Chemical Vapor Deposition (CVD)
... • This technique is suitable for the manufacture of coatings, powders, fibers and monolithic components. • This technique is often used in many thin film applications. • By varying the experimental conditions—substrate material, substrate temperature, composition of the reaction gas mixture, total p ...
... • This technique is suitable for the manufacture of coatings, powders, fibers and monolithic components. • This technique is often used in many thin film applications. • By varying the experimental conditions—substrate material, substrate temperature, composition of the reaction gas mixture, total p ...
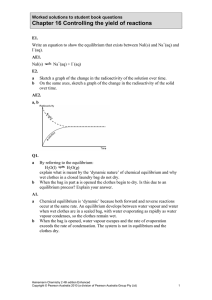
Chapter 16 Controlling the yield of reactions
... The volume of the container was increased at constant temperature and a new equilbrium was established. Predict how each of the following quantities would change at the new equilibrium compared with the initial equilibrium: a concentration of NO2 b mass of NO2 A12. ...
... The volume of the container was increased at constant temperature and a new equilbrium was established. Predict how each of the following quantities would change at the new equilibrium compared with the initial equilibrium: a concentration of NO2 b mass of NO2 A12. ...
Ch16 - WordPress.com
... The volume of the container was increased at constant temperature and a new equilbrium was established. Predict how each of the following quantities would change at the new equilibrium compared with the initial equilibrium: a concentration of NO2 b mass of NO2 A12. An increase in volume will cause a ...
... The volume of the container was increased at constant temperature and a new equilbrium was established. Predict how each of the following quantities would change at the new equilibrium compared with the initial equilibrium: a concentration of NO2 b mass of NO2 A12. An increase in volume will cause a ...
Ch16
... The volume of the container was increased at constant temperature and a new equilbrium was established. Predict how each of the following quantities would change at the new equilibrium compared with the initial equilibrium: a concentration of NO2 b mass of NO2 A12. An increase in volume will cause a ...
... The volume of the container was increased at constant temperature and a new equilbrium was established. Predict how each of the following quantities would change at the new equilibrium compared with the initial equilibrium: a concentration of NO2 b mass of NO2 A12. An increase in volume will cause a ...
On the formation of radiation fogs under heavily polluted
... curves very closely as they grow. With certain types of atmospheric clouds this would not be the case as the droplets are actually out of equilibrium because of kinetic limitations in water vapor condensation (Nenes et al., 2001). Considering gases such as HNO3 , with mixing ratios orders of magnitu ...
... curves very closely as they grow. With certain types of atmospheric clouds this would not be the case as the droplets are actually out of equilibrium because of kinetic limitations in water vapor condensation (Nenes et al., 2001). Considering gases such as HNO3 , with mixing ratios orders of magnitu ...
Solubility of Diflunisal in Supercritical Carbon Dioxide
... Experimental Section, each reported data point is the average of at least three replicate experimental measurements. The relative standard deviation (RSD) values were taken as an indication of replicate measurement reproducibility and varied between 15.1 % (T ) 328.2 K, P ) 9.1 MPa) and 2 % (T ) 328 ...
... Experimental Section, each reported data point is the average of at least three replicate experimental measurements. The relative standard deviation (RSD) values were taken as an indication of replicate measurement reproducibility and varied between 15.1 % (T ) 328.2 K, P ) 9.1 MPa) and 2 % (T ) 328 ...
The Uptake of Methyl Vinyl Ketone
... solutions were used as carrier gas at a total flow rate of 100260 STP cm3 min-1 (STP conditions: 273 K and 1 atm), a total pressure P ) 65-170 Torr (0.08-0.23 atm), and temperatures of 273-313 K. The organic compounds were introduced into the main flow of the reactor by entraining their vapor into a ...
... solutions were used as carrier gas at a total flow rate of 100260 STP cm3 min-1 (STP conditions: 273 K and 1 atm), a total pressure P ) 65-170 Torr (0.08-0.23 atm), and temperatures of 273-313 K. The organic compounds were introduced into the main flow of the reactor by entraining their vapor into a ...
IGNITION OF CNG MIXTURES IN SPARK IGNITION ENGINES
... and is almost 1.5 times bigger than in diesel engines with direct fuel injection. For these reasons the maximum value of combustion pressure can reach 180 bars. The ignition problem of CNG mixtures was considered by many researches in the past [1, 5, 7]. It corresponds also to the theory of ignition ...
... and is almost 1.5 times bigger than in diesel engines with direct fuel injection. For these reasons the maximum value of combustion pressure can reach 180 bars. The ignition problem of CNG mixtures was considered by many researches in the past [1, 5, 7]. It corresponds also to the theory of ignition ...
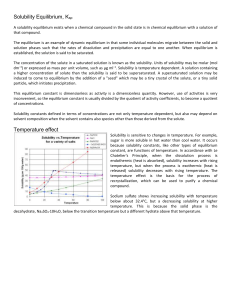
Solutions - WordPress.com
... • The most common solvent is Water – the universal solvent • The term aqueous or (aq) describes a solution in which the solvent is water • Not all solutes and solvents are liquid! ▫ Brass (zinc and nickel) ▫ Seawater (salt and other substances in water) ▫ Air (Many different gasses in Nitrogen) ...
... • The most common solvent is Water – the universal solvent • The term aqueous or (aq) describes a solution in which the solvent is water • Not all solutes and solvents are liquid! ▫ Brass (zinc and nickel) ▫ Seawater (salt and other substances in water) ▫ Air (Many different gasses in Nitrogen) ...
fast pyrolysis characteristics of sugarcane bagasse hemicellulose
... product distribution The distributions of products resulted from the pyrolysis of sugarcane bagasse hemicellulose at different temperatures in the tubular furnace are shown in Figure 2. As seen, the product distributions of hemicellulose changed with increasing temperature. The gas yield was the hig ...
... product distribution The distributions of products resulted from the pyrolysis of sugarcane bagasse hemicellulose at different temperatures in the tubular furnace are shown in Figure 2. As seen, the product distributions of hemicellulose changed with increasing temperature. The gas yield was the hig ...
Thermal Diffusion and Partial Molar Enthalpy Variations of n
... and ngb is the number of moles of butane in the gas. We verified that the ideal gas assumption was a good approximation by calculating the compressibility factor of the gas phase, which was equal to unity with an accuracy of 1% for the range of simulated pressures and temperatures. For simulations a ...
... and ngb is the number of moles of butane in the gas. We verified that the ideal gas assumption was a good approximation by calculating the compressibility factor of the gas phase, which was equal to unity with an accuracy of 1% for the range of simulated pressures and temperatures. For simulations a ...
CHEM 1412. Chapter 15. Chemical Equilibrium (Homework)
... 19. For the reaction PCl3(g) + Cl2(g) PCl5(g) at a particular temperature, Kc = 24.3. Suppose a system at that temperature is prepared with [PCl3] = 0.10 M, [Cl2] = 0.15 M, and [PCl5] = 0.60 M. Which of the following is true based on the above? A. Qc > Kc, the reaction proceeds from left to right to ...
... 19. For the reaction PCl3(g) + Cl2(g) PCl5(g) at a particular temperature, Kc = 24.3. Suppose a system at that temperature is prepared with [PCl3] = 0.10 M, [Cl2] = 0.15 M, and [PCl5] = 0.60 M. Which of the following is true based on the above? A. Qc > Kc, the reaction proceeds from left to right to ...
co2 removal from natural gas by hydrate formation
... are 19.3 bar and 60.4 mol% CO2, respectively. The temperature of the system is then raised to its initial value (20.0 °C) to dissociate the hydrates formed. In this heating process, two endothermic perturbations are observed in the temperature profile corresponding to the decomposition of the hydrat ...
... are 19.3 bar and 60.4 mol% CO2, respectively. The temperature of the system is then raised to its initial value (20.0 °C) to dissociate the hydrates formed. In this heating process, two endothermic perturbations are observed in the temperature profile corresponding to the decomposition of the hydrat ...
Lab 1: Measurement
... record the number of significant figures and units in each measurement. Mass measurement involves determining the mass of the sand in a vial. Three stations will be set up in the laboratory. Each station is set up for you to determine the two volume measurements and a temperature measurement. The me ...
... record the number of significant figures and units in each measurement. Mass measurement involves determining the mass of the sand in a vial. Three stations will be set up in the laboratory. Each station is set up for you to determine the two volume measurements and a temperature measurement. The me ...
document
... C3H8(g) + 5O2(g) → 3CO2(g) + 4H2O(g) Calculate the number of moles of oxygen required to react exactly with 4.3 moles of propane, C3H8, in the above reaction 4.3 moles of C3H8 requires how many moles of O2 There is a 1:5 ratio So 4.3(1) : 4.3(5) ...
... C3H8(g) + 5O2(g) → 3CO2(g) + 4H2O(g) Calculate the number of moles of oxygen required to react exactly with 4.3 moles of propane, C3H8, in the above reaction 4.3 moles of C3H8 requires how many moles of O2 There is a 1:5 ratio So 4.3(1) : 4.3(5) ...
Quiz Keys - Section 10
... Problem 1 (7 points). In class we discussed the problems associated with high altitude caused by lower boiling points of liquids at lower pressures. A different kind of problem may be caused by changing a boiling point of a liquid at altitudes below sea level. Northern Europe (especially Sweden and ...
... Problem 1 (7 points). In class we discussed the problems associated with high altitude caused by lower boiling points of liquids at lower pressures. A different kind of problem may be caused by changing a boiling point of a liquid at altitudes below sea level. Northern Europe (especially Sweden and ...
The speciation equilibrium coupling with phase equilibrium in the
... Fig. 1. The dissociation constant, pK1, as a function of temperature in pure water (the model of this study vs. experimental data at pressures close to water saturation pressures, where pK1 = −log10(K1). ...
... Fig. 1. The dissociation constant, pK1, as a function of temperature in pure water (the model of this study vs. experimental data at pressures close to water saturation pressures, where pK1 = −log10(K1). ...