Periodic Table
... Periodic Table Matching Match each item with the correct statement below. a. electronegativity f. b. ionization energy g. c. atomic radius h. d. metal i. e. transition metal j. ____ ____ ____ ____ ____ ...
... Periodic Table Matching Match each item with the correct statement below. a. electronegativity f. b. ionization energy g. c. atomic radius h. d. metal i. e. transition metal j. ____ ____ ____ ____ ____ ...
Unit 2 Periodic Table
... Atoms of this family have 6 valence electrons. Known as chalcogen family. Most elements in this family share electrons when forming compounds. Oxygen is the most abundant element in the earth’s crust. It is extremely active and combines with almost all elements. ...
... Atoms of this family have 6 valence electrons. Known as chalcogen family. Most elements in this family share electrons when forming compounds. Oxygen is the most abundant element in the earth’s crust. It is extremely active and combines with almost all elements. ...
Chemistry Textbook Notes
... Common properties of purse substances are colour, physical state, melting/boiling points, density electrical conductivity, solubility in different liquids and mechanical properties. Distinct colours e.g. Deep brown- liquid bromine, solid copper- reddish brown etc. States of matter are solid, liquid ...
... Common properties of purse substances are colour, physical state, melting/boiling points, density electrical conductivity, solubility in different liquids and mechanical properties. Distinct colours e.g. Deep brown- liquid bromine, solid copper- reddish brown etc. States of matter are solid, liquid ...
Periodic Table Funsheet (KEY) 1. What family has the most active
... 14. Group 17 elements are called HALOGENS. 15. Group 18 elements are called NOBLE GASES. 16. As you go from left to right across the periodic table, the elements go from (METALS / nonmetals) to (metals / NONMETALS). 17. The most active element in Group 17 is FLUORINE. 18. What sublevels are filling ...
... 14. Group 17 elements are called HALOGENS. 15. Group 18 elements are called NOBLE GASES. 16. As you go from left to right across the periodic table, the elements go from (METALS / nonmetals) to (metals / NONMETALS). 17. The most active element in Group 17 is FLUORINE. 18. What sublevels are filling ...
Chapter 13
... outermost s sublevel and the nearby d sublevel contain electrons (Groups 3-12) 4. Inner transition metals – elements whose outermost s sublevel and nearby f sublevel contain electrons… the two rows below the table ...
... outermost s sublevel and the nearby d sublevel contain electrons (Groups 3-12) 4. Inner transition metals – elements whose outermost s sublevel and nearby f sublevel contain electrons… the two rows below the table ...
Revision map for the Periodic Table
... 13. The electrons are arranged in shells. 14. The outermost shell is called the valence shell. 15. Electrons in the valence shell are called valence electrons. 16. Each atom has one or more valence electrons. 17. Most atoms have one or more inner shells of electrons inside the valence shell. 18. The ...
... 13. The electrons are arranged in shells. 14. The outermost shell is called the valence shell. 15. Electrons in the valence shell are called valence electrons. 16. Each atom has one or more valence electrons. 17. Most atoms have one or more inner shells of electrons inside the valence shell. 18. The ...
The Periodic Table
... *As you move across the period, the number of electrons in the outermost orbital increases by one. *In a group, the outermost orbital has the same number of electrons. *Because electrons are the outermost particles in an atom, they are the ones that give an atom it’s properties, so elements with sim ...
... *As you move across the period, the number of electrons in the outermost orbital increases by one. *In a group, the outermost orbital has the same number of electrons. *Because electrons are the outermost particles in an atom, they are the ones that give an atom it’s properties, so elements with sim ...
ATOMIC STRUCTURE - IGCSE STUDY BANK
... usually weak materials eg soft or brittle solids (exceptions like silicon, and carbon as diamond, which are very hard and strong) if solid, not easily beaten into shape or drawn into wire, tend to be too brittle solids not usually sonorous Chemical properties of non-metals ...
... usually weak materials eg soft or brittle solids (exceptions like silicon, and carbon as diamond, which are very hard and strong) if solid, not easily beaten into shape or drawn into wire, tend to be too brittle solids not usually sonorous Chemical properties of non-metals ...
Matter: Building Blocks of the Universe Chapter 5 Classification of
... the same number of valence electrons Family 1—Alkali—soft, silver, white, shiny—react or combine with other elements easily—never found alone in nature Family 2—Alkaline—Earth metals—very reactive Between Family 2 and 13 are the transition metals—these are the metals you are most familiar with ...
... the same number of valence electrons Family 1—Alkali—soft, silver, white, shiny—react or combine with other elements easily—never found alone in nature Family 2—Alkaline—Earth metals—very reactive Between Family 2 and 13 are the transition metals—these are the metals you are most familiar with ...
family includes a non-metal
... The hydrogen square sits atop Family AI, but it is not a member of that family. Hydrogen is in a class of its own. It’s a gas at room temperature. It has one proton and one electron in its one and only energy level. Hydrogen only needs 2 electrons to fill up its valence shell. ...
... The hydrogen square sits atop Family AI, but it is not a member of that family. Hydrogen is in a class of its own. It’s a gas at room temperature. It has one proton and one electron in its one and only energy level. Hydrogen only needs 2 electrons to fill up its valence shell. ...
Name
... Ionization energy is the energy required to remove an electron from an atom. This energy is measured when the atom is in the gaseous state. The first ionization energy is the energy required to remove the first electron from an atom. The second ionization energy is the energy required to remove ...
... Ionization energy is the energy required to remove an electron from an atom. This energy is measured when the atom is in the gaseous state. The first ionization energy is the energy required to remove the first electron from an atom. The second ionization energy is the energy required to remove ...
PERIODIC TABLE quiz study guide
... 2. Be able to draw and describe the basic structure of an atom using the following terms: Proton (its charge and a.m.u.), Neutron (and its charge and a.m.u.), Electron (and its charge and a.m.u.), Nu ...
... 2. Be able to draw and describe the basic structure of an atom using the following terms: Proton (its charge and a.m.u.), Neutron (and its charge and a.m.u.), Electron (and its charge and a.m.u.), Nu ...
Week 9 (wk9) - Riverside Local Schools
... elements in the periodic table were arranged in increasing order according to nuclear charge, or the number of… 2. The PERIODIC LAW states that the physical and chemical properties of the elements are periodic functions of their… 3. When elements are arranged in order of increasing atomic number, el ...
... elements in the periodic table were arranged in increasing order according to nuclear charge, or the number of… 2. The PERIODIC LAW states that the physical and chemical properties of the elements are periodic functions of their… 3. When elements are arranged in order of increasing atomic number, el ...
Atomic Properties and the Periodic Table
... Due to additional energy level as you go down Due to increasing nuclear charge ...
... Due to additional energy level as you go down Due to increasing nuclear charge ...
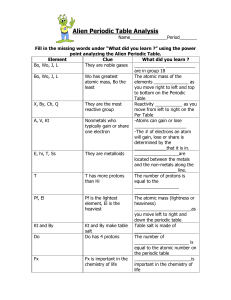
CHMR_AYS_U4AlienPeriodicTableAnalysis_V01
... _____________ as you move from right to left and top to bottom on the Periodic Table. Reactivity ______________ as you move from left to right on the Periodic Table, except for the ________ ____. Atoms can gain or lose _____________. The # of electrons an atom will gain, lose or share is determined ...
... _____________ as you move from right to left and top to bottom on the Periodic Table. Reactivity ______________ as you move from left to right on the Periodic Table, except for the ________ ____. Atoms can gain or lose _____________. The # of electrons an atom will gain, lose or share is determined ...
Uint one - pisscience
... *He classified each main group into two subgroups (A,B) because he found differences between their properties. Advantages of Mendeleev's Table: 1-He left spaces in his table as he predicted the discovery of new elements. 2-He corrected the wrong estimated atomic weights of some elements. Disadvantag ...
... *He classified each main group into two subgroups (A,B) because he found differences between their properties. Advantages of Mendeleev's Table: 1-He left spaces in his table as he predicted the discovery of new elements. 2-He corrected the wrong estimated atomic weights of some elements. Disadvantag ...
Ex. 06 Answer
... Across a period, the elements show a gradual change (decrease) in atomic size. b) 5 c) 2 d) Metals — a, b, c, d Metalloid — e Non-metals — f, g, h, i, j ...
... Across a period, the elements show a gradual change (decrease) in atomic size. b) 5 c) 2 d) Metals — a, b, c, d Metalloid — e Non-metals — f, g, h, i, j ...
Our modern Periodic Table
... b) Attempts focused on grouping elements with similar properties c) In 1867, Dimitri Mendeleev found patterns in the elements and organized them into a table d) The resulting table had holes for elements not yet discovered ...
... b) Attempts focused on grouping elements with similar properties c) In 1867, Dimitri Mendeleev found patterns in the elements and organized them into a table d) The resulting table had holes for elements not yet discovered ...
Atoms
... 1. Write out the ions present in the compound. Al3+ and O22. Determine the number of each ion to produce a neutral compound. Find the coefficient of each element that, when multiplied by the charge, will provide the lowest common multiple. So what do we have to multiple 3 by to make it 3+ and ...
... 1. Write out the ions present in the compound. Al3+ and O22. Determine the number of each ion to produce a neutral compound. Find the coefficient of each element that, when multiplied by the charge, will provide the lowest common multiple. So what do we have to multiple 3 by to make it 3+ and ...
Chemistry Ch. 5
... The periodic table organizes the elements in a particular way. A great deal of information about an element can be gathered from its position in the period table. For example, you can predict with reasonably good accuracy the physical and chemical properties of the element. You can also predict what ...
... The periodic table organizes the elements in a particular way. A great deal of information about an element can be gathered from its position in the period table. For example, you can predict with reasonably good accuracy the physical and chemical properties of the element. You can also predict what ...
PPT Periodic Families from Class
... What does it mean to be reactive? • We will be describing elements according to their reactivity. • Elements that are reactive bond easily with other elements to make compounds. • Some elements are only found in nature bonded with other elements. • What makes an element reactive? • An incomplete va ...
... What does it mean to be reactive? • We will be describing elements according to their reactivity. • Elements that are reactive bond easily with other elements to make compounds. • Some elements are only found in nature bonded with other elements. • What makes an element reactive? • An incomplete va ...
Periodicity of Elements
... The atomic number tells how many. protons the atom of that element contains in the nucleus. Use the periodic table in your textbook, or another periodic table, to look up the a t ~ m i cnumbers. It will be the whole number just above the element symbol. To find the answer to the question, use only t ...
... The atomic number tells how many. protons the atom of that element contains in the nucleus. Use the periodic table in your textbook, or another periodic table, to look up the a t ~ m i cnumbers. It will be the whole number just above the element symbol. To find the answer to the question, use only t ...
Alien Periodic Table Analysis
... They are the most Reactivity ___________ as you reactive group move from left to right on the Per Table A, V, Kt Nonmetals who -Atoms can gain or lose typically gain or share _____________________ one electron -The # of electrons an atom will gain, lose or share is determined by the _____________tha ...
... They are the most Reactivity ___________ as you reactive group move from left to right on the Per Table A, V, Kt Nonmetals who -Atoms can gain or lose typically gain or share _____________________ one electron -The # of electrons an atom will gain, lose or share is determined by the _____________tha ...
Period 3 element
A period 3 element is one of the chemical elements in the third row (or period) of the periodic table of the chemical elements. The periodic table is laid out in rows to illustrate recurring (periodic) trends in the chemical behaviour of the elements as their atomic number increases: a new row is begun when the periodic table skips a row and a chemical behaviour begins to repeat, meaning that elements with similar behavior fall into the same vertical columns. The third period contains eight elements: sodium, magnesium, aluminium, silicon, phosphorus, sulfur, chlorine, and argon. The first two, sodium and magnesium, are members of the s-block of the periodic table, while the others are members of the p-block. Note that there is a 3d orbital, but it is not filled until Period 4, such giving the period table its characteristic shape of ""two rows at a time"". All of the period 3 elements occur in nature and have at least one stable isotope.