogen bonding specifically refers to when the halogen acts as an electrophilic species.