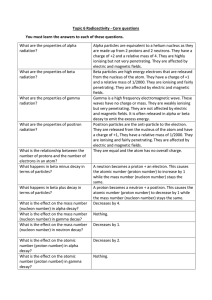
Topic 6 Radioactivity Core Questions
... Alpha particles are equivalent to a helium nucleus as they are made up from 2 protons and 2 neutrons. They have a charge of +2 and a relative mass of 4. They are highly ionising but not very penetrating. They are affected by electric and magnetic fields. Beta particles are high energy electrons that ...
... Alpha particles are equivalent to a helium nucleus as they are made up from 2 protons and 2 neutrons. They have a charge of +2 and a relative mass of 4. They are highly ionising but not very penetrating. They are affected by electric and magnetic fields. Beta particles are high energy electrons that ...
chem 1 TIFF new.indd
... For example, the smallest element is hydrogen. It has an atomic number of 1, which means it has only one proton. It also has only one electron, since the number of protons equals the number of electrons. Though atoms are very small, each one has a weight, called the atomic weight. For most atoms the ...
... For example, the smallest element is hydrogen. It has an atomic number of 1, which means it has only one proton. It also has only one electron, since the number of protons equals the number of electrons. Though atoms are very small, each one has a weight, called the atomic weight. For most atoms the ...
2:1 Foundations of Atomic Theory In the 1790s the study of matter
... The table above shows that a proton and neutron are approximately the same size and are 2000 times larger than an electron. If a large football stadium were an atom, this would make the nucleus (including the protons and neutrons) about the size of a marble. The electrons would be similar to dust pa ...
... The table above shows that a proton and neutron are approximately the same size and are 2000 times larger than an electron. If a large football stadium were an atom, this would make the nucleus (including the protons and neutrons) about the size of a marble. The electrons would be similar to dust pa ...
The average atomic mass of an element is the sum of the
... atomic number of chlorine is 17 (it has 17 protons in its nucleus). To calculate the average mass, first convert the percentages intofractions (divide them by 100). Then, calculate the mass numbers. The chlorine isotope with 18 neutrons has an abundance of 0.7577 and a mass number of 35 amu. To calc ...
... atomic number of chlorine is 17 (it has 17 protons in its nucleus). To calculate the average mass, first convert the percentages intofractions (divide them by 100). Then, calculate the mass numbers. The chlorine isotope with 18 neutrons has an abundance of 0.7577 and a mass number of 35 amu. To calc ...
Matter - Moodle
... • The chemical composition ______________________ A chemical property describes how a substance ________________ into a new substance Either by: • __________________ with other elements • _________________ __________________ into new substances ...
... • The chemical composition ______________________ A chemical property describes how a substance ________________ into a new substance Either by: • __________________ with other elements • _________________ __________________ into new substances ...
Elements, Isotopes, and Ions
... – Is the total mass of a certain ISOTOPE of an element. 1. How to calculate mass #: # of protons + # of neutrons = mass # 2. How to calculate # of neutrons from mass #: (Mass #) ...
... – Is the total mass of a certain ISOTOPE of an element. 1. How to calculate mass #: # of protons + # of neutrons = mass # 2. How to calculate # of neutrons from mass #: (Mass #) ...
1.2--NOTES--Basic Atomic Structure
... • all symbols must either be one capital letter, or one capital and one lowercase. •atomic number = number of p+. Written in bottomleft-hand corner of symbol. Identifies the element. •mass number = number of p+ & n0. Identifies the isotope. Written in top-left-hand corner. •average atomic mass = wei ...
... • all symbols must either be one capital letter, or one capital and one lowercase. •atomic number = number of p+. Written in bottomleft-hand corner of symbol. Identifies the element. •mass number = number of p+ & n0. Identifies the isotope. Written in top-left-hand corner. •average atomic mass = wei ...
Answer on Question #47967 - Chemistry – Other
... e. A model in which the protons, electrons, and neutrons are evenly distributed throughout the volume of the atom 8. The nucleus of an atom is ____________. a. Positively charged and has a high density b. Negatively charged and has a high density c. Positively charged and has a low density d. Negat ...
... e. A model in which the protons, electrons, and neutrons are evenly distributed throughout the volume of the atom 8. The nucleus of an atom is ____________. a. Positively charged and has a high density b. Negatively charged and has a high density c. Positively charged and has a low density d. Negat ...
PreAP Chemistry
... 19. Challenge Nitrogen has two naturally occurring isotopes, N-14 and N-15. Its atomic mass is 14.007. Which isotope is more abundant? Explain. ...
... 19. Challenge Nitrogen has two naturally occurring isotopes, N-14 and N-15. Its atomic mass is 14.007. Which isotope is more abundant? Explain. ...
Name
... 16. Most of the alpha particles traveled through the gold atoms showing atoms are mostly empty space. Very few positively charged alpha particles deflected revealing a tiny, dense, positive region in atoms. 17. C 18. B 19. D 20. They are isotopes b/c they have different numbers of neutrons, but they ...
... 16. Most of the alpha particles traveled through the gold atoms showing atoms are mostly empty space. Very few positively charged alpha particles deflected revealing a tiny, dense, positive region in atoms. 17. C 18. B 19. D 20. They are isotopes b/c they have different numbers of neutrons, but they ...
8b Isotopes and Ions2
... Do particle inventories for each of the following isotopes: Si-30 Ar-36 Fe-54 ...
... Do particle inventories for each of the following isotopes: Si-30 Ar-36 Fe-54 ...
Chapter 3 study guide answers
... Because a few alpha particles bounced back from the foil, Rutherford concluded that they were ...
... Because a few alpha particles bounced back from the foil, Rutherford concluded that they were ...
unit-3-atoms-and-nuclear - Waukee Community School District Blogs
... b. The daughter nuclide thus has a new atomic number because the number of protons increases by 1; mass stays the same c. New atomic number = new element symbol ...
... b. The daughter nuclide thus has a new atomic number because the number of protons increases by 1; mass stays the same c. New atomic number = new element symbol ...
What are atoms? Notes - Riverdale Middle School
... • Periodic table - a chart that shows the elements in order of increasing atomic number. • Elements on the periodic table are organized in periods (rows) and groups (columns) according to their physical and chemical properties. Electrical resistance is a measure of how difficult it is for an electri ...
... • Periodic table - a chart that shows the elements in order of increasing atomic number. • Elements on the periodic table are organized in periods (rows) and groups (columns) according to their physical and chemical properties. Electrical resistance is a measure of how difficult it is for an electri ...
File
... meaning indivisible: the Greek philosopher Demokritos (460370 BCE) maintained that all matter could be divided and sub-divided into smaller and smaller units, and eventually there would be a tiny particle that could not be divided any further - an atom Understanding of atoms didn’t progress much b ...
... meaning indivisible: the Greek philosopher Demokritos (460370 BCE) maintained that all matter could be divided and sub-divided into smaller and smaller units, and eventually there would be a tiny particle that could not be divided any further - an atom Understanding of atoms didn’t progress much b ...
The Atom
... mass #’s (or simply, • Because of this, they have different _________ masses different ___________.) • Isotopes are the same element, but the atoms weigh a different neutrons amount because of the # of ______________. Examples---> (1) Carbon-12 & Carbon-13 (2) Chlorine-35 & Chlorine-37 (The # shown ...
... mass #’s (or simply, • Because of this, they have different _________ masses different ___________.) • Isotopes are the same element, but the atoms weigh a different neutrons amount because of the # of ______________. Examples---> (1) Carbon-12 & Carbon-13 (2) Chlorine-35 & Chlorine-37 (The # shown ...
01 - cloudfront.net
... 17. Analyzing Ideas John Dalton made a number of statements about atoms that are now known to be incorrect. Why do you think his atomic theory is still found in science textbooks? _______________________________________________________________ ________________________________________________________ ...
... 17. Analyzing Ideas John Dalton made a number of statements about atoms that are now known to be incorrect. Why do you think his atomic theory is still found in science textbooks? _______________________________________________________________ ________________________________________________________ ...
Atoms: The Building Blocks of Matter Date:
... number 15, which signifies that they are the same element. However, both A and C have different masses they are isotopes of that element. ------------------------------------------------------------------------------------------------Most of us learned that to find the average of a given series of ...
... number 15, which signifies that they are the same element. However, both A and C have different masses they are isotopes of that element. ------------------------------------------------------------------------------------------------Most of us learned that to find the average of a given series of ...
Chapter 4 The structure of the atom
... • All of his ideas were NOT based on empirical (experimental) science – just ideas ...
... • All of his ideas were NOT based on empirical (experimental) science – just ideas ...
quiz1review - WordPress.com
... The measure of the gravitational force of an object on Earth is called its _____________ ...
... The measure of the gravitational force of an object on Earth is called its _____________ ...
Name: Date: ______ Period: Unit 3 – Atomic Structure Review Time
... 4. What are the 5 major principles of Dalton’s atomic theory? 5. What were the major problems of Dalton’s atomic theory? 6. Which model was introduced because of the Gold foil experiment? 7. Which atomic model had a ring of electrons surrounding the positively charged core? 8. Who had the first EXTE ...
... 4. What are the 5 major principles of Dalton’s atomic theory? 5. What were the major problems of Dalton’s atomic theory? 6. Which model was introduced because of the Gold foil experiment? 7. Which atomic model had a ring of electrons surrounding the positively charged core? 8. Who had the first EXTE ...
P2 Topic 3
... P2 6.2 The discovery of the nucleus Until 1911, the accepted model of the atom was known as the plum pudding model (top diagram). It was believed that the atom was a ball of positive charge with negatively-charged electrons (discovered in 1897) buried inside. Then Ernest Rutherford, together with h ...
... P2 6.2 The discovery of the nucleus Until 1911, the accepted model of the atom was known as the plum pudding model (top diagram). It was believed that the atom was a ball of positive charge with negatively-charged electrons (discovered in 1897) buried inside. Then Ernest Rutherford, together with h ...
P2 Knowledge Powerpoint – WIP Part 2
... P2 6.2 The discovery of the nucleus Until 1911, the accepted model of the atom was known as the plum pudding model (top diagram). It was believed that the atom was a ball of positive charge with negatively-charged electrons (discovered in 1897) buried inside. Then Ernest Rutherford, together with h ...
... P2 6.2 The discovery of the nucleus Until 1911, the accepted model of the atom was known as the plum pudding model (top diagram). It was believed that the atom was a ball of positive charge with negatively-charged electrons (discovered in 1897) buried inside. Then Ernest Rutherford, together with h ...























