
SODIUM HYDROGEN CARBONATE
... Wyoming. It is cheaper to mine Na2CO3 than to manufacture it. Furthermore, the CaCl2 byproduct of the Solvay process has not found sufficient use as a chemical to enhance the economic value of the Solvay process. About 11 million tons of Na2CO3 are produced annually. In 1995 the total production of ...
... Wyoming. It is cheaper to mine Na2CO3 than to manufacture it. Furthermore, the CaCl2 byproduct of the Solvay process has not found sufficient use as a chemical to enhance the economic value of the Solvay process. About 11 million tons of Na2CO3 are produced annually. In 1995 the total production of ...
Chapter1 - WilsonChemWiki
... Ions: are atoms or group of atoms that have lost or gained electrons, and are classified according to their charges to: Positive ions (Cations): are atoms that have lost electron(s). Like; Li loses 1e- and become Li+ ion, Ca loses 2e- and become Ca+2 ion, Al loses 3e- and become Al+3 ion,… Negative ...
... Ions: are atoms or group of atoms that have lost or gained electrons, and are classified according to their charges to: Positive ions (Cations): are atoms that have lost electron(s). Like; Li loses 1e- and become Li+ ion, Ca loses 2e- and become Ca+2 ion, Al loses 3e- and become Al+3 ion,… Negative ...
study material class X (science)
... Ans. (a) marble chips react with dilute hydrochloric acid to form calcium chloride and carbon dioxide .it is a double displacement reaction CaCO3+2HCl CaCl2 + H2O +CO2 (b) Zinc granules react with dilute hydrochloric acid to give hydrogen gas. it is a displacement reaction Zn(s)+2HCl ZnCl2(aq)+H ...
... Ans. (a) marble chips react with dilute hydrochloric acid to form calcium chloride and carbon dioxide .it is a double displacement reaction CaCO3+2HCl CaCl2 + H2O +CO2 (b) Zinc granules react with dilute hydrochloric acid to give hydrogen gas. it is a displacement reaction Zn(s)+2HCl ZnCl2(aq)+H ...
CHEM 120 WEEK 11 LECTURES (INORGANIC WEEK 2) Dr. MD
... Contains only metals, apart from boron. Boron is also the only element which does not form a stable trication (B3+) again will have too high a charge density to be stable. Why do the other elements form tri-cations (M3+ )? Soln. √ Because they have the valence electronic configuration ns2np1 and ...
... Contains only metals, apart from boron. Boron is also the only element which does not form a stable trication (B3+) again will have too high a charge density to be stable. Why do the other elements form tri-cations (M3+ )? Soln. √ Because they have the valence electronic configuration ns2np1 and ...
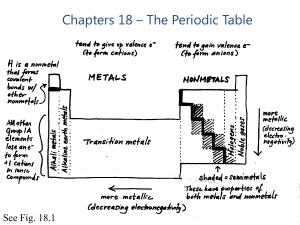
Chapters 18 – The Periodic Table
... from SO2 over V2O5 catalysts, is then converted to sulfuric acid. Sulfuric acid is the cheapest strong acid and is so widely used in industry that its production level is an indicator of a nation’s economic strength. Strong dehydrating agent that removes water from any organic source. 4. Sulfur hexa ...
... from SO2 over V2O5 catalysts, is then converted to sulfuric acid. Sulfuric acid is the cheapest strong acid and is so widely used in industry that its production level is an indicator of a nation’s economic strength. Strong dehydrating agent that removes water from any organic source. 4. Sulfur hexa ...
CHEMISTRY 101 Name Mock Final Exam Spring 2014 Signature Dr
... cobalt(II) fluoride cobalt(II) sulfate cobalt(III) fluoride cobalt(II) chloride ...
... cobalt(II) fluoride cobalt(II) sulfate cobalt(III) fluoride cobalt(II) chloride ...
153KB PDF - Clydeview Academy
... 4 If any of this information is wrong, tell the Invigilator immediately. 5 If this information is correct, print your name and seat number in the boxes provided. 6 The answer to each question is either A, B, C or D. Decide what your answer is, then, using your pencil, put a horizontal line in the sp ...
... 4 If any of this information is wrong, tell the Invigilator immediately. 5 If this information is correct, print your name and seat number in the boxes provided. 6 The answer to each question is either A, B, C or D. Decide what your answer is, then, using your pencil, put a horizontal line in the sp ...
Name - Piscataway High School
... Temperature and its measurement Temperature (which means the average kinetic energy of the molecule) can be measured using three common scales: Celsius, Kelvin, and Fahrenheit. We use the following formulas to convert form one scale to another. Celsius is the scale most desirable for laboratory wor ...
... Temperature and its measurement Temperature (which means the average kinetic energy of the molecule) can be measured using three common scales: Celsius, Kelvin, and Fahrenheit. We use the following formulas to convert form one scale to another. Celsius is the scale most desirable for laboratory wor ...
Chapter 8
... A positive ion forms when an atom loses one or more electrons. When the atom loses an electron, it does not become a different element because it still has the same number of protons. A positively charged ion is called a cation. Elements on the left side of the periodic table tend to lose some of th ...
... A positive ion forms when an atom loses one or more electrons. When the atom loses an electron, it does not become a different element because it still has the same number of protons. A positively charged ion is called a cation. Elements on the left side of the periodic table tend to lose some of th ...
Bonding 1. Which one of the following is most likely to be an ionic
... 2. Which of the following pairs of elements would be most likely to form an ionic compound? a. P and Br b. Zn and K c. C and O d. Al and Rb e. F and Ca 3. What is the name of NaI? a. sodium iodide b. sodium(I) iodide c. sodium monoiodide d. sodious iodide e. sodium iodine 4. Which of the following c ...
... 2. Which of the following pairs of elements would be most likely to form an ionic compound? a. P and Br b. Zn and K c. C and O d. Al and Rb e. F and Ca 3. What is the name of NaI? a. sodium iodide b. sodium(I) iodide c. sodium monoiodide d. sodious iodide e. sodium iodine 4. Which of the following c ...
chemistry advanced may 2010 marking scheme
... (iii) This is because the chloride ion is too weak a base to be effective as a buffer against addition of hydroxonium ion. (The H3O+ ion in the HCl-NaCl mixture will however buffer against the addition of base.) (1 mark) accept other correct answers. 2. Nitric(V) acid is an important industrial chem ...
... (iii) This is because the chloride ion is too weak a base to be effective as a buffer against addition of hydroxonium ion. (The H3O+ ion in the HCl-NaCl mixture will however buffer against the addition of base.) (1 mark) accept other correct answers. 2. Nitric(V) acid is an important industrial chem ...
Solution - gearju.com
... The charge the atom would have in a molecule (or an ionic compound) if electrons were completely transferred. 1. Free elements (uncombined state) have an oxidation number of zero. ...
... The charge the atom would have in a molecule (or an ionic compound) if electrons were completely transferred. 1. Free elements (uncombined state) have an oxidation number of zero. ...
cbse class – x science solutions
... Many metals such as copper, zinc etc. are refined electrolytic ally. In this process, the impure copper metal is made anode and a thin strip of pure copper metal is made the cathode. A solution of copper sulphate is taken as electrolyte. The apparatus is set up as shown in the figure. On passing the ...
... Many metals such as copper, zinc etc. are refined electrolytic ally. In this process, the impure copper metal is made anode and a thin strip of pure copper metal is made the cathode. A solution of copper sulphate is taken as electrolyte. The apparatus is set up as shown in the figure. On passing the ...
2007 - Thompson Rivers University
... 17. The following table lists the names and corresponding formulae of a number of compounds. Some lines in the table contain errors. i ii iii iv v vi ...
... 17. The following table lists the names and corresponding formulae of a number of compounds. Some lines in the table contain errors. i ii iii iv v vi ...
Chemistry Final Exam Review 2006-2007
... 2. On the pathway below, label the activated complex, activation energy with catalyst, and activation energy without catalyst 3 What are the five factors that affect the rate of a reaction? 4. Which of the five factors change collision frequency? 5 Which factor changes collision frequency and the e ...
... 2. On the pathway below, label the activated complex, activation energy with catalyst, and activation energy without catalyst 3 What are the five factors that affect the rate of a reaction? 4. Which of the five factors change collision frequency? 5 Which factor changes collision frequency and the e ...
1 - Study Hungary
... 20 protons, 19 neutrons, 19 electrons 19 protons, 20 neutrons, 18 electrons 39 protons, 19 neutrons, 38 electrons 20 protons, 19 neutrons, 20 electrons 40 protons, 20 neutrons, 19 electrons ...
... 20 protons, 19 neutrons, 19 electrons 19 protons, 20 neutrons, 18 electrons 39 protons, 19 neutrons, 38 electrons 20 protons, 19 neutrons, 20 electrons 40 protons, 20 neutrons, 19 electrons ...
Ch 9 Pkt - mvhs
... 1. Identify the limiting reactant when 1.22 g of O2 reacts with 1.05 g of H2 to produce water. 2. Identify the limiting reactant when 4.68 g of Fe reacts with 2.99 g of S to produce FeS. 3. Identify the limiting reactant when 5.87 g of Mg(OH)2 reacts with 12.84 g of HCl to form MgCl2 and water. 4. I ...
... 1. Identify the limiting reactant when 1.22 g of O2 reacts with 1.05 g of H2 to produce water. 2. Identify the limiting reactant when 4.68 g of Fe reacts with 2.99 g of S to produce FeS. 3. Identify the limiting reactant when 5.87 g of Mg(OH)2 reacts with 12.84 g of HCl to form MgCl2 and water. 4. I ...
Hydroxide
Hydroxide is a diatomic anion with chemical formula OH−. It consists of an oxygen and hydrogen atom held together by a covalent bond, and carries a negative electric charge. It is an important but usually minor constituent of water. It functions as a base, a ligand, a nucleophile and a catalyst. The hydroxide ion forms salts, some of which dissociate in aqueous solution, liberating solvated hydroxide ions. Sodium hydroxide is a multi-million-ton per annum commodity chemical. A hydroxide attached to a strongly electropositive center may itself ionize, liberating a hydrogen cation (H+), making the parent compound an acid.The corresponding electrically neutral compound •HO is the hydroxyl radical. The corresponding covalently-bound group -OH of atoms is the hydroxyl group.Hydroxide ion and hydroxyl group are nucleophiles and can act as a catalyst in organic chemistry.Many inorganic substances which bear the word ""hydroxide"" in their names are not ionic compounds of the hydroxide ion, but covalent compounds which contain hydroxyl groups.



![C2_revision_slides_V3_+_questions_+_MS_-_H[1]](http://s1.studyres.com/store/data/000092833_1-97fb33725e7f1ef12029ed42751d3dca-300x300.png)


















