oxidation number
... metals (+2), Ag+, Zn2+, Cd2+, and Al3+ within compounds is a set value. • Hydrogen (H) has two possible oxidation numbers: – +1 when bonded to a nonmetal – -1 when bonded to a metal ...
... metals (+2), Ag+, Zn2+, Cd2+, and Al3+ within compounds is a set value. • Hydrogen (H) has two possible oxidation numbers: – +1 when bonded to a nonmetal – -1 when bonded to a metal ...
The Bohr Atom The Bohr Atom Electronic Transitions 2.3 Light, Atom
... • Electrons are found only in these energy levels • Highest-energy orbits are farthest from the ...
... • Electrons are found only in these energy levels • Highest-energy orbits are farthest from the ...
Unit 1-4-Periodic Trends - Free WonderKids-e
... qualitative property, there is not a standardized method for calculating electronegativity. However, the scale that most chemists use in quantifying electronegativity is the Pauling Scale, named after the chemist Linus Pauling. The numbers assigned by the Pauling scale are dimensionless due to elect ...
... qualitative property, there is not a standardized method for calculating electronegativity. However, the scale that most chemists use in quantifying electronegativity is the Pauling Scale, named after the chemist Linus Pauling. The numbers assigned by the Pauling scale are dimensionless due to elect ...
Science - ExamResults.net
... Solve examples, Distinguish, Complete the table, Write characteristics, Write uses. Q. 3 and Q. 7 Give two examples and explain any one, Write law / definition and explain with example, Write meritsdemerits, Explain. Q. 4 and Q. 8 ...
... Solve examples, Distinguish, Complete the table, Write characteristics, Write uses. Q. 3 and Q. 7 Give two examples and explain any one, Write law / definition and explain with example, Write meritsdemerits, Explain. Q. 4 and Q. 8 ...
Std 10th, Science and Technology, Maharashtra Board, English
... Solve examples, Distinguish, Complete the table, Write characteristics, Write uses. Q. 3 and Q. 7 Give two examples and explain any one, Write law / definition and explain with example, Write meritsdemerits, Explain. Q. 4 and Q. 8 ...
... Solve examples, Distinguish, Complete the table, Write characteristics, Write uses. Q. 3 and Q. 7 Give two examples and explain any one, Write law / definition and explain with example, Write meritsdemerits, Explain. Q. 4 and Q. 8 ...
Atomic Nucleus and Isotopes
... roots in many concepts within chemistry and biological sciences. The properties of atoms such as their atomic and mass number are important in understanding electron configuration sequences, which also ties into bond formation between atoms to form molecules. Ionic and covalent bonds, for example de ...
... roots in many concepts within chemistry and biological sciences. The properties of atoms such as their atomic and mass number are important in understanding electron configuration sequences, which also ties into bond formation between atoms to form molecules. Ionic and covalent bonds, for example de ...
oxidation number
... Determining Oxidation Numbers • Metals form positive ions (7+ is the highest positive oxidation number). • Nonmetals tend to form negative ions. – However other than the noble gases which have an oxidation number of zero, and fluoride (F-) which is always a 1-, all other nonmetals could have positi ...
... Determining Oxidation Numbers • Metals form positive ions (7+ is the highest positive oxidation number). • Nonmetals tend to form negative ions. – However other than the noble gases which have an oxidation number of zero, and fluoride (F-) which is always a 1-, all other nonmetals could have positi ...
Quantum Thorj and th Atom - Crossroads chemistry 40S
... electron can move only from one allowable orbit to another, and therefore, can emit or absorb only certain amounts of energy. I The four electron transitions that account for visible lines in hydrogen’s atomic emission spectrum are shown in Figure 5-lOb. For example, electrons dropping from the thir ...
... electron can move only from one allowable orbit to another, and therefore, can emit or absorb only certain amounts of energy. I The four electron transitions that account for visible lines in hydrogen’s atomic emission spectrum are shown in Figure 5-lOb. For example, electrons dropping from the thir ...
Lecture 12
... Molecular mass is the sum of the masses of all atoms in a molecule Formula mass is the sum of the masses of all atoms in a formula unit of an ionic compound (or molecule - used ...
... Molecular mass is the sum of the masses of all atoms in a molecule Formula mass is the sum of the masses of all atoms in a formula unit of an ionic compound (or molecule - used ...
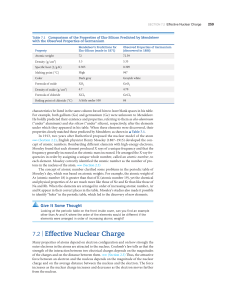
7.2 | Effective Nuclear Charge
... Moving from one element to the next in the periodic table, the increase in Zeff felt by the innermost (1s) electrons (red circles) closely tracks the increase in nuclear charge Z (black line) because these electrons are not screened much. The results of several methods to calculate Zeff for valence ...
... Moving from one element to the next in the periodic table, the increase in Zeff felt by the innermost (1s) electrons (red circles) closely tracks the increase in nuclear charge Z (black line) because these electrons are not screened much. The results of several methods to calculate Zeff for valence ...
Document
... not the first scientist to organise a seemingly unconnected mass of information into a Periodic Table based on the properties of elements. However Mendeleev was the first scientist to use a Periodic Table to predict the properties of unknown metals needed to fill gaps in the table. His 1869 predicti ...
... not the first scientist to organise a seemingly unconnected mass of information into a Periodic Table based on the properties of elements. However Mendeleev was the first scientist to use a Periodic Table to predict the properties of unknown metals needed to fill gaps in the table. His 1869 predicti ...
Unit 1: Lecture Notes
... 1e. Students know the nucleus of the atom is much smaller than the atom yet contains most of its mass. The volume of the hydrogen nucleus is about one trillion times less than the volume of the hydrogen atom, yet the nucleus contains almost all the mass in the form of one proton. The diameter of an ...
... 1e. Students know the nucleus of the atom is much smaller than the atom yet contains most of its mass. The volume of the hydrogen nucleus is about one trillion times less than the volume of the hydrogen atom, yet the nucleus contains almost all the mass in the form of one proton. The diameter of an ...
Graphing Periodic Trends
... Graphing Periodic Trends Introduction: Elements on the periodic table are arranged in such a way that they exhibit patterns in their properties. In this activity, you will graph their properties and analyze their patterns. You will determine the Periodic Trends for the following properties: atomic r ...
... Graphing Periodic Trends Introduction: Elements on the periodic table are arranged in such a way that they exhibit patterns in their properties. In this activity, you will graph their properties and analyze their patterns. You will determine the Periodic Trends for the following properties: atomic r ...
Section 2 Electron Configuration and the Periodic Table Chapter 5
... • Group 2 metals are less reactive than the alkali metals, but are still too reactive to be found in nature in pure form. Chapter menu ...
... • Group 2 metals are less reactive than the alkali metals, but are still too reactive to be found in nature in pure form. Chapter menu ...
These are the periodic trends we need to know
... of these magnificent brains, there is quite a marvelous story to tell. All right, you must first understand the times, and what was, and was not, known. Around 1908, it is pretty well established that (there are): negative particles called electrons positive particles called alpha particles el ...
... of these magnificent brains, there is quite a marvelous story to tell. All right, you must first understand the times, and what was, and was not, known. Around 1908, it is pretty well established that (there are): negative particles called electrons positive particles called alpha particles el ...
Ch09_Clicker_Questions - Saint Leo University Faculty
... A small portion of elements on Earth originated in the Sun. An even smaller portion are synthesized in laboratories. Most atoms beyond hydrogen, overwhelmingly, were manufactured in the stars. We really are made of stardust! © 2013 Pearson Education, Inc. ...
... A small portion of elements on Earth originated in the Sun. An even smaller portion are synthesized in laboratories. Most atoms beyond hydrogen, overwhelmingly, were manufactured in the stars. We really are made of stardust! © 2013 Pearson Education, Inc. ...
Problem Solving Drill - Rapid Learning Center
... College Chemistry - Problem Drill 14: The Periodic Table and Chemical Periodicity Question No. 1 of 10 Instructions: (1) Read the problem and answer choices carefully (2) Work the problems on paper as needed (3) Pick the answer (4) Go back to review the core concept tutorial as needed. 1. Atomic rad ...
... College Chemistry - Problem Drill 14: The Periodic Table and Chemical Periodicity Question No. 1 of 10 Instructions: (1) Read the problem and answer choices carefully (2) Work the problems on paper as needed (3) Pick the answer (4) Go back to review the core concept tutorial as needed. 1. Atomic rad ...
Chapter 7 Electron Configuration and the
... – Groups 3A - 7A show considerable variation among properties from metallic to nonmetallic – Transition metals do not always exhibit regular patterns in their electron configurations but have some similarities as a whole such as colored compounds and multiple oxidation states. ...
... – Groups 3A - 7A show considerable variation among properties from metallic to nonmetallic – Transition metals do not always exhibit regular patterns in their electron configurations but have some similarities as a whole such as colored compounds and multiple oxidation states. ...
the PDF
... Topic 3 – Patterns in the Periodic Table The Periodic Table The modern periodic table is based on that first drawn up by the Russian chemist, Mendeleev, in about 1870. He listed the known elements in order of increasing atomic mass and then arranged them so that those with similar chemical propertie ...
... Topic 3 – Patterns in the Periodic Table The Periodic Table The modern periodic table is based on that first drawn up by the Russian chemist, Mendeleev, in about 1870. He listed the known elements in order of increasing atomic mass and then arranged them so that those with similar chemical propertie ...
Section 6 Ionization Energy and Orbitals
... periodic table, as you go from left to right across a row, the atomic radii of the elements become smaller. In the same way, as you go from left to right across a row, the ionization energy of the elements increases. This is because the nuclear charge is increasing, placing a stronger electrostatic ...
... periodic table, as you go from left to right across a row, the atomic radii of the elements become smaller. In the same way, as you go from left to right across a row, the ionization energy of the elements increases. This is because the nuclear charge is increasing, placing a stronger electrostatic ...
60. Write the electron configuration for Zn
... Atomic Structure/Periodic Trends 34. What are the five postulates of Dalton’s atomic theory? 1. Elements are made of tiny particles called atoms. 2. All atoms of a given element are identical 3. The atoms of a given element are different from those of any other element. 4. Atoms of one element can c ...
... Atomic Structure/Periodic Trends 34. What are the five postulates of Dalton’s atomic theory? 1. Elements are made of tiny particles called atoms. 2. All atoms of a given element are identical 3. The atoms of a given element are different from those of any other element. 4. Atoms of one element can c ...
Spring Semester
... b. Solution: Find moles of KOH .025L x .150mols/1L = .00375 mols KOH 1:1 Ratio between KOH and HCl so .00375 mols HCl .00375 mols HCl x 1L/.175 mols = .0214L or 21.4mL Atomic Structure/Periodic Trends 45. What are the five postulates of Dalton’s atomic theory? 1. Elements are made of tiny particles ...
... b. Solution: Find moles of KOH .025L x .150mols/1L = .00375 mols KOH 1:1 Ratio between KOH and HCl so .00375 mols HCl .00375 mols HCl x 1L/.175 mols = .0214L or 21.4mL Atomic Structure/Periodic Trends 45. What are the five postulates of Dalton’s atomic theory? 1. Elements are made of tiny particles ...
OXIDATION AND REDUCTION
... Oxidation Number is the valency of an atom in a molecule or ion which is assigned the sign either + or -. It may be (i)electrovalency or (ii)covalency . When the proper sign is associated with valency it becomes oxidation number(ON). It is mostly a theoretical concept particularly for covalent compo ...
... Oxidation Number is the valency of an atom in a molecule or ion which is assigned the sign either + or -. It may be (i)electrovalency or (ii)covalency . When the proper sign is associated with valency it becomes oxidation number(ON). It is mostly a theoretical concept particularly for covalent compo ...
Period 3 element
A period 3 element is one of the chemical elements in the third row (or period) of the periodic table of the chemical elements. The periodic table is laid out in rows to illustrate recurring (periodic) trends in the chemical behaviour of the elements as their atomic number increases: a new row is begun when the periodic table skips a row and a chemical behaviour begins to repeat, meaning that elements with similar behavior fall into the same vertical columns. The third period contains eight elements: sodium, magnesium, aluminium, silicon, phosphorus, sulfur, chlorine, and argon. The first two, sodium and magnesium, are members of the s-block of the periodic table, while the others are members of the p-block. Note that there is a 3d orbital, but it is not filled until Period 4, such giving the period table its characteristic shape of ""two rows at a time"". All of the period 3 elements occur in nature and have at least one stable isotope.