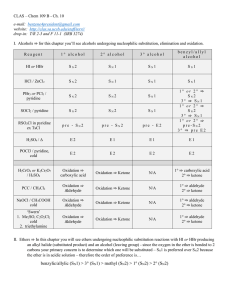
Organic Chemistry: An Indian Journal
... SCHEME 3. The reactions were carried out in the presence of potassium carbonate in acetone as solvent. The coupling of alcohols with aryl halides is a general and useful route for the synthesis of diaryl ethers. In this regard, Li et al. [19] exploited microwave irradiation in the presence of potass ...
... SCHEME 3. The reactions were carried out in the presence of potassium carbonate in acetone as solvent. The coupling of alcohols with aryl halides is a general and useful route for the synthesis of diaryl ethers. In this regard, Li et al. [19] exploited microwave irradiation in the presence of potass ...
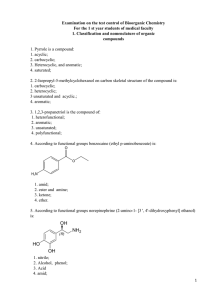
2. 2-Isopropyl-5-methylcyclohexanol on carbon skeletal
... 1. OH- group consisting of carboxylic acid functional groups; 2. hydroxyl group with sp 3-hybrid oxygen; 3. N-H acidic center; 4. OH group bonded directly to the heterocycle; 39. The most strong acidic properties of the compound shown in: 1. acetic acid; 2. propanoic acid; 3. 2-methylpropanoic acid; ...
... 1. OH- group consisting of carboxylic acid functional groups; 2. hydroxyl group with sp 3-hybrid oxygen; 3. N-H acidic center; 4. OH group bonded directly to the heterocycle; 39. The most strong acidic properties of the compound shown in: 1. acetic acid; 2. propanoic acid; 3. 2-methylpropanoic acid; ...
Shielding vs. Deshielding
... chemistry is a property of the substituents or functional groups in a chemical compound. The effect is used in a qualitative way and describes the electron withdrawing or releasing properties of the substituents based on relevant resonance structures and is symbolized by the letter M. The mesomeric ...
... chemistry is a property of the substituents or functional groups in a chemical compound. The effect is used in a qualitative way and describes the electron withdrawing or releasing properties of the substituents based on relevant resonance structures and is symbolized by the letter M. The mesomeric ...
chapter20(10-27-14)
... • For a fair comparison you would have to compare the boiling point of dimethylamine with that of ethylamine. They are isomers of each other - each contains exactly the same number of the same atoms. • The boiling point of the secondary amine is a little lower than the corresponding primary amine wi ...
... • For a fair comparison you would have to compare the boiling point of dimethylamine with that of ethylamine. They are isomers of each other - each contains exactly the same number of the same atoms. • The boiling point of the secondary amine is a little lower than the corresponding primary amine wi ...
Chapter 19. Aldehydes and Ketones: Nucleophilic Addition Reactions
... CrO3 in aqueous acid oxidizes aldehydes to carboxylic acids ...
... CrO3 in aqueous acid oxidizes aldehydes to carboxylic acids ...
NITRO COMPOUNDS
... as nitration of alkanes requires a very high temperature . This may be due to the fact that the nitration of arenes involves the initial formation of electrophilic reagent while the nitration of alkanes is supposed to involve the initial formation of free radical . The formation of free radical take ...
... as nitration of alkanes requires a very high temperature . This may be due to the fact that the nitration of arenes involves the initial formation of electrophilic reagent while the nitration of alkanes is supposed to involve the initial formation of free radical . The formation of free radical take ...