THE ATOM
... • Isotopes – different atoms of the same element that have the same number of protons but different numbers of neutrons • some isotopes are radioactive – they emit energy when the nucleus of the atom breaks down spontaneously • most radioactive isotopes are not dangerous • to determine if an isotope ...
... • Isotopes – different atoms of the same element that have the same number of protons but different numbers of neutrons • some isotopes are radioactive – they emit energy when the nucleus of the atom breaks down spontaneously • most radioactive isotopes are not dangerous • to determine if an isotope ...
K,7th Grade Test Review: Atoms and Chemical Reactions PART
... 1. __________ is the smallest unit of an element that is still that element. 2. __________ is a substance that cannot be broken down into similar substances by physical or chemical changes. 3. Protons and neutrons have a __________ of 1 unit. Electrons have almost none. 4. An atom with more protons ...
... 1. __________ is the smallest unit of an element that is still that element. 2. __________ is a substance that cannot be broken down into similar substances by physical or chemical changes. 3. Protons and neutrons have a __________ of 1 unit. Electrons have almost none. 4. An atom with more protons ...
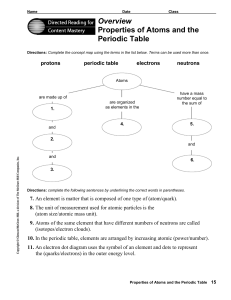
Overview Properties of Atoms and the Periodic Table
... 8. The unit of measurement used for atomic particles is the (atom size/atomic mass unit). 9. Atoms of the same element that have different numbers of neutrons are called (isotopes/electron clouds). 10. In the periodic table, elements are arranged by increasing atomic (power/number). 11. An electron ...
... 8. The unit of measurement used for atomic particles is the (atom size/atomic mass unit). 9. Atoms of the same element that have different numbers of neutrons are called (isotopes/electron clouds). 10. In the periodic table, elements are arranged by increasing atomic (power/number). 11. An electron ...
7.3 – Nuclear Reactions, Fission and Fusion 7.3.1 – Describe and
... We are all familiar with the equation E = mc 2. In this equation the rest energy of an object is given by E, the rest mass of an object is given by m and c is the speed of light which is quite large c = 3.0 X 108 m/s. The reason we say rest mass is due to relativity the mass of an object actually in ...
... We are all familiar with the equation E = mc 2. In this equation the rest energy of an object is given by E, the rest mass of an object is given by m and c is the speed of light which is quite large c = 3.0 X 108 m/s. The reason we say rest mass is due to relativity the mass of an object actually in ...
How many protons, electrons and neutrons are in an atom of krypton
... charged electrons. Atoms must have equal numbers of protons and electrons. In our example, an atom of krypton must contain 36 electrons since it contains 36 protons. Electrons are arranged around atoms in a special way. If you need to know how the electrons are arranged around an atom, take a look ...
... charged electrons. Atoms must have equal numbers of protons and electrons. In our example, an atom of krypton must contain 36 electrons since it contains 36 protons. Electrons are arranged around atoms in a special way. If you need to know how the electrons are arranged around an atom, take a look ...
Chapter 4 Atomic Structure
... identical. Atoms of any one element are different from those of any other element. ...
... identical. Atoms of any one element are different from those of any other element. ...
Chemistry Unit 2: Atomic Structure Unit Assignment #1 1. State the
... 12. How do the three isotopes of hydrogen (H–1, H–2, H–3) compare in terms of the numbers of subatomic particles in each? 13. Write the nuclear symbol for deuterium (H-2): a. Identify the atomic number b. Identify the mass number 14. Determine the number of protons, neutrons, and electrons in Co–59. ...
... 12. How do the three isotopes of hydrogen (H–1, H–2, H–3) compare in terms of the numbers of subatomic particles in each? 13. Write the nuclear symbol for deuterium (H-2): a. Identify the atomic number b. Identify the mass number 14. Determine the number of protons, neutrons, and electrons in Co–59. ...
Chem - Humble ISD
... Alpha radiation decay Occurs spontaneously in atoms with an atomic number greater than ______ Alpha particles – two protons and two neutrons are physically ejected from the atom o Which atom has 2 protons and 2 neutrons? Charge Symbol – Used by ______________________ Blocked by paper Nuc ...
... Alpha radiation decay Occurs spontaneously in atoms with an atomic number greater than ______ Alpha particles – two protons and two neutrons are physically ejected from the atom o Which atom has 2 protons and 2 neutrons? Charge Symbol – Used by ______________________ Blocked by paper Nuc ...
SUBATOMIC PARTICLES The three main subatomic particles found
... table is arranged in order of increasing atomic number, beginning with hydrogen (atomic number 1) and moving down the table to larger atoms such as uranium (atomic number 92). If an atom is electrically neutral, the number of protons (+) will equal the number of electrons (-). Therefore, if you know ...
... table is arranged in order of increasing atomic number, beginning with hydrogen (atomic number 1) and moving down the table to larger atoms such as uranium (atomic number 92). If an atom is electrically neutral, the number of protons (+) will equal the number of electrons (-). Therefore, if you know ...
Atoms - McEachern High School
... • All you have to know the rules that atoms follows. • Rules – The first energy level (or ring around the nucleus) only can hold 2 electrons – Once the first ring is “full”, the next set of electrons will begin to fill in to a new energy level. – With the exception of the first energy level, All ou ...
... • All you have to know the rules that atoms follows. • Rules – The first energy level (or ring around the nucleus) only can hold 2 electrons – Once the first ring is “full”, the next set of electrons will begin to fill in to a new energy level. – With the exception of the first energy level, All ou ...
Chapter3 atoms
... Dalton’s Atomic Theory (1808) All matter is composed of extremely small particles called atoms Atoms of a given element are identical in size, mass, and other properties; atoms of different John Dalton elements differ in size, mass, and other properties Atoms cannot be subdivided, created, or ...
... Dalton’s Atomic Theory (1808) All matter is composed of extremely small particles called atoms Atoms of a given element are identical in size, mass, and other properties; atoms of different John Dalton elements differ in size, mass, and other properties Atoms cannot be subdivided, created, or ...
Name Parts of an Atom Worksheet Date_______ Substances that
... With the exception of hydrogen, all atoms have three main parts. The parts of an atom are protons, electrons, and neutrons. A proton is positively charged and is located in the center or nucleus of the atom. Electrons are negatively charged and are located in rings or orbits spinning around the nucl ...
... With the exception of hydrogen, all atoms have three main parts. The parts of an atom are protons, electrons, and neutrons. A proton is positively charged and is located in the center or nucleus of the atom. Electrons are negatively charged and are located in rings or orbits spinning around the nucl ...
Zn 8 p + 8 p + 30 p + 8 n 8 n 35 n 8 e
... You will know what isotopes are You will be able to figure out the number of neutrons for each isotope ...
... You will know what isotopes are You will be able to figure out the number of neutrons for each isotope ...
File
... 4. For each of the following elements give the atomic number, mass number and the number of electrons: Hydrogen, Carbon, Neon and Calcium (you may make use of log tables). 5. Draw the Bohr structure of the following elements: Hydrogen, Carbon, Neon, Calcium (you may make use of log tables). 6. What ...
... 4. For each of the following elements give the atomic number, mass number and the number of electrons: Hydrogen, Carbon, Neon and Calcium (you may make use of log tables). 5. Draw the Bohr structure of the following elements: Hydrogen, Carbon, Neon, Calcium (you may make use of log tables). 6. What ...
What is the difference between artificial and natural transmutation
... Real-World examples of fission and fusion: 1. Summarize each scenario in 1-2 sentences. 2. Determine if the reaction depicts fission or fusion. 3. Write out the nuclear reaction associated with the scenario (the answers are posted at the bottom of the page—but not in the correct order) Nuclear Power ...
... Real-World examples of fission and fusion: 1. Summarize each scenario in 1-2 sentences. 2. Determine if the reaction depicts fission or fusion. 3. Write out the nuclear reaction associated with the scenario (the answers are posted at the bottom of the page—but not in the correct order) Nuclear Power ...
Thomson`s Atomic Model
... • The actual results did not fit Thompson’s model, so Rutherford proposed a new one. • He hypothesized that almost all the mass of the atom and all of its positive charge are crammed into an incredibly small region of space at the center of the atom called the nucleus. Most of the atom is empty spac ...
... • The actual results did not fit Thompson’s model, so Rutherford proposed a new one. • He hypothesized that almost all the mass of the atom and all of its positive charge are crammed into an incredibly small region of space at the center of the atom called the nucleus. Most of the atom is empty spac ...
Science - Byron High School
... ex. Think of cutting a piece of aluminum foil into smaller and smaller pieces. How far can it be cut? ...
... ex. Think of cutting a piece of aluminum foil into smaller and smaller pieces. How far can it be cut? ...
Atoms
... •Isotopes are atoms of the same element that differ in the number of neutrons. Isotopes of the same element have the same chemical properties, because they have the same number of protons and electrons. Isotopes are identified by mass number. Neutrons affect mass, so, isotopes with more neutrons ar ...
... •Isotopes are atoms of the same element that differ in the number of neutrons. Isotopes of the same element have the same chemical properties, because they have the same number of protons and electrons. Isotopes are identified by mass number. Neutrons affect mass, so, isotopes with more neutrons ar ...
PS 2.2 - S2TEM Centers SC
... Easter Egg Isotopes Introduction to the lesson: Isotopes have the same atomic number and hence nearly identical chemical behavior but different atomic masses. Most elements found in nature are mixtures of several isotopes; tin, for example, has 10 isotopes. In most cases, only stable isotopes of ele ...
... Easter Egg Isotopes Introduction to the lesson: Isotopes have the same atomic number and hence nearly identical chemical behavior but different atomic masses. Most elements found in nature are mixtures of several isotopes; tin, for example, has 10 isotopes. In most cases, only stable isotopes of ele ...
Structure of the Atom
... 3) Atoms combine in whole-number ratios to form compounds 4) In chemical reactions, atoms are combined, separated, or rearranged – but never changed into atoms of another element. ...
... 3) Atoms combine in whole-number ratios to form compounds 4) In chemical reactions, atoms are combined, separated, or rearranged – but never changed into atoms of another element. ...
Everything around us is made up of atoms. Atoms are one of the
... The center of the atom is called the nucleus. Neutrons and protons are located in the atomic nucleus. Electrons are very small particles located outside the nucleus. They orbit the nucleus at fantasist speeds, like the Earth orbits the sun. Each type of subatomic particle has a different electrical ...
... The center of the atom is called the nucleus. Neutrons and protons are located in the atomic nucleus. Electrons are very small particles located outside the nucleus. They orbit the nucleus at fantasist speeds, like the Earth orbits the sun. Each type of subatomic particle has a different electrical ...
protons and neutrons
... whole-number ratios to form chemical compounds In chemical reactions, atoms are combined, ...
... whole-number ratios to form chemical compounds In chemical reactions, atoms are combined, ...
Atomic Structure
... Weighted grades analogy Masses of subatomic particles ridiculously small – needed something more convenient Arbitrary amount set: Carbon-12 atom = 12.00000 amu 1 amu (atomic mass unit) = 1/12 mass of C-12 atom Mass of a single proton/neutron – 1 amu Why are the masses decimals? Most elements exist ...
... Weighted grades analogy Masses of subatomic particles ridiculously small – needed something more convenient Arbitrary amount set: Carbon-12 atom = 12.00000 amu 1 amu (atomic mass unit) = 1/12 mass of C-12 atom Mass of a single proton/neutron – 1 amu Why are the masses decimals? Most elements exist ...