Cr 6+ ions
... spectrum of a high-temperature YAG:Cr3+(Fig. 18). The first signal at g = 1.99 corresponds with ions of chrome taking place in nonuniform field of hydrated ions ligandes. The second signal at g ~ 3.56 – 3.50 corresponds to the most intensive line of thin structure of a spectrum of Cr3+, which occupi ...
... spectrum of a high-temperature YAG:Cr3+(Fig. 18). The first signal at g = 1.99 corresponds with ions of chrome taking place in nonuniform field of hydrated ions ligandes. The second signal at g ~ 3.56 – 3.50 corresponds to the most intensive line of thin structure of a spectrum of Cr3+, which occupi ...
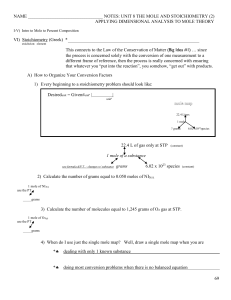
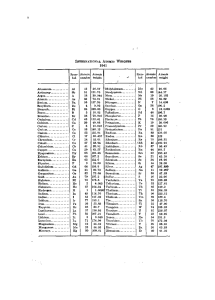
NAME NOTES: UNIT 8 THE MOLE AND STOICHIOMETRY (2
... These aqueous solutions may be described in terms of their concentration or their MOLARITY. If you were to update your Concept Map of Matter you would have the following: ...
... These aqueous solutions may be described in terms of their concentration or their MOLARITY. If you were to update your Concept Map of Matter you would have the following: ...
Hale.ICNAA.paper14
... The experiments generally quote data for “onset” nucleation conditions. For all but one set of the experimental data a rate of 1 cm-3s-1 is used in making the plot. For the data of Fladerer113 an approximate rate of 107 2 cm-3s-1 is used. At large scaled supersaturations (small values of the absci ...
... The experiments generally quote data for “onset” nucleation conditions. For all but one set of the experimental data a rate of 1 cm-3s-1 is used in making the plot. For the data of Fladerer113 an approximate rate of 107 2 cm-3s-1 is used. At large scaled supersaturations (small values of the absci ...
Flammability Limits _MS thesis_
... Flammable substances, which undergo exothermic reaction in the presence of air when exposed to an ignition source, are prevalent in today’s chemical and petrochemical industries. Most hydrocarbons are extremely volatile under relatively normal operation conditions. To prevent workplace explosions of ...
... Flammable substances, which undergo exothermic reaction in the presence of air when exposed to an ignition source, are prevalent in today’s chemical and petrochemical industries. Most hydrocarbons are extremely volatile under relatively normal operation conditions. To prevent workplace explosions of ...
Correlated/non-correlated ion dynamics of charge
... The title salt, 3-butyl-1-methyl-1H-imidazolium hexafluorophosphate, [C4mim][PF6] (see Fig. 1), has to date received much attention in the arena of molecular dynamics (MD), usually employing static high field approaches that probe fast motions.9–11 Our interest in this particular salt, which possess ...
... The title salt, 3-butyl-1-methyl-1H-imidazolium hexafluorophosphate, [C4mim][PF6] (see Fig. 1), has to date received much attention in the arena of molecular dynamics (MD), usually employing static high field approaches that probe fast motions.9–11 Our interest in this particular salt, which possess ...
Fluorian garnets from the host rocks of the Skaergaard intrusion
... mineral assemblage, and stratigraphic reconstructions indicate pressures of 1 kbar. Assessment of isobaric, isothermal phase relations in the system CaO-A1203-Si02-H20HF allows estimation of fluid characteristics in equilibrium with fluorian grandite-bearing assemblages. The presence of fluorite, wo ...
... mineral assemblage, and stratigraphic reconstructions indicate pressures of 1 kbar. Assessment of isobaric, isothermal phase relations in the system CaO-A1203-Si02-H20HF allows estimation of fluid characteristics in equilibrium with fluorian grandite-bearing assemblages. The presence of fluorite, wo ...
X: Ag, Ca, In, Li, Na, Sn, Sr and Zn
... additions of Li, Na, Ca, Zn, Ag, In, Sr, and Sn can improve the mechanical properties of Mgbased alloys, by forming secondary precipitates in the Mg matrix. In developing new magnesium alloys, it is important to understand their constitution (microstructure) and thermodynamic behaviour. Obtaining su ...
... additions of Li, Na, Ca, Zn, Ag, In, Sr, and Sn can improve the mechanical properties of Mgbased alloys, by forming secondary precipitates in the Mg matrix. In developing new magnesium alloys, it is important to understand their constitution (microstructure) and thermodynamic behaviour. Obtaining su ...
Liquid Intrusion and Alternative Methods for the Characterisation of
... In the application of mercury porosimetry, the volume of mercury entering the pore structure is measured as the applied (hydrostatic) pressure is gradually increased. The value νi(Hg) at the applied pressure pi apparently gives the cumulative volume of all available pores of radius equal to, or grea ...
... In the application of mercury porosimetry, the volume of mercury entering the pore structure is measured as the applied (hydrostatic) pressure is gradually increased. The value νi(Hg) at the applied pressure pi apparently gives the cumulative volume of all available pores of radius equal to, or grea ...
Open DongwonShin - ETDA
... 2.3 Heat capacity of aluminum from the SGTE pure element database[11]. 2.4 Gibbs energies of the individual phases of pure aluminum. The reference state is given as fcc phase at all temperatures. . . . . . . . 2.5 Geometry of the Redlich-Kister type polynomial interaction parameters in the A-B binar ...
... 2.3 Heat capacity of aluminum from the SGTE pure element database[11]. 2.4 Gibbs energies of the individual phases of pure aluminum. The reference state is given as fcc phase at all temperatures. . . . . . . . 2.5 Geometry of the Redlich-Kister type polynomial interaction parameters in the A-B binar ...
Electrodeposition of rare earth metals in ionic liquids
... Ionic liquids gain increasing attention in the recent years, as electrolytes for the recovery of metals more electropositive than hydrogen. It concerns for salts that are generally liquid at ambient conditions, consisting of a bulky organic cation and a smaller organic or inorganic anion [1]. The ...
... Ionic liquids gain increasing attention in the recent years, as electrolytes for the recovery of metals more electropositive than hydrogen. It concerns for salts that are generally liquid at ambient conditions, consisting of a bulky organic cation and a smaller organic or inorganic anion [1]. The ...
Ternary nucleation of inorganic acids, ammonia, and water
... The Gibbs free energy of formation of an incompressible liquid cluster in an ideal ternary vapor can be written as13 ...
... The Gibbs free energy of formation of an incompressible liquid cluster in an ideal ternary vapor can be written as13 ...
Cliffs Notes
... Acquisitions Editor: Sherry Gomoll Technical Editor: Christopher Bushee Production Proofreader: Joel K. Draper Hungry Minds Indianapolis Production Services ...
... Acquisitions Editor: Sherry Gomoll Technical Editor: Christopher Bushee Production Proofreader: Joel K. Draper Hungry Minds Indianapolis Production Services ...
IIT-JEE (Advanced) - Brilliant Public School Sitamarhi
... wt. of the mixture taken = 2g Loss in weight on heating = 0.124 g. Q.17 A 10 g sample of a mixture of calcium chloride and sodium chloride is treated with Na2CO3 to precipitate calcium as calcium carbonate. This CaCO3 is heated to convert all the calcium to CaO and the final mass of CaO is 1.62g. Ca ...
... wt. of the mixture taken = 2g Loss in weight on heating = 0.124 g. Q.17 A 10 g sample of a mixture of calcium chloride and sodium chloride is treated with Na2CO3 to precipitate calcium as calcium carbonate. This CaCO3 is heated to convert all the calcium to CaO and the final mass of CaO is 1.62g. Ca ...
Fundamentals of Combustion
... mixtures Some general issues . . . . . . . . . . . . . . . . . . . Ideal and non-ideal mixtures . . . . . . . . . . . . . . Ideal mixtures of ideal gases . . . . . . . . . . . . . . 2.3.1 Dalton model . . . . . . . . . . . . . . . . . . 2.3.1.1 Binary mixtures . . . . . . . . . . . 2.3.1.2 Entropy o ...
... mixtures Some general issues . . . . . . . . . . . . . . . . . . . Ideal and non-ideal mixtures . . . . . . . . . . . . . . Ideal mixtures of ideal gases . . . . . . . . . . . . . . 2.3.1 Dalton model . . . . . . . . . . . . . . . . . . 2.3.1.1 Binary mixtures . . . . . . . . . . . 2.3.1.2 Entropy o ...
1. (a) (i) 2Ca(NO3)2 → 2CaO + 4NO2 + O2 formulae correct (1
... With vertical section 3–5 units in length (1) at a volume of HCl of 20 cm3 (1) Final pH of between 1 and 2 (1) Named indicator consequential on vertical part of their graph (1) Because all of its range is within the vertical part of the graph / pKind ± 1 is within vertical part of graph / it changes ...
... With vertical section 3–5 units in length (1) at a volume of HCl of 20 cm3 (1) Final pH of between 1 and 2 (1) Named indicator consequential on vertical part of their graph (1) Because all of its range is within the vertical part of the graph / pKind ± 1 is within vertical part of graph / it changes ...
Chemical Vapor Deposition of Si and SiGe Films for High
... technology is built on two important innovations concerning the emitter and the base. The current gain for a transistor was greatly increased when a polycrystalline Si (poly-Si) layer was used to contact the intrinsic emitter of single crystalline Si. This is now widely termed as poly-Si emitter tec ...
... technology is built on two important innovations concerning the emitter and the base. The current gain for a transistor was greatly increased when a polycrystalline Si (poly-Si) layer was used to contact the intrinsic emitter of single crystalline Si. This is now widely termed as poly-Si emitter tec ...
DYNAMICS OF GLASS-FORMING LIQUIDS:
... likely similar to highly aged ordinary glasses, with low enthalpy and high thermal stability. Two recent experiments document additional features of these tightly packed glasses: 1) the beta relaxation can be suppressed by nearly a factor of four in vapor-deposited toluene glasses, and 2) the trans- ...
... likely similar to highly aged ordinary glasses, with low enthalpy and high thermal stability. Two recent experiments document additional features of these tightly packed glasses: 1) the beta relaxation can be suppressed by nearly a factor of four in vapor-deposited toluene glasses, and 2) the trans- ...
chemistry worksheet # 2: the mole as a unit of mass
... of particles. While a dozen is only 12 particles a mole is a much larger number—6.02 x 1023 particles. Elements generally exist as the particles we call atoms. A mole of carbon contains 6.02 x 1023 atoms of carbon. A mole of helium contains 6.02 x 1023 atoms of helium. A mole of sodium contains 6.02 ...
... of particles. While a dozen is only 12 particles a mole is a much larger number—6.02 x 1023 particles. Elements generally exist as the particles we call atoms. A mole of carbon contains 6.02 x 1023 atoms of carbon. A mole of helium contains 6.02 x 1023 atoms of helium. A mole of sodium contains 6.02 ...
Basic Stoichometry
... Imagine if you made a batch of cookies and used way too many eggs, or not enough sugar. YUCK! In chemistry, reactions proceed with very specific recipes. The study of these recipes is stoichiometry. When the reactants are present in the correct amounts, the reaction will produce products. What happe ...
... Imagine if you made a batch of cookies and used way too many eggs, or not enough sugar. YUCK! In chemistry, reactions proceed with very specific recipes. The study of these recipes is stoichiometry. When the reactants are present in the correct amounts, the reaction will produce products. What happe ...
2 H 2
... – Qualitative: quality (Colors, textures, smells, tastes, appearance) of the substances – can be observed. – Quantitative: quantity (mass, length, temperature, volume) of the substances – can be measured. ...
... – Qualitative: quality (Colors, textures, smells, tastes, appearance) of the substances – can be observed. – Quantitative: quantity (mass, length, temperature, volume) of the substances – can be measured. ...