Wiggler_dynamics_PRST-AB
... the chamber wall. Secondaries can also strike the wall, producing more secondaries, so that the population of secondaries under some circumstances can outnumber the primary electrons. An “electron cloud” formed in this way can negatively impact beam stability and quality [1]. If the beam particles a ...
... the chamber wall. Secondaries can also strike the wall, producing more secondaries, so that the population of secondaries under some circumstances can outnumber the primary electrons. An “electron cloud” formed in this way can negatively impact beam stability and quality [1]. If the beam particles a ...
Electricity - Deakin University Blogs
... protons. However, atoms can lose or gain electrons quite easily (gaining and losing protons is quite difficult) and so an atom that has more electrons than it has protons will have a net negative charge, and an atom that has fewer electrons than it has protons has a net positive charge. Atoms that h ...
... protons. However, atoms can lose or gain electrons quite easily (gaining and losing protons is quite difficult) and so an atom that has more electrons than it has protons will have a net negative charge, and an atom that has fewer electrons than it has protons has a net positive charge. Atoms that h ...
ABSTRACT Title of Document:
... I have measured the conditions for electrical breakdown between two electrodes separated by a gap of about 1 μm, a regime which has not been characterized adequately previously. Initially, as mentioned above, my desire was to better understand the energetics and transport through a SAM by measuring ...
... I have measured the conditions for electrical breakdown between two electrodes separated by a gap of about 1 μm, a regime which has not been characterized adequately previously. Initially, as mentioned above, my desire was to better understand the energetics and transport through a SAM by measuring ...
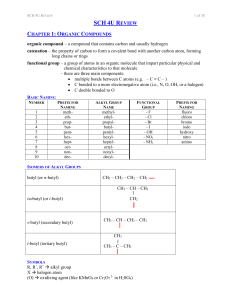
SCH 4U REVIEW Notes
... CHAPTER 1: ORGANIC COMPOUNDS organic compound – a compound that contains carbon and usually hydrogen catenation – the property of carbon to form a covalent bond with another carbon atom, forming long chains or rings functional group – a group of atoms in an organic molecule that impart particular ph ...
... CHAPTER 1: ORGANIC COMPOUNDS organic compound – a compound that contains carbon and usually hydrogen catenation – the property of carbon to form a covalent bond with another carbon atom, forming long chains or rings functional group – a group of atoms in an organic molecule that impart particular ph ...
Ch 17 Solutions
... It is often thought that the electron, with its greater acceleration and greater final speed, will have the greater final kinetic energy. However, the increase in kinetic energy is equal to the decrease in potential energy, which is the product of the object’s charge and the change in potential. How ...
... It is often thought that the electron, with its greater acceleration and greater final speed, will have the greater final kinetic energy. However, the increase in kinetic energy is equal to the decrease in potential energy, which is the product of the object’s charge and the change in potential. How ...
ELECTROSTATICS UNIT I TOPICS TO BE COVERED
... For a parallel combination of capacitors, connected across a dc source, potential difference (or voltage) across each capacitor will be the same. From here we get Cp = C1 + C2 + C3 + ............... Energy stored in a charged capacitor is given by U = ...
... For a parallel combination of capacitors, connected across a dc source, potential difference (or voltage) across each capacitor will be the same. From here we get Cp = C1 + C2 + C3 + ............... Energy stored in a charged capacitor is given by U = ...
Biographies and Definitions
... Sciences in Paris (in 1773). But it was also an extraordinary paper – it contained solutions to a range of unsolved problems in structural mechanics, mostly obtained by the calculus of variations. A few years later, in 1777, he shared with Jan Hendrik van Swinden (1746-1823) the Grand Prix of the Ac ...
... Sciences in Paris (in 1773). But it was also an extraordinary paper – it contained solutions to a range of unsolved problems in structural mechanics, mostly obtained by the calculus of variations. A few years later, in 1777, he shared with Jan Hendrik van Swinden (1746-1823) the Grand Prix of the Ac ...
Electric and Magnetic Properties of Ultra Thin (Ga,Mn)As Epilayers
... between the localized magnetic moments of Mn2+ is mediated by the conduction holes (S = 1/2) in the (Ga,Mn)As valence band, through an antiferromagnetic coupling to the Mn2+ ions. Mean-field theory calculations have shown very good agreement with experiments [15, 19, 20]. The main approach is the ab ...
... between the localized magnetic moments of Mn2+ is mediated by the conduction holes (S = 1/2) in the (Ga,Mn)As valence band, through an antiferromagnetic coupling to the Mn2+ ions. Mean-field theory calculations have shown very good agreement with experiments [15, 19, 20]. The main approach is the ab ...
Effect of interlayer trapping and detrapping on the determination of
... effect of SiNx : H thickness, tbl, in the Dit measurement while keeping constant the thickness, thk, of the high-k 共HfO2兲 dielectric film. Since thk does not change, the high-k dielectric must show the same properties for all the samples and we cannot attribute the discrepancies to any mechanism occ ...
... effect of SiNx : H thickness, tbl, in the Dit measurement while keeping constant the thickness, thk, of the high-k 共HfO2兲 dielectric film. Since thk does not change, the high-k dielectric must show the same properties for all the samples and we cannot attribute the discrepancies to any mechanism occ ...
Supplemental Informaton
... • Prefixes are indication of the number of atoms: mono-, di-, tri-, tetra-, penta-, hexa• order of naming nonmetal 1 & nonmetal (ide)2 nonmetal 1 is to the left and bottom of nonmetal 2 based on it is named first in the nomenclature scheme. Si - C - As - P - N - H - Se - S - I - Br - Cl - O - F • S ...
... • Prefixes are indication of the number of atoms: mono-, di-, tri-, tetra-, penta-, hexa• order of naming nonmetal 1 & nonmetal (ide)2 nonmetal 1 is to the left and bottom of nonmetal 2 based on it is named first in the nomenclature scheme. Si - C - As - P - N - H - Se - S - I - Br - Cl - O - F • S ...
unusual behavior of radiant barrier materials in fire causation
... Aluminum is an electrical conductor. All conductors have a certain amount of electrical resistance. If enough electrical current flows through that resistance, heat is generated. If enough heat is generated and there is ignitable material close by, a fire very often results. It turns out that a roof ...
... Aluminum is an electrical conductor. All conductors have a certain amount of electrical resistance. If enough electrical current flows through that resistance, heat is generated. If enough heat is generated and there is ignitable material close by, a fire very often results. It turns out that a roof ...
Study material of Science for class X
... Block your nose by pressing it between your thumb and index finger. Now eat sugar again. Is there any difference in its taste? While eating lunch, block your nose in the same way and notice if you can fully appreciate the taste of the food you are eating. Observations:1. When we put sugar in our ...
... Block your nose by pressing it between your thumb and index finger. Now eat sugar again. Is there any difference in its taste? While eating lunch, block your nose in the same way and notice if you can fully appreciate the taste of the food you are eating. Observations:1. When we put sugar in our ...
colossal magnetoresistance of systems with Magnetic and electric
... polarisation is defined as P = (n↑ − n↓ )/(n↑ + n↓ ), n↑,↓ where n↑,↓ are the densities of up and down spin carriers at the Fermi energy. If P = 1 the carriers are fully polarised. This is the case of manganites [22], double perovskites [23] and chromium dioxide [24]. These materials are called half ...
... polarisation is defined as P = (n↑ − n↓ )/(n↑ + n↓ ), n↑,↓ where n↑,↓ are the densities of up and down spin carriers at the Fermi energy. If P = 1 the carriers are fully polarised. This is the case of manganites [22], double perovskites [23] and chromium dioxide [24]. These materials are called half ...
On wind-driven electrojets at magnetic cusps
... spheric density was largest. Similary, the weakest currents were found in regions of low ionospheric density, in plasma voids. This is due in large part to the fact that the current density is directly proportional to the ionospheric density given an approximately constant difference between the ion ...
... spheric density was largest. Similary, the weakest currents were found in regions of low ionospheric density, in plasma voids. This is due in large part to the fact that the current density is directly proportional to the ionospheric density given an approximately constant difference between the ion ...
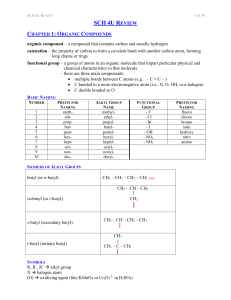
12 U Chem Review
... CHAPTER 1: ORGANIC COMPOUNDS organic compound – a compound that contains carbon and usually hydrogen catenation – the property of carbon to form a covalent bond with another carbon atom, forming long chains or rings functional group – a group of atoms in an organic molecule that impart particular ph ...
... CHAPTER 1: ORGANIC COMPOUNDS organic compound – a compound that contains carbon and usually hydrogen catenation – the property of carbon to form a covalent bond with another carbon atom, forming long chains or rings functional group – a group of atoms in an organic molecule that impart particular ph ...
Chapter wise Theoretical Important Questions in Physics for Class-XII
... Distinguish between conductors, insulators and semiconductors on the basis of energy bands. What are extrinsic semiconductors? Mention its types and explain the mechanism of conduction in each. Explain the conduction in N Type and P Type semiconductor on the basis of band theory. Explain the formati ...
... Distinguish between conductors, insulators and semiconductors on the basis of energy bands. What are extrinsic semiconductors? Mention its types and explain the mechanism of conduction in each. Explain the conduction in N Type and P Type semiconductor on the basis of band theory. Explain the formati ...