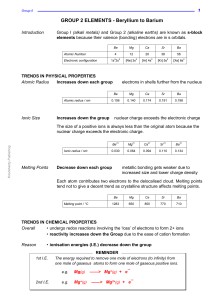
The s-Block Elements
... Reactions of chlorides 1. All group I chlorides are ionic and readily soluble in water. No hydrolysis occurs. 2. Group II chlorides show some degree of covalent character. Beryllium chloride is covalent and hydrolysis to form Be(OH)2(s) and HCl(aq). Magnesium chloride is intermediate, it dissolves ...
... Reactions of chlorides 1. All group I chlorides are ionic and readily soluble in water. No hydrolysis occurs. 2. Group II chlorides show some degree of covalent character. Beryllium chloride is covalent and hydrolysis to form Be(OH)2(s) and HCl(aq). Magnesium chloride is intermediate, it dissolves ...
Title - Iowa State University
... 6) Which of the following are insoluble? I.Ag2SO4 II. Ni3PO4 III. NaI a) I only b) II only c) III only d) I and II e)I, II and II 7) Aqueous sodium carbonate reacts with barium chloride Molecular equation: Ionic equation: Spectator ions: Net ionic equation: 1060 Hixson-Lied Student Success Center ...
... 6) Which of the following are insoluble? I.Ag2SO4 II. Ni3PO4 III. NaI a) I only b) II only c) III only d) I and II e)I, II and II 7) Aqueous sodium carbonate reacts with barium chloride Molecular equation: Ionic equation: Spectator ions: Net ionic equation: 1060 Hixson-Lied Student Success Center ...
Name ______ Write formulas for the reactants and predicted
... Write formulas for the reactants and predicted products for the chemical reactions that follow. Assume that in all cases a reaction occurs. The equation must be balanced. Write all substances in their proper form –as ions if appropriate – and cancel any spectator ions. ...
... Write formulas for the reactants and predicted products for the chemical reactions that follow. Assume that in all cases a reaction occurs. The equation must be balanced. Write all substances in their proper form –as ions if appropriate – and cancel any spectator ions. ...
1.0 basic concepts
... • This means that water, H2O cannot be formed, therefore H2 is the product • If you look at the reactants in (a) – (d), you’ll notice that the metal has oxygen present. • This means that water, H2O can be formed, therefore H2O is the product Carbon dioxide or not • If you look at the reactants in (d ...
... • This means that water, H2O cannot be formed, therefore H2 is the product • If you look at the reactants in (a) – (d), you’ll notice that the metal has oxygen present. • This means that water, H2O can be formed, therefore H2O is the product Carbon dioxide or not • If you look at the reactants in (d ...
experiment 10 - Faculty Web Pages
... Introduction: You will study double displacement reactions using a small-scale method and predict the products of double displacement reactions. Background: You will combine two water solutions, each containing positive and negative ions. Consider this generalized reaction between two ionic compound ...
... Introduction: You will study double displacement reactions using a small-scale method and predict the products of double displacement reactions. Background: You will combine two water solutions, each containing positive and negative ions. Consider this generalized reaction between two ionic compound ...
Writing Net Ionic Equations
... HCl (aq) + H2O (l) → H3O+ (aq) + Cl- (aq) This reaction may be abbreviated as: HCl (aq) → H+ (aq) + Cl- (aq) 2. The common strong bases are the soluble hydroxides (those of Group IA elements and Ba2+) and the slightly soluble hydroxides (those of Ca2+ and Sr2+). Strong bases, like strong acids, are ...
... HCl (aq) + H2O (l) → H3O+ (aq) + Cl- (aq) This reaction may be abbreviated as: HCl (aq) → H+ (aq) + Cl- (aq) 2. The common strong bases are the soluble hydroxides (those of Group IA elements and Ba2+) and the slightly soluble hydroxides (those of Ca2+ and Sr2+). Strong bases, like strong acids, are ...
activity series
... 4. Before beginning to balance an equation, check each formula to see that it is correct. NEVER change a formula during the balancing of an equation. 5. Balancing is done by placing coefficients in front of the formulas to insure the same number of atoms of each element on both sides of the arrow. 6 ...
... 4. Before beginning to balance an equation, check each formula to see that it is correct. NEVER change a formula during the balancing of an equation. 5. Balancing is done by placing coefficients in front of the formulas to insure the same number of atoms of each element on both sides of the arrow. 6 ...
ATOMS, MOLECULES, AND IONS
... positively charged, compact, heavy nucleus. The charge on the atomic nucleus is +Ze (Z is the atomic number of the atom). The fundamental unit of positive charge in the nucleus is the proton. ♦ Chemical identity of an atom is determined by the number of protons. ♦ The nucleus also contains neutral p ...
... positively charged, compact, heavy nucleus. The charge on the atomic nucleus is +Ze (Z is the atomic number of the atom). The fundamental unit of positive charge in the nucleus is the proton. ♦ Chemical identity of an atom is determined by the number of protons. ♦ The nucleus also contains neutral p ...
LN_atoms_etc
... charged particles within an atom. Electrons have a charge of e = 1.6021773 10–19 C and a mass of 9.109390 10–31 kg. Later experiments by Rutherford determined that at the center of an atom is a positively charged, compact, heavy nucleus. The charge on the atomic nucleus is +Ze (Z is the atomic n ...
... charged particles within an atom. Electrons have a charge of e = 1.6021773 10–19 C and a mass of 9.109390 10–31 kg. Later experiments by Rutherford determined that at the center of an atom is a positively charged, compact, heavy nucleus. The charge on the atomic nucleus is +Ze (Z is the atomic n ...
AP Chemistry Summer Assignment
... g. Potassium Chloride. 42. The hormone, thyroxine is secreted by the thyroid gland, and has the formula: C15H17NO4I4. How many milligrams of Iodine can be extracted from 15.0 Grams of thyroxine? 43. Determine the formula weight for the following: a. N2O5 b. CuSO4 C. Ca(HCO3)2 d. CaSO4 . 2 H2O 44. Wr ...
... g. Potassium Chloride. 42. The hormone, thyroxine is secreted by the thyroid gland, and has the formula: C15H17NO4I4. How many milligrams of Iodine can be extracted from 15.0 Grams of thyroxine? 43. Determine the formula weight for the following: a. N2O5 b. CuSO4 C. Ca(HCO3)2 d. CaSO4 . 2 H2O 44. Wr ...
Notes on QA - Scarsdale Public Schools
... mixture of dozens of cations based on their insolubility as chloride salts. Hence the Group 1 cations consist of silver, lead(II) and mercury(II) ions which all form insoluble chlorides. The Group II cations are those that form insoluble sulfides in acidic solution, while the Group III cations form ...
... mixture of dozens of cations based on their insolubility as chloride salts. Hence the Group 1 cations consist of silver, lead(II) and mercury(II) ions which all form insoluble chlorides. The Group II cations are those that form insoluble sulfides in acidic solution, while the Group III cations form ...
Midterm 2 from Summer 2012
... Suppose I synthesize 0.35 mole of Cuprous Chloride, CuCl. What mass of product ...
... Suppose I synthesize 0.35 mole of Cuprous Chloride, CuCl. What mass of product ...
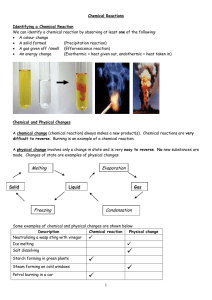
Chemical Reactions
... The reactants are separated from each other by a plus sign and the products are separated from each other by a plus sign. There should be an arrow in the middle. Examples: When sodium is mixed with water, a purple alkaline solution of sodium hydroxide is produced and hydrogen gas is evolved. Sodium ...
... The reactants are separated from each other by a plus sign and the products are separated from each other by a plus sign. There should be an arrow in the middle. Examples: When sodium is mixed with water, a purple alkaline solution of sodium hydroxide is produced and hydrogen gas is evolved. Sodium ...
final exam review chapter 1-4
... Proton versus Neutrons versus Electrons Percent abundance, and mass spectrometry Periodic trends (diatomic, metals, nonmetals, metalloids, cations versus anions) Ionic (transferring of electrons) versus covalent (shared electrons) Naming: ionic, covalent, ions (oxy-ions), acids (binary and ...
... Proton versus Neutrons versus Electrons Percent abundance, and mass spectrometry Periodic trends (diatomic, metals, nonmetals, metalloids, cations versus anions) Ionic (transferring of electrons) versus covalent (shared electrons) Naming: ionic, covalent, ions (oxy-ions), acids (binary and ...
No Slide Title
... 2. The active agent in many hair bleaches is hydrogen peroxide. The amount of hydrogen peroxide in 13.8 g of hair bleach was determined by titration with a standard potassium permanganate solution. Unbalanced equation: ...
... 2. The active agent in many hair bleaches is hydrogen peroxide. The amount of hydrogen peroxide in 13.8 g of hair bleach was determined by titration with a standard potassium permanganate solution. Unbalanced equation: ...
S3 Chemistry - eduBuzz.org
... State whether covalent substances form discrete molecular or giant network structures Predict the typical properties of discrete molecular and giant network covalent structures State that ionic substances form giant lattices of oppositely charged ions and predict the typical properties of ioni ...
... State whether covalent substances form discrete molecular or giant network structures Predict the typical properties of discrete molecular and giant network covalent structures State that ionic substances form giant lattices of oppositely charged ions and predict the typical properties of ioni ...
S3 Chemistry - eduBuzz.org
... State whether covalent substances form discrete molecular or giant network structures Predict the typical properties of discrete molecular and giant network covalent structures State that ionic substances form giant lattices of oppositely charged ions and predict the typical properties of ioni ...
... State whether covalent substances form discrete molecular or giant network structures Predict the typical properties of discrete molecular and giant network covalent structures State that ionic substances form giant lattices of oppositely charged ions and predict the typical properties of ioni ...
Hydroxide
Hydroxide is a diatomic anion with chemical formula OH−. It consists of an oxygen and hydrogen atom held together by a covalent bond, and carries a negative electric charge. It is an important but usually minor constituent of water. It functions as a base, a ligand, a nucleophile and a catalyst. The hydroxide ion forms salts, some of which dissociate in aqueous solution, liberating solvated hydroxide ions. Sodium hydroxide is a multi-million-ton per annum commodity chemical. A hydroxide attached to a strongly electropositive center may itself ionize, liberating a hydrogen cation (H+), making the parent compound an acid.The corresponding electrically neutral compound •HO is the hydroxyl radical. The corresponding covalently-bound group -OH of atoms is the hydroxyl group.Hydroxide ion and hydroxyl group are nucleophiles and can act as a catalyst in organic chemistry.Many inorganic substances which bear the word ""hydroxide"" in their names are not ionic compounds of the hydroxide ion, but covalent compounds which contain hydroxyl groups.