AP Chem Chapter 13 Homework
... b. All other factors being equal, if adhesive forces are strong, capillary action is likely to occur less readily than if adhesive forces are weak. c. The shape of a meniscus depends on the difference between the strengths of cohesive forces and adhesive forces. d. Liquids with strong cohesive force ...
... b. All other factors being equal, if adhesive forces are strong, capillary action is likely to occur less readily than if adhesive forces are weak. c. The shape of a meniscus depends on the difference between the strengths of cohesive forces and adhesive forces. d. Liquids with strong cohesive force ...
NOBLE-GAS CHEMISTRY
... molecules (where M = Cu, Ag, and Au; X = F, Cl, and Br), and the binding energy was estimated to be as large as a quarter an eV for the Ar…AgF derivative.34 The work started from the most inert element (Ar) of the Ar–Kr–Xe set, and Kr and Xe were conquered in the following steps. To date, most of th ...
... molecules (where M = Cu, Ag, and Au; X = F, Cl, and Br), and the binding energy was estimated to be as large as a quarter an eV for the Ar…AgF derivative.34 The work started from the most inert element (Ar) of the Ar–Kr–Xe set, and Kr and Xe were conquered in the following steps. To date, most of th ...
N,N`-diisopropylthiourea and N,N` - South African Journal of Science
... polyhedra around the Zn(II) ion a distorted tetrahedral. The two N,N’-dicyclohexylthiourea ligands were sulphur-bonded to the metal atom with the two chloride ligands facing opposite to the two organic ligands. In this four-coordinate structure, molecular units are arranged so that one of the chlori ...
... polyhedra around the Zn(II) ion a distorted tetrahedral. The two N,N’-dicyclohexylthiourea ligands were sulphur-bonded to the metal atom with the two chloride ligands facing opposite to the two organic ligands. In this four-coordinate structure, molecular units are arranged so that one of the chlori ...
The Role of Tetrahedral Building Blocks in Low-Dimensional Oxohalide Materials Iwan Zimmermann
... having a stereochemically active lone-pair such as Se4+, Sb3+ Te4+, Bi3+ or I5+ can be introduced into the crystal structure. Due to their non-bonding electron pair, such elements adopt a one-sided, asymmetric coordination caused by a second order Jahn Teller (SOJT) distortion.[3-4] Lone-pairs do no ...
... having a stereochemically active lone-pair such as Se4+, Sb3+ Te4+, Bi3+ or I5+ can be introduced into the crystal structure. Due to their non-bonding electron pair, such elements adopt a one-sided, asymmetric coordination caused by a second order Jahn Teller (SOJT) distortion.[3-4] Lone-pairs do no ...
Photoactivation mechanism of PAmCherry based on crystal
... cellular and even macromolecular structure that were not previously discernible with less than 40 nm resolution (1). There is significant demand for a broader and more diverse range of photoactivatable fluorescent probes (2), in particular irreversibly photoactivatable fluorescent proteins (PAFPs). ...
... cellular and even macromolecular structure that were not previously discernible with less than 40 nm resolution (1). There is significant demand for a broader and more diverse range of photoactivatable fluorescent probes (2), in particular irreversibly photoactivatable fluorescent proteins (PAFPs). ...
getting started 3.1 hydrocarbons
... 1. A functional group is a structural arrangement of atoms that, because of their electronegativity and bonding type, imparts particular characteristics to the molecule. 2. C=C and C)C bonds are more reactive than C–C bonds because the second and third bonds formed are weaker than the single bonds f ...
... 1. A functional group is a structural arrangement of atoms that, because of their electronegativity and bonding type, imparts particular characteristics to the molecule. 2. C=C and C)C bonds are more reactive than C–C bonds because the second and third bonds formed are weaker than the single bonds f ...
Modular Chemistry: Secondary Building Units as a
... the case of BDC having one carboxylate oxygen as part of the SBU, the expansion is done with a benzyl unit) (Figure 4c). We found that all methanol, including the ligands, can be removed from the voids to give a porous network (Table 2) having open zinc sites. It is interesting to note that MOF-3 ma ...
... the case of BDC having one carboxylate oxygen as part of the SBU, the expansion is done with a benzyl unit) (Figure 4c). We found that all methanol, including the ligands, can be removed from the voids to give a porous network (Table 2) having open zinc sites. It is interesting to note that MOF-3 ma ...
Structure and Bonding of Ordered Organic Monolayers of 1, 3, 5, 7
... coplanar and are separated by 3.14 Å, slightly shorter than the 3.85 Å separation between adjacent SidSi dimers. Figure 4b-e depicts four of the ways in which COT might interact with Si(001). One possibility is for four π electrons of COT to interact with one SidSi dimer, resulting in a [4 + 2] cycl ...
... coplanar and are separated by 3.14 Å, slightly shorter than the 3.85 Å separation between adjacent SidSi dimers. Figure 4b-e depicts four of the ways in which COT might interact with Si(001). One possibility is for four π electrons of COT to interact with one SidSi dimer, resulting in a [4 + 2] cycl ...
Vorlesung Supramolekulare Chemie
... but they add up. If we now look at the enthalpies of covalent bonds, it is obvious that entropy does not play a significant role in the formation of bonds. Example: Reaction of two molecules forming one new C-C bond: ΔG = - 415 KJ/mol + 23 KJ/mol. The entropic effect accounts for less than 5% of the ...
... but they add up. If we now look at the enthalpies of covalent bonds, it is obvious that entropy does not play a significant role in the formation of bonds. Example: Reaction of two molecules forming one new C-C bond: ΔG = - 415 KJ/mol + 23 KJ/mol. The entropic effect accounts for less than 5% of the ...
12 - einstein classes
... It is obtained by heating white phosphorous at 470 K and under high pressure. A series of phase of block phosphorous are formed. One of these phases consists of an extended layer structure in which each P is bound to three neighbours by single bonds. ...
... It is obtained by heating white phosphorous at 470 K and under high pressure. A series of phase of block phosphorous are formed. One of these phases consists of an extended layer structure in which each P is bound to three neighbours by single bonds. ...
GCE Getting Started - Edexcel
... Understand the interactions in molecules, such as H2O, liquid NH3 and liquid HF, which give rise to hydrogen bonding. Understand the following anomalous properties of water resulting from hydrogen bonding: i. its relatively high melting temperature and boiling temperature ii. the density of ice comp ...
... Understand the interactions in molecules, such as H2O, liquid NH3 and liquid HF, which give rise to hydrogen bonding. Understand the following anomalous properties of water resulting from hydrogen bonding: i. its relatively high melting temperature and boiling temperature ii. the density of ice comp ...
Chemical Bonding
... In Chapter 1 you studied various representations, or models, of the structure of atoms. In this activity, you will build models of a variety of molecules. This exercise will help you to see that different chemicals have different molecular structures. Materials: molecular models kit • In your molecu ...
... In Chapter 1 you studied various representations, or models, of the structure of atoms. In this activity, you will build models of a variety of molecules. This exercise will help you to see that different chemicals have different molecular structures. Materials: molecular models kit • In your molecu ...
CHEM-1411 Final Practice Exam
... sulfur atom in the first structure is therefore sp3. However, the sulfur is not simply sp3 hybridized in the second structure, which has an “expanded octet” around the sulfur atom. Hybridizations that allow more than an octet of electrons around an atom are sp3d (10 electrons) and sp3d2 (12 electron ...
... sulfur atom in the first structure is therefore sp3. However, the sulfur is not simply sp3 hybridized in the second structure, which has an “expanded octet” around the sulfur atom. Hybridizations that allow more than an octet of electrons around an atom are sp3d (10 electrons) and sp3d2 (12 electron ...
1411FINALSAMPLE+KEY - Houston Community College
... sulfur atom in the first structure is therefore sp3. However, the sulfur is not simply sp3 hybridized in the second structure, which has an “expanded octet” around the sulfur atom. Hybridizations that allow more than an octet of electrons around an atom are sp3d (10 electrons) and sp3d2 (12 electron ...
... sulfur atom in the first structure is therefore sp3. However, the sulfur is not simply sp3 hybridized in the second structure, which has an “expanded octet” around the sulfur atom. Hybridizations that allow more than an octet of electrons around an atom are sp3d (10 electrons) and sp3d2 (12 electron ...
Nickel(II) cis- and trans-Dimethyl Complexes of
... monitoring the decrease of the Me resonance in the 1H NMR spectrum relative to an internal reference of maleic anhydride flame-sealed in a glass capillary. Decomposition of 2 displayed first-order kinetics and was independent of the concentration of 2 and the addition of excess tBuCCeth, as shown in ...
... monitoring the decrease of the Me resonance in the 1H NMR spectrum relative to an internal reference of maleic anhydride flame-sealed in a glass capillary. Decomposition of 2 displayed first-order kinetics and was independent of the concentration of 2 and the addition of excess tBuCCeth, as shown in ...
Minimum electrophilicity principle in Lewis acid–base complexes of
... and makes it hard Lewis acid. Conversely, addition of three soft, electropositive atoms (such as hydrogen) softens the boron and makes it a soft Lewis acid. It is expected that more electronegative species pull charge from the boron more effectively; therefore, they would have strong interactions wi ...
... and makes it hard Lewis acid. Conversely, addition of three soft, electropositive atoms (such as hydrogen) softens the boron and makes it a soft Lewis acid. It is expected that more electronegative species pull charge from the boron more effectively; therefore, they would have strong interactions wi ...
Answers to Selected Exercises
... Some energy is released as heat through friction between the ball and the sand. 4.11 The energy source of a 100-watt light bulb is electrical current from household wiring. Energy is radiated in the form of heat and visible light. The energy source for an adult person is food. After a series of comp ...
... Some energy is released as heat through friction between the ball and the sand. 4.11 The energy source of a 100-watt light bulb is electrical current from household wiring. Energy is radiated in the form of heat and visible light. The energy source for an adult person is food. After a series of comp ...
1411FINALSAMPLEs and Key
... sulfur atom in the first structure is therefore sp3. However, the sulfur is not simply sp3 hybridized in the second structure, which has an “expanded octet” around the sulfur atom. Hybridizations that allow more than an octet of electrons around an atom are sp3d (10 electrons) and sp3d2 (12 electron ...
... sulfur atom in the first structure is therefore sp3. However, the sulfur is not simply sp3 hybridized in the second structure, which has an “expanded octet” around the sulfur atom. Hybridizations that allow more than an octet of electrons around an atom are sp3d (10 electrons) and sp3d2 (12 electron ...
Interaction between hydrogen molecules and - FHI
... total energy required to form a structure with respect to that of a graphene. Small fullerenes have relatively high formation energy, thus less stable. It is due to the fact that they cannot satisfy the isolated pentagon rule.20 We introduced a La atom inside each fullerene and carefully optimized t ...
... total energy required to form a structure with respect to that of a graphene. Small fullerenes have relatively high formation energy, thus less stable. It is due to the fact that they cannot satisfy the isolated pentagon rule.20 We introduced a La atom inside each fullerene and carefully optimized t ...
Crystal Structures
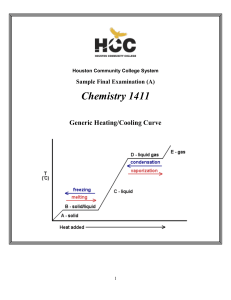
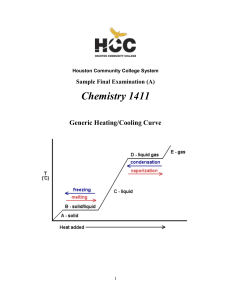
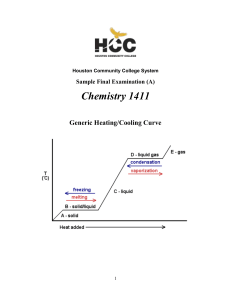
... Heating ice produces water (T > 0 C, 32 F – melting point) Heating water produces steam (water vapor) (T > 100 C, 212 F) These transitions are phase transitions. Some materials have triple points, where all three forms can exist together at the same time. ...
... Heating ice produces water (T > 0 C, 32 F – melting point) Heating water produces steam (water vapor) (T > 100 C, 212 F) These transitions are phase transitions. Some materials have triple points, where all three forms can exist together at the same time. ...
Chemical bonding
... 52) Ans: Sigma and pi 53) Ans: It is a type of covalent bond formed due to overlap of atomic orbitals side ways. 54) Ans: A pi electron cloud consists of two saucer shaped clouds above and below the plane of participating atoms. 55) Ans: No 56) Ans: Sigma bonds. 57) Ans: Hydrogen bond is defined as ...
... 52) Ans: Sigma and pi 53) Ans: It is a type of covalent bond formed due to overlap of atomic orbitals side ways. 54) Ans: A pi electron cloud consists of two saucer shaped clouds above and below the plane of participating atoms. 55) Ans: No 56) Ans: Sigma bonds. 57) Ans: Hydrogen bond is defined as ...
Chapter 8 "Ionic versus Covalent Bonding"
... 1. Atoms interact with one another to form aggregates such as molecules, compounds, and crystals because doing so lowers the total energy of the system; that is, the aggregates are more stable than the isolated atoms. 2. Energy is required to dissociate bonded atoms or ions into isolated atoms or io ...
... 1. Atoms interact with one another to form aggregates such as molecules, compounds, and crystals because doing so lowers the total energy of the system; that is, the aggregates are more stable than the isolated atoms. 2. Energy is required to dissociate bonded atoms or ions into isolated atoms or io ...
Amidine: Structure, Reactivity and Complexation Behaviour
... disruption and formation of chemical bonds, being essential, to establish databases with experimental reliable data of bond dissociation energies (BDEs) as a direct information of the strength of chemical bonds[26]. Chemical species containing the N-H bond form an important class of compounds with a ...
... disruption and formation of chemical bonds, being essential, to establish databases with experimental reliable data of bond dissociation energies (BDEs) as a direct information of the strength of chemical bonds[26]. Chemical species containing the N-H bond form an important class of compounds with a ...
Molecular Recognition of Trans-Chiral Schiff Base
... ution paramagnetism of Ni(II) complexes, structural phase transition of Cu(II) complexes, chiral catalysts, and some types of molecule-based magnets and other interesting facts about correlation between structures and properties are known and these facts are cooperative ef‐ fect involving intermolec ...
... ution paramagnetism of Ni(II) complexes, structural phase transition of Cu(II) complexes, chiral catalysts, and some types of molecule-based magnets and other interesting facts about correlation between structures and properties are known and these facts are cooperative ef‐ fect involving intermolec ...
Topological Analysis of Electron Density
... very much like the balls and spheres of molecular models !!! The simple binary hydrides of the second period elements show that the relative volumes of space associated with each element is determined by their relative electronegativities. Surfaces are truncated at 0.001 au. ...
... very much like the balls and spheres of molecular models !!! The simple binary hydrides of the second period elements show that the relative volumes of space associated with each element is determined by their relative electronegativities. Surfaces are truncated at 0.001 au. ...
Halogen bond

Halogen bonding (XB) is the non-covalent interaction that occurs between a halogen atom (Lewis acid) and a Lewis base. Although halogens are involved in other types of bonding (e.g. covalent), halogen bonding specifically refers to when the halogen acts as an electrophilic species.