American-Journal-of-Oil-and-Chemical-Technologies
... In conclusion, continuing with our previous works on synthesizing supramolecular compounds containing pyridinedicarboxylic acid N-oxides [9-11], two new coordination complexes have been synthesized and characterized. The red shift of bands ʋas(COO−), ʋs(COO−), and ʋas(COO−) and ʋ(NO) confirm formati ...
... In conclusion, continuing with our previous works on synthesizing supramolecular compounds containing pyridinedicarboxylic acid N-oxides [9-11], two new coordination complexes have been synthesized and characterized. The red shift of bands ʋas(COO−), ʋs(COO−), and ʋas(COO−) and ʋ(NO) confirm formati ...
Topic 2 notes - WordPress.com
... Chemical equation: CaO (s) + H2O (l) Ca(OH)2 (s) In this reaction calcium oxide dissolves in water to form calcium hydroxide – a crumbly, white solid... o Heat is given off o Fizzing o Steam is produced When more water is added, calcium hydroxide dissolves to form a colourless calcium hydroxide so ...
... Chemical equation: CaO (s) + H2O (l) Ca(OH)2 (s) In this reaction calcium oxide dissolves in water to form calcium hydroxide – a crumbly, white solid... o Heat is given off o Fizzing o Steam is produced When more water is added, calcium hydroxide dissolves to form a colourless calcium hydroxide so ...
RULES OF CHEMICAL NOMENCLATURE I. Elements (periodic
... (because ‘+’ is always first), nonmetal is last. B. Named by adding name of first element (metal) to second element (non-metal) whose name is modified to end in “ide.” (metals are to the left of the staircase; non-metals are to the right in periodic table.) ...
... (because ‘+’ is always first), nonmetal is last. B. Named by adding name of first element (metal) to second element (non-metal) whose name is modified to end in “ide.” (metals are to the left of the staircase; non-metals are to the right in periodic table.) ...
Formulae and equations
... A substance which cannot be split into anything simpler by chemical means. ...
... A substance which cannot be split into anything simpler by chemical means. ...
Chemical Reactions
... we were to decrease pressure by increasing volume, the equilibrium of the above reaction will shift to the left, because the reactant side has greater number of moles than does the product side. The system tries to counteract the decrease in partial pressure of gas molecules by shifting to the side ...
... we were to decrease pressure by increasing volume, the equilibrium of the above reaction will shift to the left, because the reactant side has greater number of moles than does the product side. The system tries to counteract the decrease in partial pressure of gas molecules by shifting to the side ...
Click to download. - Life Learning Cloud
... molecules are held together by strong forces called covalent bonds, but there are only very weak forces between the molecules( weak intermolecular forces). This means: 1) They have low melting and boiling points (many are liquids or gases). 2) They tend to be soft and/or have little strength. 3) The ...
... molecules are held together by strong forces called covalent bonds, but there are only very weak forces between the molecules( weak intermolecular forces). This means: 1) They have low melting and boiling points (many are liquids or gases). 2) They tend to be soft and/or have little strength. 3) The ...
Chapter 4 - GEOCITIES.ws
... the attractions between the ions are so large that the water molecule will not separate the ion, and the substance remains mostly ...
... the attractions between the ions are so large that the water molecule will not separate the ion, and the substance remains mostly ...
handout 4
... Lecture Example: Baking soda (NaHCO3) is used as an antacid. It neutralizes HCl in the stomach. How many grams of HCl are neutralized per 1.00 gram of baking soda? If all the baking soda reacts, how many grams of water is produced? ...
... Lecture Example: Baking soda (NaHCO3) is used as an antacid. It neutralizes HCl in the stomach. How many grams of HCl are neutralized per 1.00 gram of baking soda? If all the baking soda reacts, how many grams of water is produced? ...
AQA GCSE Chemistry My Revision Notes
... A school chemistry technician was tidying up in the outside chemical store. Four bottles of chemicals were found where the labels were unclear. The technician thought they were potassium carbonate, potassium chloride, potassium sulfate and aluminium sulfate. The following reagents were readily avail ...
... A school chemistry technician was tidying up in the outside chemical store. Four bottles of chemicals were found where the labels were unclear. The technician thought they were potassium carbonate, potassium chloride, potassium sulfate and aluminium sulfate. The following reagents were readily avail ...
SAMPLE PAPER -2 Time Allowed: 3 Hrs
... (b) State Henry's law. Give its mathematical expression and write two applications of it. Q.29 (a) Account for the following: (i) Transition elements show highest oxidation state in their oxides than fluorides. (ii) Cu has positive electrode potential in the first transition series. (b)Write balance ...
... (b) State Henry's law. Give its mathematical expression and write two applications of it. Q.29 (a) Account for the following: (i) Transition elements show highest oxidation state in their oxides than fluorides. (ii) Cu has positive electrode potential in the first transition series. (b)Write balance ...
X012/11/02
... B OH–(aq) ions decreases C H+(aq) ions and the concentration of OH–(aq) ions become equal D H+(aq) ions and the concentration of OH–(aq) ions remain unchanged. ...
... B OH–(aq) ions decreases C H+(aq) ions and the concentration of OH–(aq) ions become equal D H+(aq) ions and the concentration of OH–(aq) ions remain unchanged. ...
Arrhenius theory of ionization and its factors
... A gas that is composed of nearly equal numbers of negative and positive ions is called a plasma. The atmospheres of most stars, the gas within the glass tubing of neon advertising signs, and the gases of the upper atmosphere of Earth are examples of plasmas. A gas becomes a plasma when the kinetic e ...
... A gas that is composed of nearly equal numbers of negative and positive ions is called a plasma. The atmospheres of most stars, the gas within the glass tubing of neon advertising signs, and the gases of the upper atmosphere of Earth are examples of plasmas. A gas becomes a plasma when the kinetic e ...
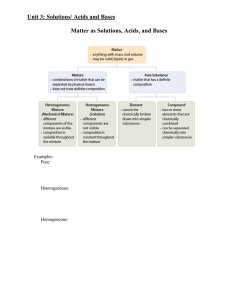
Outline for Unit 1 Solutions, Acid/Base, and Gases
... Have high concentrations of hydronium ions, making their pH closer to 1 Have a high conductivity React quicker React with active metals vigorously producing hydrogen gas. Weak Acids: Have a low concentration of hydronium ions, making their pH closer to 7 Have a low conductivity React m ...
... Have high concentrations of hydronium ions, making their pH closer to 1 Have a high conductivity React quicker React with active metals vigorously producing hydrogen gas. Weak Acids: Have a low concentration of hydronium ions, making their pH closer to 7 Have a low conductivity React m ...
Practice Writing AP Questions
... For the following reactions write a net ionic equation. Box your final answer. 1. Carbon disulfide vapor is burned in excess oxygen. a. What would be the products if the oxygen gas was not in excess? 2. Potassium permanganate solution is added to an acidic solution of hydrogen peroxide. a. This reac ...
... For the following reactions write a net ionic equation. Box your final answer. 1. Carbon disulfide vapor is burned in excess oxygen. a. What would be the products if the oxygen gas was not in excess? 2. Potassium permanganate solution is added to an acidic solution of hydrogen peroxide. a. This reac ...
Skill Sheet 19-B Chemical Formulas
... Have you ever heard of sodium nitrate? It’s a preservative used in foods like hot dogs. The chemical formula for sodium nitrate is NaNO3. How many types of atoms does this compound contain? You are right if you said three: sodium, nitrogen, and oxygen. The nitrogen and oxygen atoms have a shared-ele ...
... Have you ever heard of sodium nitrate? It’s a preservative used in foods like hot dogs. The chemical formula for sodium nitrate is NaNO3. How many types of atoms does this compound contain? You are right if you said three: sodium, nitrogen, and oxygen. The nitrogen and oxygen atoms have a shared-ele ...
A Classification of AP Chemistry Reactions
... - Chlorine gas is bubbled into a cold solution of sodium hydroxide. Cl2 + OH- Cl- + ClO- + HOH - Solutions of potassiom iodide and potassium iodate are mixed in acid solution. I- + IO3- + H+ I2 + HOH Hydrogen Hydrogen gas, H2, is an effective reducing agent for some metal oxides. - Hydrogen gas ...
... - Chlorine gas is bubbled into a cold solution of sodium hydroxide. Cl2 + OH- Cl- + ClO- + HOH - Solutions of potassiom iodide and potassium iodate are mixed in acid solution. I- + IO3- + H+ I2 + HOH Hydrogen Hydrogen gas, H2, is an effective reducing agent for some metal oxides. - Hydrogen gas ...
AP CHEMISTRY - An Incomplete List of Topics
... Types of acid/base - Bronsted-Lowry, Arrhenius, Lewis: (Remember Lewis is the opposite of Bronsted-Lowry) Know formulas of strong acids and bases Be able to calculate the Ka for either a strong or weak acid/base dissociation Know the chemical equation for either a strong or weak acid/base dissociati ...
... Types of acid/base - Bronsted-Lowry, Arrhenius, Lewis: (Remember Lewis is the opposite of Bronsted-Lowry) Know formulas of strong acids and bases Be able to calculate the Ka for either a strong or weak acid/base dissociation Know the chemical equation for either a strong or weak acid/base dissociati ...
Chapter 4 Packet
... A solution is a homogeneous mixture made by dissolving one substance (the solute) in another substance (the solvent). An aqueous solution is a solution where water is the solvent. Electrolytes are substances which produce ions in solution. An electrolytic solution is a solution which conducts an ele ...
... A solution is a homogeneous mixture made by dissolving one substance (the solute) in another substance (the solvent). An aqueous solution is a solution where water is the solvent. Electrolytes are substances which produce ions in solution. An electrolytic solution is a solution which conducts an ele ...
Question paper - Unit A173/02 - Module C7 - Higher tier (PDF
... energy needed to break all the bonds in ethanol and oxygen energy given out when all the bonds in carbon dioxide and water are made energy change when ethanol burns ...
... energy needed to break all the bonds in ethanol and oxygen energy given out when all the bonds in carbon dioxide and water are made energy change when ethanol burns ...
Word - chemmybear.com
... multiple valences like Fe° (Fe2+ & Fe3+), Cu°, or Sn° and gases like O2, F2, Cl2 the ions usually form oxidize to the “-ic” ion. Example: 2Fe°(s) + 3Cl2(g) + heat 2FeCl3(s) 2. When you identify an oxidation product, make certain you also have a reduction product. Ex: “Free halogens + dilute OH- hypo ...
... multiple valences like Fe° (Fe2+ & Fe3+), Cu°, or Sn° and gases like O2, F2, Cl2 the ions usually form oxidize to the “-ic” ion. Example: 2Fe°(s) + 3Cl2(g) + heat 2FeCl3(s) 2. When you identify an oxidation product, make certain you also have a reduction product. Ex: “Free halogens + dilute OH- hypo ...
Chemistry 20 Lesson 36 – The Whole Enchilada
... How many milliliters of 2.50 mol/L HNO3 (aq) is required to dissolve an old copper penny with a mass of 3.94 g according to the following unbalanced equation? ___ Cu(s) + ___ HNO3 (aq) → ___Cu(NO3)2 (aq) + ___ NO(g) + ___ H2O(1) ...
... How many milliliters of 2.50 mol/L HNO3 (aq) is required to dissolve an old copper penny with a mass of 3.94 g according to the following unbalanced equation? ___ Cu(s) + ___ HNO3 (aq) → ___Cu(NO3)2 (aq) + ___ NO(g) + ___ H2O(1) ...
+ H 2 O(l )
... the chemical reaction that occurs when solid sodium oxide is added to water at room temperature and forms sodium hydroxide (dissolved in the water). Include symbol for physical states and then balance. ...
... the chemical reaction that occurs when solid sodium oxide is added to water at room temperature and forms sodium hydroxide (dissolved in the water). Include symbol for physical states and then balance. ...
CHEMISTRY REVISION GUIDE for CIE IGCSE Coordinated Science
... A mole is 6.02x1023 of something. It is chosen so that a mole of something has the same mass in grams (molar mass, Mm) as its formula mass. For example the Mr of water is 18.02 so the Mm of water is 18.02g; the Mr of decane is 142.34 so the Mm of decane is 142.34g. Importantly this means that 18.02 ...
... A mole is 6.02x1023 of something. It is chosen so that a mole of something has the same mass in grams (molar mass, Mm) as its formula mass. For example the Mr of water is 18.02 so the Mm of water is 18.02g; the Mr of decane is 142.34 so the Mm of decane is 142.34g. Importantly this means that 18.02 ...
Hydroxide
Hydroxide is a diatomic anion with chemical formula OH−. It consists of an oxygen and hydrogen atom held together by a covalent bond, and carries a negative electric charge. It is an important but usually minor constituent of water. It functions as a base, a ligand, a nucleophile and a catalyst. The hydroxide ion forms salts, some of which dissociate in aqueous solution, liberating solvated hydroxide ions. Sodium hydroxide is a multi-million-ton per annum commodity chemical. A hydroxide attached to a strongly electropositive center may itself ionize, liberating a hydrogen cation (H+), making the parent compound an acid.The corresponding electrically neutral compound •HO is the hydroxyl radical. The corresponding covalently-bound group -OH of atoms is the hydroxyl group.Hydroxide ion and hydroxyl group are nucleophiles and can act as a catalyst in organic chemistry.Many inorganic substances which bear the word ""hydroxide"" in their names are not ionic compounds of the hydroxide ion, but covalent compounds which contain hydroxyl groups.