A) electrons B) neutrons C) positrons D) protons 1. According to the
... A) aluminum C) magnesium ...
... A) aluminum C) magnesium ...
Ch 04 AtomicStructure
... A. The actual mass of an electron is very large compared to the actual mass of a proton. B. The actual masses of atoms are very small and difficult to work with. C. The number of subatomic particles in atoms of different elements varies. D. The actual masses of protons, electrons, and neutrons are n ...
... A. The actual mass of an electron is very large compared to the actual mass of a proton. B. The actual masses of atoms are very small and difficult to work with. C. The number of subatomic particles in atoms of different elements varies. D. The actual masses of protons, electrons, and neutrons are n ...
10.1 RG and answer key
... 5. _______________ An element’s mass number tells the number of protons in its nucleus. 6. _______________ Negatively charged particles in an atom are called electrons. 7. _______________ The cloud model of the atom describes the location of electrons as specific orbits around the nucleus. 8. ______ ...
... 5. _______________ An element’s mass number tells the number of protons in its nucleus. 6. _______________ Negatively charged particles in an atom are called electrons. 7. _______________ The cloud model of the atom describes the location of electrons as specific orbits around the nucleus. 8. ______ ...
Snímek 1
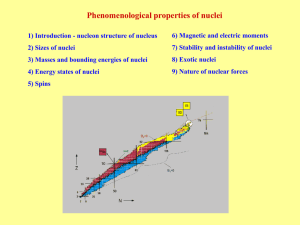
... Magic numbers – observed values of N and Z with increased stability. At 1896 H. Becquerel observed first sign of instability of nuclei – radioactivity. Instable nuclei irradiate: Alpha decay → nucleus transformation by 4He irradiation Beta decay → nucleus transformation by e-, e+ irradiation or capt ...
... Magic numbers – observed values of N and Z with increased stability. At 1896 H. Becquerel observed first sign of instability of nuclei – radioactivity. Instable nuclei irradiate: Alpha decay → nucleus transformation by 4He irradiation Beta decay → nucleus transformation by e-, e+ irradiation or capt ...
Chapter 16 Atomic Energy
... sometimes, particles. • The particles and energy given off are called nuclear radiation. • Unstable nuclei continue to decay until they form stable nuclei. Three kinds of radioactive decay are alpha decay, beta decay, and gamma decay. Copyright © Houghton Mifflin Harcourt Publishing Company ...
... sometimes, particles. • The particles and energy given off are called nuclear radiation. • Unstable nuclei continue to decay until they form stable nuclei. Three kinds of radioactive decay are alpha decay, beta decay, and gamma decay. Copyright © Houghton Mifflin Harcourt Publishing Company ...
Chem Ch. 4.3
... • The atomic mass of an element is the weighted average mass of the isotopes of that element. • Atomic mass, therefore, takes into account the percent abundance of each isotope as well as the mass of an atom of that isotope. • The isotope in greater abundance will have a greater effect on determinin ...
... • The atomic mass of an element is the weighted average mass of the isotopes of that element. • Atomic mass, therefore, takes into account the percent abundance of each isotope as well as the mass of an atom of that isotope. • The isotope in greater abundance will have a greater effect on determinin ...
7.2- Nuclear reactions (PPT)
... balanced and the star collapses into a neutron star. ▪ This is called the iron catastrophe. ▪ Where do the elements above iron come from? ▪ For an even more massive star, there is enough gravity to overcome the neutron barrier to collapse. ▪ Collapse continues and nothing can stop it! ▪ The star bec ...
... balanced and the star collapses into a neutron star. ▪ This is called the iron catastrophe. ▪ Where do the elements above iron come from? ▪ For an even more massive star, there is enough gravity to overcome the neutron barrier to collapse. ▪ Collapse continues and nothing can stop it! ▪ The star bec ...
Slideshow
... In the case of Hydrogen, all three of these isotopes are naturally occurring. However, they don’t occur with equal frequency – 1H is the most common! The same applies to many other types of atoms. Carbon, for example, has three naturally occurring isotopes (12C, 13C, 14C). How many protons, neutrons ...
... In the case of Hydrogen, all three of these isotopes are naturally occurring. However, they don’t occur with equal frequency – 1H is the most common! The same applies to many other types of atoms. Carbon, for example, has three naturally occurring isotopes (12C, 13C, 14C). How many protons, neutrons ...
1. Atomic Structure
... positively charged because of the protons dense – it contains nearly all the mass of the atom in a tiny space. Electrons are: very small and light, and negatively charged able to be lost or gained in chemical reactions found thinly spread around the outside of the nucleus, orbiting in laye ...
... positively charged because of the protons dense – it contains nearly all the mass of the atom in a tiny space. Electrons are: very small and light, and negatively charged able to be lost or gained in chemical reactions found thinly spread around the outside of the nucleus, orbiting in laye ...
4.80 Chapter Outline
... have gold or water or any other recognizable substance. If broken apart, almost all atoms contain three smaller particles called protons, neutrons, and electrons. Because these particles are even smaller than an atom, they are called subatomic particles. These three types of particles are arranged i ...
... have gold or water or any other recognizable substance. If broken apart, almost all atoms contain three smaller particles called protons, neutrons, and electrons. Because these particles are even smaller than an atom, they are called subatomic particles. These three types of particles are arranged i ...
Elements and Compounds
... Electron Properties • R.A. Millikan (Univ. Of Chicago) determined charge magnitude of electrons which led to the determination of the mass • q= -1.6 x 10-19 Coulombs • Mass = 9.11 x 10-28 grams (from charge to mass ratio) ...
... Electron Properties • R.A. Millikan (Univ. Of Chicago) determined charge magnitude of electrons which led to the determination of the mass • q= -1.6 x 10-19 Coulombs • Mass = 9.11 x 10-28 grams (from charge to mass ratio) ...
atomic number
... Isotopes: are atoms that have the same number of protons but different numbers of neutrons. Because isotopes of an element have different numbers of neutrons, they also have different mass numbers. Isotopes are chemically alike because they have identical numbers of protons and electrons, which are ...
... Isotopes: are atoms that have the same number of protons but different numbers of neutrons. Because isotopes of an element have different numbers of neutrons, they also have different mass numbers. Isotopes are chemically alike because they have identical numbers of protons and electrons, which are ...
Topic exploration pack
... consider the nuclei as stable and indivisible. While learners, especially those who have also been learning chemistry, and indeed to a lesser extent biology, will already have encountered examples of atoms losing and gaining electrons, but, as far as most of the other topics in the course are concer ...
... consider the nuclei as stable and indivisible. While learners, especially those who have also been learning chemistry, and indeed to a lesser extent biology, will already have encountered examples of atoms losing and gaining electrons, but, as far as most of the other topics in the course are concer ...
Physics 9 Fall 2009 - faculty.ucmerced.edu
... A proton and an alpha particle (q = +2e, m = 4u) are fired directly toward each other from far away, each with an initial speed of 0.010c. What is their distance of closest approach, as measured between their centers? ...
... A proton and an alpha particle (q = +2e, m = 4u) are fired directly toward each other from far away, each with an initial speed of 0.010c. What is their distance of closest approach, as measured between their centers? ...
review of the review
... 5) As the alpha particle got closer to the nucleus, there was an increase in a) electric force, kinetic energy b) electric potential energy, kinetic energy c) electric potential energy, electric force, d) all three 6) The closest possible approach of an -particle can be calculated using conservatio ...
... 5) As the alpha particle got closer to the nucleus, there was an increase in a) electric force, kinetic energy b) electric potential energy, kinetic energy c) electric potential energy, electric force, d) all three 6) The closest possible approach of an -particle can be calculated using conservatio ...
PHY303 1 TURN OVER PHY303 Data Provided: A formula sheet
... prompt and delayed neutrons, chain reaction and fission fragments. (c) Consider neutrons of energy 0.1 eV incident on a piece of natural uranium. The cross section for fission of 235U at that energy is 250 barns. The amount of 235U in natural uranium is 0.72% and the density of uranium is 19 g cm3. ...
... prompt and delayed neutrons, chain reaction and fission fragments. (c) Consider neutrons of energy 0.1 eV incident on a piece of natural uranium. The cross section for fission of 235U at that energy is 250 barns. The amount of 235U in natural uranium is 0.72% and the density of uranium is 19 g cm3. ...
Atomic Structure - davis.k12.ut.us
... Thompson passed electric current through gases in a glass tube. The result was a glowing beam flowing from the negative side to the ...
... Thompson passed electric current through gases in a glass tube. The result was a glowing beam flowing from the negative side to the ...
Powerpoint covering atomic structure and isotopes
... positively charged because of the protons dense – it contains nearly all the mass of the atom in a tiny space. Electrons are: very small and light, and negatively charged able to be lost or gained in chemical reactions found thinly spread around the outside of the nucleus, orbiting in laye ...
... positively charged because of the protons dense – it contains nearly all the mass of the atom in a tiny space. Electrons are: very small and light, and negatively charged able to be lost or gained in chemical reactions found thinly spread around the outside of the nucleus, orbiting in laye ...
Decay Mechanisms - High Energy Physics Research at Minnesota
... absorption of a photon ( a topic to be covered later in the course). Note that emission of an Auger electron increases the number of vacancies in the atomic shells by one unit. Often we see Auger cascades in relatively heavy atoms as inner-shell vacancies are successively filled by the Auger process ...
... absorption of a photon ( a topic to be covered later in the course). Note that emission of an Auger electron increases the number of vacancies in the atomic shells by one unit. Often we see Auger cascades in relatively heavy atoms as inner-shell vacancies are successively filled by the Auger process ...
vibrations and waves
... ____________________ 3. Both Democritus and Dalton suggested that matter is made up of atoms. ____________________ 4. Dalton’s atomic theory stated that atoms separate, combine, or rearrange in chemical reactions. ____________________ 5. Dalton’s atomic theory stated that matter is mostly empty spac ...
... ____________________ 3. Both Democritus and Dalton suggested that matter is made up of atoms. ____________________ 4. Dalton’s atomic theory stated that atoms separate, combine, or rearrange in chemical reactions. ____________________ 5. Dalton’s atomic theory stated that matter is mostly empty spac ...
Structures of Matter
... 1. Write the word “BOOK” in your lab journal. With your class, look up the element names for each symbol in the word and list the element names. (Remember some of the elements might have 2 letters attached) Example: the word CAT : C-Carbon At-Astatine 2. Write the name “Cesar” in your lab journal. L ...
... 1. Write the word “BOOK” in your lab journal. With your class, look up the element names for each symbol in the word and list the element names. (Remember some of the elements might have 2 letters attached) Example: the word CAT : C-Carbon At-Astatine 2. Write the name “Cesar” in your lab journal. L ...
1.10 Atomic structure - Pearson Schools and FE Colleges
... 6 C. This is known as carbon-14. The nuclei of carbon-14 atoms have two extra neutrons when compared with carbon-12, so the two atoms have different mass numbers. The two different carbon atoms are called isotopes of the element carbon. There is also a third isotope of carbon with a mass number of ...
... 6 C. This is known as carbon-14. The nuclei of carbon-14 atoms have two extra neutrons when compared with carbon-12, so the two atoms have different mass numbers. The two different carbon atoms are called isotopes of the element carbon. There is also a third isotope of carbon with a mass number of ...
Matter - TeacherWeb
... one or more nonmetal atoms Often form ionic bonds with metal cation(s) (+) and nonmetal ...
... one or more nonmetal atoms Often form ionic bonds with metal cation(s) (+) and nonmetal ...
Chapter 4 Reviewing Content - Huber Heights City Schools
... Atoms are the smallest particle of an element that retains the properties of that element (68)The following table shows some of the data collected by Rutherford and his colleagues during ...
... Atoms are the smallest particle of an element that retains the properties of that element (68)The following table shows some of the data collected by Rutherford and his colleagues during ...