equilibrium - eVirtualGuru
... H2O (l) ⇌ H2O (vap) The double half arrows indicate that the processes in both the directions are going on simultaneously. The mixture of reactants and products in the equilibrium state is called an equilibrium mixture. Equilibrium can be established for both physical processes and chemical reaction ...
... H2O (l) ⇌ H2O (vap) The double half arrows indicate that the processes in both the directions are going on simultaneously. The mixture of reactants and products in the equilibrium state is called an equilibrium mixture. Equilibrium can be established for both physical processes and chemical reaction ...
Chemistry HL Syllabus Details
... TOK: The early discoverers of the elements allowed chemistry to make great steps with limited apparatus, often derived from the pseudoscience of alchemy. Lavoisier’s work with oxygen, which overturned the phlogiston theory of heat, could be discussed as an example of a paradigm shift. Int: The disco ...
... TOK: The early discoverers of the elements allowed chemistry to make great steps with limited apparatus, often derived from the pseudoscience of alchemy. Lavoisier’s work with oxygen, which overturned the phlogiston theory of heat, could be discussed as an example of a paradigm shift. Int: The disco ...
Section – B - About iTutoring
... The catalyst reduces time required to complete the reaction but does not affect equilibrium state. (30). In the reaction A2 + B2 2AB + Q cal., what should be decreased to increase the proportion of AB? On decreasing temperature, the rate of endothermic i.e., forward reaction increases. (31). Mention ...
... The catalyst reduces time required to complete the reaction but does not affect equilibrium state. (30). In the reaction A2 + B2 2AB + Q cal., what should be decreased to increase the proportion of AB? On decreasing temperature, the rate of endothermic i.e., forward reaction increases. (31). Mention ...
IIT-JEE (Advanced) - Brilliant Public School Sitamarhi
... Silver salt method : (for organic acids) Basicity of an acid : No. of replacable H+ atoms in an acid (H contained to more electronegative atom is acidic) Procedure : Some known amount of silver salt (w1 gm) is heated to obtain w2 gm of while shining residue of silver. Then if the basicity of acid is ...
... Silver salt method : (for organic acids) Basicity of an acid : No. of replacable H+ atoms in an acid (H contained to more electronegative atom is acidic) Procedure : Some known amount of silver salt (w1 gm) is heated to obtain w2 gm of while shining residue of silver. Then if the basicity of acid is ...
Amines - ncert
... present at different positions in the parent chain, their positions are specified by giving numbers to the carbon atoms bearing –NH2 groups and suitable prefix such as di, tri, etc. is attached to the amine. The letter ‘e’ of the suffix of the hydrocarbon part is retained. For example, H2N–CH2–CH2–N ...
... present at different positions in the parent chain, their positions are specified by giving numbers to the carbon atoms bearing –NH2 groups and suitable prefix such as di, tri, etc. is attached to the amine. The letter ‘e’ of the suffix of the hydrocarbon part is retained. For example, H2N–CH2–CH2–N ...
- Kendriya Vidyalaya Jamuna Colliery
... 5. In terms of band theory what is the difference between a conductor, an insulator and a semiconductor? The energy gap between the valence band and conduction band in an insulator is very large while in a conductor, the energy gap is very small or there is overlapping between valence band and condu ...
... 5. In terms of band theory what is the difference between a conductor, an insulator and a semiconductor? The energy gap between the valence band and conduction band in an insulator is very large while in a conductor, the energy gap is very small or there is overlapping between valence band and condu ...
chemistry - Textbooks Online
... (i) The magnitude of order of a reaction may be zero, or fractional or integral values. For an elementary reaction, its order is never fractional since it is a one step process. (ii) Order of a reaction should be determined only by experiments. It cannot be predicted interms of stoichiometry of reac ...
... (i) The magnitude of order of a reaction may be zero, or fractional or integral values. For an elementary reaction, its order is never fractional since it is a one step process. (ii) Order of a reaction should be determined only by experiments. It cannot be predicted interms of stoichiometry of reac ...
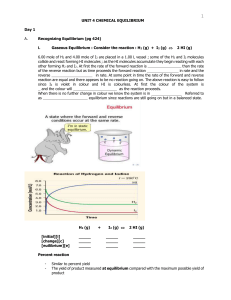
Unit 4 - Chemical Equilibrium
... Recognizing Equilibrium (pg 424) Gaseous Equilibrium : Consider the reaction : H2 (g) + I2 (g) ...
... Recognizing Equilibrium (pg 424) Gaseous Equilibrium : Consider the reaction : H2 (g) + I2 (g) ...
Problem 1-2
... of air a ternary, white, crystalline compound B and a gas C form. In the presence of air gas C burns with a light blue flame. The elementary analysis of B shows 24.5 % (w/w) of carbon and 28.6 % (w/w) of nitrogen. When annealed with carbon another ionic compound D also results in compound B, too, bu ...
... of air a ternary, white, crystalline compound B and a gas C form. In the presence of air gas C burns with a light blue flame. The elementary analysis of B shows 24.5 % (w/w) of carbon and 28.6 % (w/w) of nitrogen. When annealed with carbon another ionic compound D also results in compound B, too, bu ...
Acid rain
Acid rain is a rain or any other form of precipitation that is unusually acidic, meaning that it possesses elevated levels of hydrogen ions (low pH). It can have harmful effects on plants, aquatic animals and infrastructure. Acid rain is caused by emissions of sulfur dioxide and nitrogen oxide, which react with the water molecules in the atmosphere to produce acids. Governments have made efforts since the 1970s to reduce the release of sulfur dioxide into the atmosphere with positive results. Nitrogen oxides can also be produced naturally by lightning strikes and sulfur dioxide is produced by volcanic eruptions. The chemicals in acid rain can cause paint to peel, corrosion of steel structures such as bridges, and erosion of stone statues.