basic thermodynamics
... If the water is cooled at a constant pressure of 1 atm from 75 oCto 5oC the heat given up may be used as a source for a series of Carnot engines each using the surroundings as a sink. It is assumed that the amount of energy received by any engine is small relative to that in the source and the tempe ...
... If the water is cooled at a constant pressure of 1 atm from 75 oCto 5oC the heat given up may be used as a source for a series of Carnot engines each using the surroundings as a sink. It is assumed that the amount of energy received by any engine is small relative to that in the source and the tempe ...
Chemistry: Matter and Change
... • Distillation is a separation technique for homogeneous mixtures that is based on the differences in boiling points of substances. • Crystallization is a separation technique for homogenous mixtures that results in the formation of pure solid particles from a solution containing the dissolved subst ...
... • Distillation is a separation technique for homogeneous mixtures that is based on the differences in boiling points of substances. • Crystallization is a separation technique for homogenous mixtures that results in the formation of pure solid particles from a solution containing the dissolved subst ...
dx cx dx and x - Cameron University
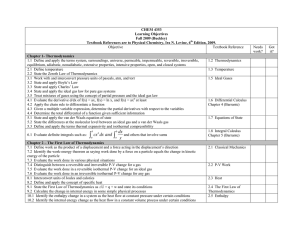
... 12.1 Apply heat, work, internal energy and enthalpy concepts to the reversible isothermal P-V change in a perfect gas 12.2 Apply heat, work, internal energy and enthalpy concepts to the reversible constant-P (or constant-V) process in a perfect gas 12.3 Apply heat, work, internal energy and enthalpy ...
... 12.1 Apply heat, work, internal energy and enthalpy concepts to the reversible isothermal P-V change in a perfect gas 12.2 Apply heat, work, internal energy and enthalpy concepts to the reversible constant-P (or constant-V) process in a perfect gas 12.3 Apply heat, work, internal energy and enthalpy ...
Assignment CHE-04 TMA-01,02 Year 2005
... A weightage of 30 percent, as you are aware, has been earmarked for continuous evaluation. This would consist of two tutor-marked assignments (TMA-1 and TMA-2) for CHE - 04 Course. You can find both these assignments in this booklet. TMA-1 is based on Blocks 1 and 2 and, TMA-2 is based on Blocks 3, ...
... A weightage of 30 percent, as you are aware, has been earmarked for continuous evaluation. This would consist of two tutor-marked assignments (TMA-1 and TMA-2) for CHE - 04 Course. You can find both these assignments in this booklet. TMA-1 is based on Blocks 1 and 2 and, TMA-2 is based on Blocks 3, ...
Multiple Choice Math Practice File
... (raw score) (Raw score) x 0.95 = _________________ (weighted MC score) The following statement is at the top of the multiple choice section…please be familiar with the symbols used on the test. Note: For all questions, assume that the temperature is 298 K, the pressure is 1.00 atmospheres and soluti ...
... (raw score) (Raw score) x 0.95 = _________________ (weighted MC score) The following statement is at the top of the multiple choice section…please be familiar with the symbols used on the test. Note: For all questions, assume that the temperature is 298 K, the pressure is 1.00 atmospheres and soluti ...
Ch 17 Equilibrium Notes
... n = sum of coefficients of gaseous products minus sum of coefficients of gaseous reactants. Calculate Kp for the following reaction: PCl3(g) + Cl2(g) ↔ PCl3(g) where Kc= 1.67(500 K) Do Follow up Problem 17.5 Pg.735. Solving Equilibrium Problems 1. Balance the equation. 2.Write the equilibrium expre ...
... n = sum of coefficients of gaseous products minus sum of coefficients of gaseous reactants. Calculate Kp for the following reaction: PCl3(g) + Cl2(g) ↔ PCl3(g) where Kc= 1.67(500 K) Do Follow up Problem 17.5 Pg.735. Solving Equilibrium Problems 1. Balance the equation. 2.Write the equilibrium expre ...
Stag3D: A code for modeling thermo
... The finite-volume method MPDATA by Smolarkiewicz [6] is used to advect the temperature field, while finite differences are used for the other terms in Eq 3. For the nondiffusive C field (Eq. 4), particles are used, as in Tackley and King [7] and Tackley [8]. 3.2 Melting and differentiation Partial m ...
... The finite-volume method MPDATA by Smolarkiewicz [6] is used to advect the temperature field, while finite differences are used for the other terms in Eq 3. For the nondiffusive C field (Eq. 4), particles are used, as in Tackley and King [7] and Tackley [8]. 3.2 Melting and differentiation Partial m ...
Units of Measurement
... – the ratio by mass of the elements in a chemical compound is always the same, regardless of the source of the compound. – The law of constant composition can be used to distinguish between compounds and mixtures of elements: – Compounds have a constant composition; mixtures do not. – Water is alway ...
... – the ratio by mass of the elements in a chemical compound is always the same, regardless of the source of the compound. – The law of constant composition can be used to distinguish between compounds and mixtures of elements: – Compounds have a constant composition; mixtures do not. – Water is alway ...
Balanced Equations And Equilibrium Constants
... that when solving for equilibrium constants, the activities of pure solids and liquids are one, so (NH4)2CO3(s) is not included in the equation. Kc = [NH3]2[CO2][H2O] It is more appropriate to use Kp because all the constituents in the equation are gases: Kp = (PNH3)2(PCO2)(PH2O) Side note: Remember ...
... that when solving for equilibrium constants, the activities of pure solids and liquids are one, so (NH4)2CO3(s) is not included in the equation. Kc = [NH3]2[CO2][H2O] It is more appropriate to use Kp because all the constituents in the equation are gases: Kp = (PNH3)2(PCO2)(PH2O) Side note: Remember ...
Chapter 5 Diffusion Availability
... The availability is the maximum work that can be extracted from the substance as it undergoes a reversible process from a given state to the environmental state. When a substance is in the environmental state, it is in thermal and mechanical equilibrium with the environment and therefore has no work ...
... The availability is the maximum work that can be extracted from the substance as it undergoes a reversible process from a given state to the environmental state. When a substance is in the environmental state, it is in thermal and mechanical equilibrium with the environment and therefore has no work ...
A Pool Boiling Map: Water on a Horizontal Surface at
... D the number of vapor columns has increased to the point where they now restrict the flow of fluid to the surface. Partial film boiling or transition boiling, region D to E, occurs after departure from nucleate boiling has been reached. The vapor columns and vapor film are unstable and collapse and ...
... D the number of vapor columns has increased to the point where they now restrict the flow of fluid to the surface. Partial film boiling or transition boiling, region D to E, occurs after departure from nucleate boiling has been reached. The vapor columns and vapor film are unstable and collapse and ...
Computational thermodynamics - IS MU
... equations, global minimization of Gibbs energy, driving force for a phase. 3. Phase diagrams: definition and types, mapping a phase diagram, implicitly defined functions and their derivatives. Optimization methods: the principle of the leastsquares method, the weighting factor. Marquardt’s algorithm ...
... equations, global minimization of Gibbs energy, driving force for a phase. 3. Phase diagrams: definition and types, mapping a phase diagram, implicitly defined functions and their derivatives. Optimization methods: the principle of the leastsquares method, the weighting factor. Marquardt’s algorithm ...
2.4 Chemical equilibria
... 2. Will heating the mixture give an equilibrium less mixture with more or less ammonia? 3. Are there more gas molecules of reactant or ...
... 2. Will heating the mixture give an equilibrium less mixture with more or less ammonia? 3. Are there more gas molecules of reactant or ...
Chapter 17 - Bakersfield College
... Product (coefficient of 2 becomes power of 2) Reactants (coefficients of 1 & 3 become powers of 1 & 3) ...
... Product (coefficient of 2 becomes power of 2) Reactants (coefficients of 1 & 3 become powers of 1 & 3) ...
Review - Discount Flies
... Know the relationship between pressure and volume plus temperature and volume. Combined Gas Law: deals with changing conditions. P1V 1 = P2V2 T1 T2 Calculate the volume of 2 liters of gas at 100 oC and 3 atm if it is changed to STP. (3 Atm) (2 L) (373 K) ...
... Know the relationship between pressure and volume plus temperature and volume. Combined Gas Law: deals with changing conditions. P1V 1 = P2V2 T1 T2 Calculate the volume of 2 liters of gas at 100 oC and 3 atm if it is changed to STP. (3 Atm) (2 L) (373 K) ...