Document
... How many moles of CO2 can be produced from the combustion of 8.7 mol octane (C8H18)? (Use coefficients from balanced chemical equation to ...
... How many moles of CO2 can be produced from the combustion of 8.7 mol octane (C8H18)? (Use coefficients from balanced chemical equation to ...
Question paper - Edexcel
... Answer ALL the questions in this section. You should aim to spend no more than 20 minutes on this section. For each question, select one answer from A to D and put a cross in the box . and then mark your new answer with If you change your mind, put a line through the box a cross . 1 Methods for inve ...
... Answer ALL the questions in this section. You should aim to spend no more than 20 minutes on this section. For each question, select one answer from A to D and put a cross in the box . and then mark your new answer with If you change your mind, put a line through the box a cross . 1 Methods for inve ...
Chapter 4
... BaCI2 (aq) + Na2SO4 (aq) Ba2+(aq) + 2Cl-(aq) + 2Na+(aq)+ SO4 2-(aq) 2. Match cation from one salt with the anion from the other salt” Ba2+(aq) + Cl-(aq) + Na+(aq)+ SO4 2-(aq) NaCl+ BaSO4 Note: Always keep the metal on the left in all salts! 3. Balance charges in salts and put in coefficients Ba2+( ...
... BaCI2 (aq) + Na2SO4 (aq) Ba2+(aq) + 2Cl-(aq) + 2Na+(aq)+ SO4 2-(aq) 2. Match cation from one salt with the anion from the other salt” Ba2+(aq) + Cl-(aq) + Na+(aq)+ SO4 2-(aq) NaCl+ BaSO4 Note: Always keep the metal on the left in all salts! 3. Balance charges in salts and put in coefficients Ba2+( ...
HIGHER TIER CHEMISTRY MINI-MOCK UNIT 2
... Electrolysis can be used to remove unwanted hair from the skin. The hair is first coated with a layer of gel containing ions in solution. The positive electrode is connected by a patch to the skin.The negative electrode is connected to the hair. Electricity flows through the gel and causes electroly ...
... Electrolysis can be used to remove unwanted hair from the skin. The hair is first coated with a layer of gel containing ions in solution. The positive electrode is connected by a patch to the skin.The negative electrode is connected to the hair. Electricity flows through the gel and causes electroly ...
CHEMICAL REACTIONS
... aided by the addition of a small amount of an inorganic acid such as sulfuric to act as a dehydrating agent. • e. g. CH3COOH + CH3OH Æ CH3COOCH3 + HOH H. Complex ion reactions (coordination chemistry) : Usually involve transition metals and common ligands (see below). Since many complex ion reaction ...
... aided by the addition of a small amount of an inorganic acid such as sulfuric to act as a dehydrating agent. • e. g. CH3COOH + CH3OH Æ CH3COOCH3 + HOH H. Complex ion reactions (coordination chemistry) : Usually involve transition metals and common ligands (see below). Since many complex ion reaction ...
Writing And Balancing Equations
... The compounds Ca(OH)2, H2SO4, CaSO4, and H2O are like correctly spelled and chosen words ...
... The compounds Ca(OH)2, H2SO4, CaSO4, and H2O are like correctly spelled and chosen words ...
File
... Identify one isomer that will react with aqueous sodium hydroxide almost exclusively by an SN2 mechanism. Draw the mechanism for this reaction using curly arrows to represent the movement of electron pairs. Include the structural formulas of the transition state and the organic product. ...
... Identify one isomer that will react with aqueous sodium hydroxide almost exclusively by an SN2 mechanism. Draw the mechanism for this reaction using curly arrows to represent the movement of electron pairs. Include the structural formulas of the transition state and the organic product. ...
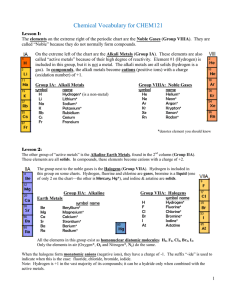
Vocabulary CHEM121
... All the elements in this group exist as homonuclear diatomic molecules: H2, F2, Cl2, Br2, I2. Only the elements in air (Oxygen*, O2 and Nitrogen*, N2) do the same. When the halogens form monatomic anions (negative ions), they have a charge of -1. The suffix “-ide” is used to indicate when this is th ...
... All the elements in this group exist as homonuclear diatomic molecules: H2, F2, Cl2, Br2, I2. Only the elements in air (Oxygen*, O2 and Nitrogen*, N2) do the same. When the halogens form monatomic anions (negative ions), they have a charge of -1. The suffix “-ide” is used to indicate when this is th ...
Exam 2, Fall 2001
... 2. (3 points) You wish to make a 0.125 M hydrochloric acid solution from 12.0 M hydrochloric acid (concentrated HCl). How much concentrated acid must you add to obtain a total volume of 100 mL of the dilute solution ? (a) 0.00104 mL (b) 1.04 mL (c) 120 mL (d) 150 mL 3. (4 points) You have a 0.207 M ...
... 2. (3 points) You wish to make a 0.125 M hydrochloric acid solution from 12.0 M hydrochloric acid (concentrated HCl). How much concentrated acid must you add to obtain a total volume of 100 mL of the dilute solution ? (a) 0.00104 mL (b) 1.04 mL (c) 120 mL (d) 150 mL 3. (4 points) You have a 0.207 M ...
Chapter 4 (Hill/Petrucci/McCreary/Perry Chemical Reactions in
... <<100% ionization in water = weak base Most common weak bases: NH3 and amines Reaction of Acids with Bases: Neutralization neutralization reaction: the reaction of ionizable H+ ions on acid molecules with OH1- or other anions (such as HCO3 1- or CO3 2-) on base “molecules” Example. We represent an a ...
... <<100% ionization in water = weak base Most common weak bases: NH3 and amines Reaction of Acids with Bases: Neutralization neutralization reaction: the reaction of ionizable H+ ions on acid molecules with OH1- or other anions (such as HCO3 1- or CO3 2-) on base “molecules” Example. We represent an a ...
Name……………………………………............................. Index number
... Write your name, index number class and admission number in the spaces provided Sign and write the date of examination in the spaces provided. Answer all the questions in the spaces provided. Mathematical tables and silent electronic calculators may be used. All working must be clearly shown where n ...
... Write your name, index number class and admission number in the spaces provided Sign and write the date of examination in the spaces provided. Answer all the questions in the spaces provided. Mathematical tables and silent electronic calculators may be used. All working must be clearly shown where n ...
Old EXAM I - gozips.uakron.edu
... III. A compound is formed when atoms of different elements combine in a specific ratio. IV. Atoms of an element can not be converted into atoms of other elements. Which of these postulates have not required modification since 1808? (a) (b) (c) (d) (e) ...
... III. A compound is formed when atoms of different elements combine in a specific ratio. IV. Atoms of an element can not be converted into atoms of other elements. Which of these postulates have not required modification since 1808? (a) (b) (c) (d) (e) ...
AP Chemistry - Shoreline Public Schools
... There are several reasons why a student might want to take AP Chemistry, including (but not limited to) the following. 1. AP Chemistry will challenge you to the limits of your academic ability. In the past you may have found classes “too easy”, and therefore not stimulated you do your very best. Thi ...
... There are several reasons why a student might want to take AP Chemistry, including (but not limited to) the following. 1. AP Chemistry will challenge you to the limits of your academic ability. In the past you may have found classes “too easy”, and therefore not stimulated you do your very best. Thi ...
Chapter 4
... The charge the atom would have in a molecule (or an ionic compound) if electrons were completely transferred. 1. Free elements (uncombined state) have an oxidation number of zero. ...
... The charge the atom would have in a molecule (or an ionic compound) if electrons were completely transferred. 1. Free elements (uncombined state) have an oxidation number of zero. ...
AP Chemistry Note Outline
... different element) is zero, regardless of how complex its molecules might be. The oxidation number for any simple, monoatomic ion is equal to the charge on the ion. The sum of all the oxidation numbers of the atoms in a molecule or ion must be equal the charge of the particle. In its compound, ...
... different element) is zero, regardless of how complex its molecules might be. The oxidation number for any simple, monoatomic ion is equal to the charge on the ion. The sum of all the oxidation numbers of the atoms in a molecule or ion must be equal the charge of the particle. In its compound, ...
Unit A Review Questions
... and are being reduced by the zinc metal being oxidized. This would also account for the colour change in the copper nitrate solution. As the copper ions come out of the solution, the solution becomes a fainter colour. b. The voltage reading is zero because the electrons do not have to travel through ...
... and are being reduced by the zinc metal being oxidized. This would also account for the colour change in the copper nitrate solution. As the copper ions come out of the solution, the solution becomes a fainter colour. b. The voltage reading is zero because the electrons do not have to travel through ...
Hints for Names and Formulas (Ch. 4 in Zumdahl Chemistry)
... ● IUPAC stands for the International Union of Pure and Applied Chemistry committee (9) most covalent inorganic molecules are gases (sometimes liquids) at room temperature (R.T.) IONIC COMPOUNDS (1) ionic compounds are never called molecules and have covalent bonds only in their polyatomic ions (2) i ...
... ● IUPAC stands for the International Union of Pure and Applied Chemistry committee (9) most covalent inorganic molecules are gases (sometimes liquids) at room temperature (R.T.) IONIC COMPOUNDS (1) ionic compounds are never called molecules and have covalent bonds only in their polyatomic ions (2) i ...
Final Exam Review Packet
... ____ 45. Sulfuric acid is probably the most important industrial chemical because it is used in so many industrial processes to produce or purify other chemicals. It can be produced by a three step process. First, sulfur is burned in air to give sulfur dioxide. Second, the sulfur dioxide is converte ...
... ____ 45. Sulfuric acid is probably the most important industrial chemical because it is used in so many industrial processes to produce or purify other chemicals. It can be produced by a three step process. First, sulfur is burned in air to give sulfur dioxide. Second, the sulfur dioxide is converte ...
FREE Sample Here
... Chemicals used as reagents, such as bromthymol blue or sodium iodide, may permanently stain clothing. Use with caution. ...
... Chemicals used as reagents, such as bromthymol blue or sodium iodide, may permanently stain clothing. Use with caution. ...
AQA C2 revision book
... held together by strong forces called covalent bonds, but there are only very weak forces between the molecules. This means: 1) They have low melting and boiling points (many are liquids or gases). 2) They tend to be soft and/or have little strength. 3) They do not conduct electricity Simple molecul ...
... held together by strong forces called covalent bonds, but there are only very weak forces between the molecules. This means: 1) They have low melting and boiling points (many are liquids or gases). 2) They tend to be soft and/or have little strength. 3) They do not conduct electricity Simple molecul ...
Hydroxide
Hydroxide is a diatomic anion with chemical formula OH−. It consists of an oxygen and hydrogen atom held together by a covalent bond, and carries a negative electric charge. It is an important but usually minor constituent of water. It functions as a base, a ligand, a nucleophile and a catalyst. The hydroxide ion forms salts, some of which dissociate in aqueous solution, liberating solvated hydroxide ions. Sodium hydroxide is a multi-million-ton per annum commodity chemical. A hydroxide attached to a strongly electropositive center may itself ionize, liberating a hydrogen cation (H+), making the parent compound an acid.The corresponding electrically neutral compound •HO is the hydroxyl radical. The corresponding covalently-bound group -OH of atoms is the hydroxyl group.Hydroxide ion and hydroxyl group are nucleophiles and can act as a catalyst in organic chemistry.Many inorganic substances which bear the word ""hydroxide"" in their names are not ionic compounds of the hydroxide ion, but covalent compounds which contain hydroxyl groups.