Exam2
... Ethylene glycol is the primary component in antifreeze. How many grams ofethylene glycol, C2H4(OH)2 are in 394 mL of a 9.087 M solution of antifreeze? (molar mass C2H4(OH)2 = 62 g/mol) a. 222 g b. 43.7 g c. 3.58 g d. 2.69 g e. 0.157 g Calculate the number of oxygen atoms in 45.0 g of Co2(SO4)3 (mola ...
... Ethylene glycol is the primary component in antifreeze. How many grams ofethylene glycol, C2H4(OH)2 are in 394 mL of a 9.087 M solution of antifreeze? (molar mass C2H4(OH)2 = 62 g/mol) a. 222 g b. 43.7 g c. 3.58 g d. 2.69 g e. 0.157 g Calculate the number of oxygen atoms in 45.0 g of Co2(SO4)3 (mola ...
Chapter 4 (Phosphines)
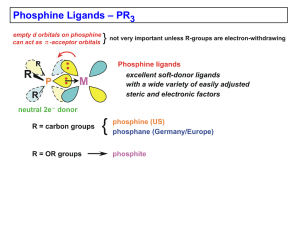
... are used to enforce cisoidal coordination geometries when needed. Some typical first row M-PR3 average bond distances: ...
... are used to enforce cisoidal coordination geometries when needed. Some typical first row M-PR3 average bond distances: ...
Structural Characterization of Natural Nickel and Copper
... unknown (Michael et al., 1993; Bertram and Pattenden, 2007; Comba et al., 2014). Although not directly produced by microbes, marine humic compounds, structurally heterogeneous refractory degradation products of organic matter decay, may also complex Cu and Ni (Baker and Khalili, 2003; Yang and Van d ...
... unknown (Michael et al., 1993; Bertram and Pattenden, 2007; Comba et al., 2014). Although not directly produced by microbes, marine humic compounds, structurally heterogeneous refractory degradation products of organic matter decay, may also complex Cu and Ni (Baker and Khalili, 2003; Yang and Van d ...
Journal of the American Chemical Society, 120(15)
... Abstract: In contrast to porphyrins and chlorins, the direct metalation of bacteriochlorins is difficult. Nevertheless, Cu2+ and Zn2+ can be introduced into bacteriopheophytin in acetic acid, whereas Cd2+ can be inserted in dimethylformamide. The former reactions depend on the substituents of the is ...
... Abstract: In contrast to porphyrins and chlorins, the direct metalation of bacteriochlorins is difficult. Nevertheless, Cu2+ and Zn2+ can be introduced into bacteriopheophytin in acetic acid, whereas Cd2+ can be inserted in dimethylformamide. The former reactions depend on the substituents of the is ...
The Nomenclature of Polyoxometalates: How To Connect a Name
... polyhedrons are fused, having common vertices and common edges, even sometimes common faces. The various sharings may also occur in the same compound, thus increasing the difficulty. Thus the central part of the problem is to name the core of fused octahedrons or, to be more precise, how to refer to ...
... polyhedrons are fused, having common vertices and common edges, even sometimes common faces. The various sharings may also occur in the same compound, thus increasing the difficulty. Thus the central part of the problem is to name the core of fused octahedrons or, to be more precise, how to refer to ...
http://doc.rero.ch
... The coordination chemistry of groups 1 and 2 metal compounds with organic ligands in the widest sense has been, until relatively recently, largely unknown compared to transition metal coordination networks. This is true despite the fact that many s-block metal–organic compounds are already of commer ...
... The coordination chemistry of groups 1 and 2 metal compounds with organic ligands in the widest sense has been, until relatively recently, largely unknown compared to transition metal coordination networks. This is true despite the fact that many s-block metal–organic compounds are already of commer ...
9. Coordination Compounds
... 6. Co-ordination sphere: The central atom/ion and the ligands attached to it are enclosed in square bracket and is collectively termed as the co-ordination sphere. The ionisable groups are written outside the bracket and are called simple ions or counter ions. For example, in the complex K4[Fe(CN)6] ...
... 6. Co-ordination sphere: The central atom/ion and the ligands attached to it are enclosed in square bracket and is collectively termed as the co-ordination sphere. The ionisable groups are written outside the bracket and are called simple ions or counter ions. For example, in the complex K4[Fe(CN)6] ...
Copper(II) and lead(II) complexation by humic acid and
... and Cu(II)–humate in the total metal and ligand concentration range. This is in accordance with the number of binding sites in the ligands. In addition, the differences between the percentages of the total metal bound to the resin for same ligand were greater with increasing concentration of ligand. ...
... and Cu(II)–humate in the total metal and ligand concentration range. This is in accordance with the number of binding sites in the ligands. In addition, the differences between the percentages of the total metal bound to the resin for same ligand were greater with increasing concentration of ligand. ...
Effect of an industrial chemical waste on the uptake
... and Cu(II)–humate in the total metal and ligand concentration range. This is in accordance with the number of binding sites in the ligands. In addition, the differences between the percentages of the total metal bound to the resin for same ligand were greater with increasing concentration of ligand. ...
... and Cu(II)–humate in the total metal and ligand concentration range. This is in accordance with the number of binding sites in the ligands. In addition, the differences between the percentages of the total metal bound to the resin for same ligand were greater with increasing concentration of ligand. ...
Role of Amine–Cavity Interactions in Determining the Structure and
... mechanical properties. In such materials, interactions between the protonated amine and the metal−formate cavity have a large impact on the mechanical properties. We use complementary single-crystal X-ray diffraction and 1H solid state nuclear magnetic resonance spectroscopy to investigate amine−cavi ...
... mechanical properties. In such materials, interactions between the protonated amine and the metal−formate cavity have a large impact on the mechanical properties. We use complementary single-crystal X-ray diffraction and 1H solid state nuclear magnetic resonance spectroscopy to investigate amine−cavi ...
Document
... • For this reason, a transition metal is defined as being an element which forms at least one ion with a partially filled sub-shell of d electrons. – In period 4 only Ti-Cu are TM’s! – Note that when d block elements form ions the s electrons are lost first ...
... • For this reason, a transition metal is defined as being an element which forms at least one ion with a partially filled sub-shell of d electrons. – In period 4 only Ti-Cu are TM’s! – Note that when d block elements form ions the s electrons are lost first ...
Coordination Chemistry Prof. Debashis Ray Department of
... coordination from the fifth coordination site and the sixth coordination site like two donor groups like X and Y, we get a typical octahedral coordination around the metal center. If the metal center what we are just seeing in the slide, that iron is there, so if we consider that iron is present; so ...
... coordination from the fifth coordination site and the sixth coordination site like two donor groups like X and Y, we get a typical octahedral coordination around the metal center. If the metal center what we are just seeing in the slide, that iron is there, so if we consider that iron is present; so ...
Laser and its applications
... The resonator is an optical “feed back device” that directs photons back and forth through the laser medium. Resonator or “optical activity” consists of a pair of carefully aligned plane or curved mirrors (see figure 2). One of them is chosen with a reflectivity 100% as possible. The other mirror is ...
... The resonator is an optical “feed back device” that directs photons back and forth through the laser medium. Resonator or “optical activity” consists of a pair of carefully aligned plane or curved mirrors (see figure 2). One of them is chosen with a reflectivity 100% as possible. The other mirror is ...
The Transition Metals
... Metals are classified in terms of what kinds of ores they form (Interactive Figure 23.2.1). The elements on the left of the transition metals tend to form oxide ores; these metals are called lithophiles. Those on the right side are called chalcophiles and form sulfide ores. Siderophiles have an affi ...
... Metals are classified in terms of what kinds of ores they form (Interactive Figure 23.2.1). The elements on the left of the transition metals tend to form oxide ores; these metals are called lithophiles. Those on the right side are called chalcophiles and form sulfide ores. Siderophiles have an affi ...
Inorganic Chemistry Sixth Edition Chapter 7
... Valence Bond theory provides the hybridization for octahedral complexes. For the first row transition metals, the hybridization can be: d2sp3 (using the 3d, 4s and 4p orbitals), or sp3d2 (using the 4s, 4p and 4d orbitals). The valence bond approach isn’t used because it fails to explain the electron ...
... Valence Bond theory provides the hybridization for octahedral complexes. For the first row transition metals, the hybridization can be: d2sp3 (using the 3d, 4s and 4p orbitals), or sp3d2 (using the 4s, 4p and 4d orbitals). The valence bond approach isn’t used because it fails to explain the electron ...
5. Bonding in Complexes
... Further ideas, models, and theories to explain the nature of the bond between the central ion and its ligands in transition metal complexes are: Crystal field theory (CFT) of H. Bethe , J.H. van Vleck , and L. Orgel (192935), treats the ligands as point charges or dipoles and describes how they infl ...
... Further ideas, models, and theories to explain the nature of the bond between the central ion and its ligands in transition metal complexes are: Crystal field theory (CFT) of H. Bethe , J.H. van Vleck , and L. Orgel (192935), treats the ligands as point charges or dipoles and describes how they infl ...
Degrees of freedom effect on fragmentation in tandem mass
... significantly different for various homopolymers. The slope was found to decrease with the activation energy of dissociation, decreasing in the order of polyethers > polymethacrylates > polyesters > polysaccharides. This trend suggested that the slope of the CCE trendline is linearly proportional to ...
... significantly different for various homopolymers. The slope was found to decrease with the activation energy of dissociation, decreasing in the order of polyethers > polymethacrylates > polyesters > polysaccharides. This trend suggested that the slope of the CCE trendline is linearly proportional to ...
II. Main types of organometallic compounds
... Many transition metal organometallics have significant catalysis activity, becoming one important catalyst in organic synthesis. Organometallic compounds have catalytic activity, which is realted to the weak bonding between L and M, making the formation and rupture of M-C bonds more easily, and tran ...
... Many transition metal organometallics have significant catalysis activity, becoming one important catalyst in organic synthesis. Organometallic compounds have catalytic activity, which is realted to the weak bonding between L and M, making the formation and rupture of M-C bonds more easily, and tran ...
3-3 More bonding.pptx
... Materials that are insulators (eg diamond) there is a large energy gap between the valence and conducKon ...
... Materials that are insulators (eg diamond) there is a large energy gap between the valence and conducKon ...
193 - Wayne State Chemistry
... To help understand the bonding, ab initio calculations14 were carried out at the B3LYP/6-311G* level of theory on a simplified model15 of the complex, trichloro(3,5-dimethylpyrazolato)titanium(IV). An attempt to optimize the monodentate complex proceeded smoothly to the bidentate complex, suggesting ...
... To help understand the bonding, ab initio calculations14 were carried out at the B3LYP/6-311G* level of theory on a simplified model15 of the complex, trichloro(3,5-dimethylpyrazolato)titanium(IV). An attempt to optimize the monodentate complex proceeded smoothly to the bidentate complex, suggesting ...
52142_present
... The significant IR bands of dihydrazones and their platinum complexes are summarized in Table 2. Ligands: Oxloyldihydrazones (L1-L4) can exist either in the trans (staggered) configuration or cis-configuration, Fig. 2, [27]. In cis- configuration, the dihydrazone can adopt either anti-cis- configura ...
... The significant IR bands of dihydrazones and their platinum complexes are summarized in Table 2. Ligands: Oxloyldihydrazones (L1-L4) can exist either in the trans (staggered) configuration or cis-configuration, Fig. 2, [27]. In cis- configuration, the dihydrazone can adopt either anti-cis- configura ...
Coordination properties of the diethyl 2-quinolilmethylphosphonate ligand with chloride
... remains almost at the same position as in the free ligand, indicating monodentate coordination of 2-qmpe. In fact, X-ray structural determination [25] of trans-[Pd(2qmpe)2Cl2] confirmed that the metal ion is bound in a N-monodentate way to the pyridine. Other ligand bands characteristic of the phosp ...
... remains almost at the same position as in the free ligand, indicating monodentate coordination of 2-qmpe. In fact, X-ray structural determination [25] of trans-[Pd(2qmpe)2Cl2] confirmed that the metal ion is bound in a N-monodentate way to the pyridine. Other ligand bands characteristic of the phosp ...
chemistry of transition elements
... configurations of the M+ ions are disrupted, with considerable loss of exchange energy. The value for Zn is correspondingly low as the ionisation leads to the production of the stable d10 configuration. The trend in the third ionisation enthalpies is not complicated by the 4s orbital factor and show ...
... configurations of the M+ ions are disrupted, with considerable loss of exchange energy. The value for Zn is correspondingly low as the ionisation leads to the production of the stable d10 configuration. The trend in the third ionisation enthalpies is not complicated by the 4s orbital factor and show ...
N-Ligand2011
... Ligand Assignment 1: Find one ligand that binds to transition metal ions through nitrogen (only through N, no mixed ligands) and answer the following questions (that will apply to all subsequent ligand assignments in addition to other questions that we will add over the course of the term). Please s ...
... Ligand Assignment 1: Find one ligand that binds to transition metal ions through nitrogen (only through N, no mixed ligands) and answer the following questions (that will apply to all subsequent ligand assignments in addition to other questions that we will add over the course of the term). Please s ...
Structural Chemistry of Titanium Alkoxides Substituted by the
... at low temperature [6]. Secondary amines can be coordinated in the same way; several isostructural amine adducts were recently structurally characterized [7]. ...
... at low temperature [6]. Secondary amines can be coordinated in the same way; several isostructural amine adducts were recently structurally characterized [7]. ...