Exam Review_Key_All Topics.082
... Solid - Molecules are held close to each other by their attractions of charge. They will bend and/or vibrate, but will stay in close proximity. Liquid - Molecules will flow or glide over one another, but stay toward the bottom of the container. Motion is a bit more random than that of a solid. ...
... Solid - Molecules are held close to each other by their attractions of charge. They will bend and/or vibrate, but will stay in close proximity. Liquid - Molecules will flow or glide over one another, but stay toward the bottom of the container. Motion is a bit more random than that of a solid. ...
Solutions
... case the amount dissolved will be _________the line of solubility. Solution____________. When a solution contains more than the maximum amount of solute that will dissolve at a specific temperature it is____________. In this case the amount dissolved will be ________the line of solubility. Made from ...
... case the amount dissolved will be _________the line of solubility. Solution____________. When a solution contains more than the maximum amount of solute that will dissolve at a specific temperature it is____________. In this case the amount dissolved will be ________the line of solubility. Made from ...
AP Chem Test 5 preview Gases
... the partial pressure of Ar in the flask is 2.00 atm. The temperature is 127 oC. a.) Calculate the total pressure in the flask. b.) Calculate the mole fraction of H2 in the flask. c.) Calculate the density (in g L-1) of the mixture in the flask. The mixture in the flask is ignited by a spark, and the ...
... the partial pressure of Ar in the flask is 2.00 atm. The temperature is 127 oC. a.) Calculate the total pressure in the flask. b.) Calculate the mole fraction of H2 in the flask. c.) Calculate the density (in g L-1) of the mixture in the flask. The mixture in the flask is ignited by a spark, and the ...
More Than You Ever Cared to Know About Solution Thermodynamics
... The one dimensional hard line model, originally introduced by Rayleigh [1] and extended to hard line mixtures [2], was used to explicitly discuss solution thermodynamics [3]. Unfortunately, the hard line mixture (suitably extended to three dimensions) is an ideal solution, and as a result, part of t ...
... The one dimensional hard line model, originally introduced by Rayleigh [1] and extended to hard line mixtures [2], was used to explicitly discuss solution thermodynamics [3]. Unfortunately, the hard line mixture (suitably extended to three dimensions) is an ideal solution, and as a result, part of t ...
Latent Heat of Vaporization and Speci c Heat - Physlab
... how is this energy shared? James Clerk Maxwell solved this problem for a large number of molecules. He said that energy is equally divided in all the directions a molecule is free to move. The average energy, when the number of molecules is large, per molecule is 12 kB T for each independent degree ...
... how is this energy shared? James Clerk Maxwell solved this problem for a large number of molecules. He said that energy is equally divided in all the directions a molecule is free to move. The average energy, when the number of molecules is large, per molecule is 12 kB T for each independent degree ...
Avogadro`s Law is relation between
... 7- Calculate the value of [–OH] from the given [H3O+] and label the solution as acidic or basic. a. 10–8 M c. 3.0 × 10–4 M b. 10–10 M d. 2.5 × 10–11 M 8- Calculate the value of [–OH] from the given [H3O+] and label the solution as acidic or basic. a. 10–1 M c. 2.6 × 10–7 M b. 10–13 M d. 1.2 × 10–12 ...
... 7- Calculate the value of [–OH] from the given [H3O+] and label the solution as acidic or basic. a. 10–8 M c. 3.0 × 10–4 M b. 10–10 M d. 2.5 × 10–11 M 8- Calculate the value of [–OH] from the given [H3O+] and label the solution as acidic or basic. a. 10–1 M c. 2.6 × 10–7 M b. 10–13 M d. 1.2 × 10–12 ...
1 mole = 6.02 X 10 23 Particles
... One mole of $1 bills stacked one on top of another would reach from the Sun to Pluto and back 7.5 million times. It would take light 9500 years to travel from the bottom to the top of a stack of 1 mole of $1 bills. ...
... One mole of $1 bills stacked one on top of another would reach from the Sun to Pluto and back 7.5 million times. It would take light 9500 years to travel from the bottom to the top of a stack of 1 mole of $1 bills. ...
Spring 2002 - Kwantlen Polytechnic University
... a. The solution obey’s Raoult’s Law. b. The solution shows a positive deviation from Raoult’s Law. c. The solution shows a negative deviation from Raoult’s Law and possesses a minimum boiling point azeotrope. d. The solution shows a negative deviation from Raoult’s Law and possesses a maximum boilin ...
... a. The solution obey’s Raoult’s Law. b. The solution shows a positive deviation from Raoult’s Law. c. The solution shows a negative deviation from Raoult’s Law and possesses a minimum boiling point azeotrope. d. The solution shows a negative deviation from Raoult’s Law and possesses a maximum boilin ...
2013 - NESACS
... Use the following information for questions 36-38. A student wanted to find out how much stomach acid would be neutralized by one TUMS antacid tablet whose active ingredient is CaCO3 (100.8 g/mole). After weighing the tablets in one bottle, the student found that the average mass of TUMS tablet was ...
... Use the following information for questions 36-38. A student wanted to find out how much stomach acid would be neutralized by one TUMS antacid tablet whose active ingredient is CaCO3 (100.8 g/mole). After weighing the tablets in one bottle, the student found that the average mass of TUMS tablet was ...
Pdf - Text of NPTEL IIT Video Lectures
... of liquid and vapour exists. If we do this experiment for different temperatures, we will have different points for saturated vapour. Similarly, we will have different points for saturated liquid. If we combine all these points by some sort of a smooth curve, then you will get some sort of a dome li ...
... of liquid and vapour exists. If we do this experiment for different temperatures, we will have different points for saturated vapour. Similarly, we will have different points for saturated liquid. If we combine all these points by some sort of a smooth curve, then you will get some sort of a dome li ...
AP `99 Multiple Choice
... (NH4)2SO4(s) (E) Sr(NO3)2(s) 66. When solid ammonium chloride, NH4Cl(s), is added to water at 25_C it dissolves and the temperature of the solution decreases. Which of the following is true for the values of _H and _S for the dissolving process? (A) Positive ...
... (NH4)2SO4(s) (E) Sr(NO3)2(s) 66. When solid ammonium chloride, NH4Cl(s), is added to water at 25_C it dissolves and the temperature of the solution decreases. Which of the following is true for the values of _H and _S for the dissolving process? (A) Positive ...
1999 Advanced Placement Chemistry Exam
... (NH4)2SO4(s) (E) Sr(NO3)2(s) 66. When solid ammonium chloride, NH4Cl(s), is added to water at 25_C it dissolves and the temperature of the solution decreases. Which of the following is true for the values of _H and _S for the dissolving process? (A) Positive ...
... (NH4)2SO4(s) (E) Sr(NO3)2(s) 66. When solid ammonium chloride, NH4Cl(s), is added to water at 25_C it dissolves and the temperature of the solution decreases. Which of the following is true for the values of _H and _S for the dissolving process? (A) Positive ...
2 - College of Arts and Sciences
... melting point at each pressure The liquid – vapor line is the boiling point at that pressure The high critical temperature and critical pressure are due to strong van der Waals forces between water molecules Phase Diagram of Water ...
... melting point at each pressure The liquid – vapor line is the boiling point at that pressure The high critical temperature and critical pressure are due to strong van der Waals forces between water molecules Phase Diagram of Water ...
Chapter 2 - Portal UniMAP
... • Pressure units: dynes/cm2, lbf/in2 or psi, (N/m2 is called a Pascal (Pa) ...
... • Pressure units: dynes/cm2, lbf/in2 or psi, (N/m2 is called a Pascal (Pa) ...
DERS TANITIM BİLGİLERİ (İNGİLİZCE)
... The course will provide the student with basic thermodynamic tools for dealing with some of chemical problems occurring in industry. It will also help the student to obtain a practical knowledge of classical thermodynamics specifically by including the calculation of thermophysical properties. per ...
... The course will provide the student with basic thermodynamic tools for dealing with some of chemical problems occurring in industry. It will also help the student to obtain a practical knowledge of classical thermodynamics specifically by including the calculation of thermophysical properties. per ...
Designing a simple Electronic Timer Circuit using Crocodile Clips
... • The first components to be connected together are the two resistors and the battery. • Move the mouse cursor until it touches the positive battery terminal and press the Select on the mouse. • The arrow cursor will change to a symbol which looks like a roll of tape. Keeping the button pressed, dra ...
... • The first components to be connected together are the two resistors and the battery. • Move the mouse cursor until it touches the positive battery terminal and press the Select on the mouse. • The arrow cursor will change to a symbol which looks like a roll of tape. Keeping the button pressed, dra ...
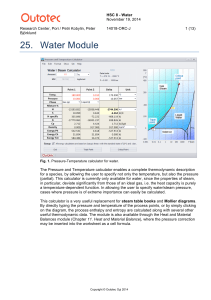
Water - HSC Chemistry 9
... than 0.032 bar, the Cp functions for steam follow the saturated steam curve below the boiling point. This means that the pressure is not kept constant below this point and thus the pressure value in the species name, i.e. X in H2O(Xbarg), is no longer valid for these temperatures. This may be seen i ...
... than 0.032 bar, the Cp functions for steam follow the saturated steam curve below the boiling point. This means that the pressure is not kept constant below this point and thus the pressure value in the species name, i.e. X in H2O(Xbarg), is no longer valid for these temperatures. This may be seen i ...
Choosing Trays and Packings for Distillation
... outlet), the pressure drop across the tray, and the frictional losses in the downcomer. As the tray pressure drop increases and the liquid rate increases, the froth level in the downcomer will increase. When the froth level, or downcomer backup, exceeds the downcomer height, the tray above will floo ...
... outlet), the pressure drop across the tray, and the frictional losses in the downcomer. As the tray pressure drop increases and the liquid rate increases, the froth level in the downcomer will increase. When the froth level, or downcomer backup, exceeds the downcomer height, the tray above will floo ...