Chapter 2: Atoms, Ions, and the Periodic Table
... A) Pure water is composed of the elements oxygen and hydrogen in a mass ratio of 8 to 1. B) Any sample of a given compound always contains the same proportions by mass of the component elements. C) The mass of the products of a chemical reaction is equal to the mass of the starting materials of the ...
... A) Pure water is composed of the elements oxygen and hydrogen in a mass ratio of 8 to 1. B) Any sample of a given compound always contains the same proportions by mass of the component elements. C) The mass of the products of a chemical reaction is equal to the mass of the starting materials of the ...
Chapter 2: Atoms, Ions, and the Periodic Table
... A) Pure water is composed of the elements oxygen and hydrogen in a mass ratio of 8 to 1. B) Any sample of a given compound always contains the same proportions by mass of the component elements. C) The mass of the products of a chemical reaction is equal to the mass of the starting materials of the ...
... A) Pure water is composed of the elements oxygen and hydrogen in a mass ratio of 8 to 1. B) Any sample of a given compound always contains the same proportions by mass of the component elements. C) The mass of the products of a chemical reaction is equal to the mass of the starting materials of the ...
Chapter 2: Atoms, Ions, and the Periodic Table
... A) Pure water is composed of the elements oxygen and hydrogen in a mass ratio of 8 to 1. B) Any sample of a given compound always contains the same proportions by mass of the component elements. C) The mass of the products of a chemical reaction is equal to the mass of the starting materials of the ...
... A) Pure water is composed of the elements oxygen and hydrogen in a mass ratio of 8 to 1. B) Any sample of a given compound always contains the same proportions by mass of the component elements. C) The mass of the products of a chemical reaction is equal to the mass of the starting materials of the ...
The Atomic Theory
... and others had observed was to suggest that materials like nitrogen, carbon, and oxygen were composed of small, indivisible quantities, which Dalton called “atoms” (in reference to Democritus’s original idea). Dalton used this idea to generate what is now known as Dalton’s atomic theory. Dalton’s at ...
... and others had observed was to suggest that materials like nitrogen, carbon, and oxygen were composed of small, indivisible quantities, which Dalton called “atoms” (in reference to Democritus’s original idea). Dalton used this idea to generate what is now known as Dalton’s atomic theory. Dalton’s at ...
4.3 Distinguishing Among Atoms
... Copyright © Pearson Education, Inc., or its affiliates. All Rights Reserved. ...
... Copyright © Pearson Education, Inc., or its affiliates. All Rights Reserved. ...
C 4 The Atomic Theory
... and others had observed was to suggest that materials like nitrogen, carbon, and oxygen were composed of small, indivisible quantities, which Dalton called “atoms” (in reference to Democritus’s original idea). Dalton used this idea to generate what is now known as Dalton’s atomic theory. Dalton’s at ...
... and others had observed was to suggest that materials like nitrogen, carbon, and oxygen were composed of small, indivisible quantities, which Dalton called “atoms” (in reference to Democritus’s original idea). Dalton used this idea to generate what is now known as Dalton’s atomic theory. Dalton’s at ...
Name: Date: ______ 1. Which of the following is a property of both
... (1) A molecule is a group of two or more atoms that functions as a unit because the atoms are bound together by chemical forces. (2) The crushing of ice to make ice chips is a physical procedure that involves a chemical change. (3) Most naturally occurring samples of matter are mixtures rather than ...
... (1) A molecule is a group of two or more atoms that functions as a unit because the atoms are bound together by chemical forces. (2) The crushing of ice to make ice chips is a physical procedure that involves a chemical change. (3) Most naturally occurring samples of matter are mixtures rather than ...
- Te Kura
... have tried to turn lead into gold to the physicists trying to work out what happens when matter is accelerated at tremendously high speeds. The last 150 years have seen an enormous increase in our knowledge about this fundamental building block. As new observations have been made, scientists have mo ...
... have tried to turn lead into gold to the physicists trying to work out what happens when matter is accelerated at tremendously high speeds. The last 150 years have seen an enormous increase in our knowledge about this fundamental building block. As new observations have been made, scientists have mo ...
Chapter 2 The Components of Matter
... The Modern Reassessment of the Atomic Theory 1. All matter is composed of atoms. The atom is the smallest body that retains the unique identity of the element. 2. Atoms of one element cannot be converted into atoms of another element in a chemical reaction. Elements can only be converted into other ...
... The Modern Reassessment of the Atomic Theory 1. All matter is composed of atoms. The atom is the smallest body that retains the unique identity of the element. 2. Atoms of one element cannot be converted into atoms of another element in a chemical reaction. Elements can only be converted into other ...
Inorganometallic Chemistry
... the Journal of Organometallic Chemistry write “Papers dealing with those aspects of organophosphorous and organoselenium chemistry that are of interest to the organometallic chemist will also be considered.” In most organometallic compounds, the metal-carbon bond has predominantly covalent character ...
... the Journal of Organometallic Chemistry write “Papers dealing with those aspects of organophosphorous and organoselenium chemistry that are of interest to the organometallic chemist will also be considered.” In most organometallic compounds, the metal-carbon bond has predominantly covalent character ...
No Slide Title - MDC Faculty Home Pages
... Write the atomic symbols for atoms with the following subatomic particles: A. 8 protons 8 neutrons 8 electrons B. 17 protons 20 neutrons 17 electrons C. 47 protons 60 neutrons 47 electrons ...
... Write the atomic symbols for atoms with the following subatomic particles: A. 8 protons 8 neutrons 8 electrons B. 17 protons 20 neutrons 17 electrons C. 47 protons 60 neutrons 47 electrons ...
Chapter 2: Atoms, Ions, and the Periodic Table
... A) Pure water is composed of the elements oxygen and hydrogen in a mass ratio of 8 to 1. B) Any sample of a given compound always contains the same proportions by mass of the component elements. C) The mass of the products of a chemical reaction is equal to the mass of the starting materials of the ...
... A) Pure water is composed of the elements oxygen and hydrogen in a mass ratio of 8 to 1. B) Any sample of a given compound always contains the same proportions by mass of the component elements. C) The mass of the products of a chemical reaction is equal to the mass of the starting materials of the ...
Chapter 2: Atoms, Ions, and the Periodic Table
... A) Pure water is composed of the elements oxygen and hydrogen in a mass ratio of 8 to 1. B) Any sample of a given compound always contains the same proportions by mass of the component elements. C) The mass of the products of a chemical reaction is equal to the mass of the starting materials of the ...
... A) Pure water is composed of the elements oxygen and hydrogen in a mass ratio of 8 to 1. B) Any sample of a given compound always contains the same proportions by mass of the component elements. C) The mass of the products of a chemical reaction is equal to the mass of the starting materials of the ...
1.9 M - Thierry Karsenti
... 2. Atom: the smallest particle of an element that retains the identify and properties of the element and can take part in a chemical change. 3. Atomic number (symbol Z): the number of protons in the nucleus of each atom. 4. Compound: a substance that is formed when two or more elements combine chemi ...
... 2. Atom: the smallest particle of an element that retains the identify and properties of the element and can take part in a chemical change. 3. Atomic number (symbol Z): the number of protons in the nucleus of each atom. 4. Compound: a substance that is formed when two or more elements combine chemi ...
Chapter 2 – Atoms, Ions, and the Periodic Table
... number of neutrons is 46 (N = A – Z). Since the charge is 1–, there must be 36 electrons. (b) The number of protons is 38, so the element is Sr. The mass number, 88, is the sum of the protons and neutrons. Since there are two more protons than electrons, the ion has a charge of 2+. (c) The number of ...
... number of neutrons is 46 (N = A – Z). Since the charge is 1–, there must be 36 electrons. (b) The number of protons is 38, so the element is Sr. The mass number, 88, is the sum of the protons and neutrons. Since there are two more protons than electrons, the ion has a charge of 2+. (c) The number of ...
Preview Sample 1
... 8) The nucleus of a nitrogen atom contains 7 neutrons and 7 protons. Which of the following is a correct statement concerning nitrogen? A) The nitrogen atom has a mass number of approximately 7 daltons and an atomic mass of 14. B) The nitrogen atom has a mass number of approximately 14 daltons and a ...
... 8) The nucleus of a nitrogen atom contains 7 neutrons and 7 protons. Which of the following is a correct statement concerning nitrogen? A) The nitrogen atom has a mass number of approximately 7 daltons and an atomic mass of 14. B) The nitrogen atom has a mass number of approximately 14 daltons and a ...
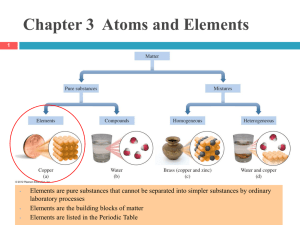
Chapter 4 Elements and the Periodic Table
... neutrons, and negatively charged electrons. Protons and neutrons are about equal in mass. An electron has about 1/2,000 the mass of a proton or neutron. ...
... neutrons, and negatively charged electrons. Protons and neutrons are about equal in mass. An electron has about 1/2,000 the mass of a proton or neutron. ...
Chapter 4 Elements and the Periodic Table
... neutrons, and negatively charged electrons. Protons and neutrons are about equal in mass. An electron has about 1/2,000 the mass of a proton or neutron. ...
... neutrons, and negatively charged electrons. Protons and neutrons are about equal in mass. An electron has about 1/2,000 the mass of a proton or neutron. ...
FREE Sample Here
... elements can be generated depending on the masses used. e. When two different elements combine to form a mixture, they do so in definite proportions by weight. Answer: c Section 0.4 Difficulty Level: medium ...
... elements can be generated depending on the masses used. e. When two different elements combine to form a mixture, they do so in definite proportions by weight. Answer: c Section 0.4 Difficulty Level: medium ...
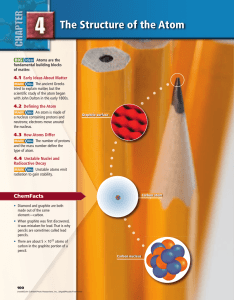
Chapter 4: The Structure of the Atom
... Many experiments since Dalton’s time have proven that atoms do exist. So what exactly is the definition of an atom? To answer this question, consider a gold ring. Suppose you decide to grind the ring down into a pile of gold dust. Each fragment of gold dust still retains all of the properties of gol ...
... Many experiments since Dalton’s time have proven that atoms do exist. So what exactly is the definition of an atom? To answer this question, consider a gold ring. Suppose you decide to grind the ring down into a pile of gold dust. Each fragment of gold dust still retains all of the properties of gol ...
FREE Sample Here
... The atomic mass of naturally occurring cobalt, which exists in nature as a single isotope, is listed as 58.9332 u. This means that a. all cobalt atoms found in nature have a mass which is 58.9332/12.000 times as great as that of a 12C atom. b. all cobalt atoms found in nature have a mass which is 58 ...
... The atomic mass of naturally occurring cobalt, which exists in nature as a single isotope, is listed as 58.9332 u. This means that a. all cobalt atoms found in nature have a mass which is 58.9332/12.000 times as great as that of a 12C atom. b. all cobalt atoms found in nature have a mass which is 58 ...
File
... The above diagram shows a cathode ray being deflected by an electric field. Which plate is positively charged? a. A c. Neither one is charged b. B Who was the man who lived from 460B.C.–370B.C. and was among the first to suggest the idea of atoms? a. Atomos c. Democritus b. Dalton d. Thomson The sma ...
... The above diagram shows a cathode ray being deflected by an electric field. Which plate is positively charged? a. A c. Neither one is charged b. B Who was the man who lived from 460B.C.–370B.C. and was among the first to suggest the idea of atoms? a. Atomos c. Democritus b. Dalton d. Thomson The sma ...
Chapter 0 A Very Brief History of Chemistry Multiple Choice Questions
... a. The nebula that formed the earth had elements that were not evenly distributed. b. Winds on the surface of the earth have moved around the heavy and light elements into bands. c. The early earth liquefied, resulting in heavier elements migrating towards the core, and lighter elements towards the ...
... a. The nebula that formed the earth had elements that were not evenly distributed. b. Winds on the surface of the earth have moved around the heavy and light elements into bands. c. The early earth liquefied, resulting in heavier elements migrating towards the core, and lighter elements towards the ...
The Atomic Theory
... Dalton’s Atomic Theory Let’s consider a simple but important experiment that suggested matter might be made up of atoms. In the late 1700s and early 1800s, scientists began noticing that when certain substances, like hydrogen and oxygen, were combined to produce a new substance, the reactants (hydro ...
... Dalton’s Atomic Theory Let’s consider a simple but important experiment that suggested matter might be made up of atoms. In the late 1700s and early 1800s, scientists began noticing that when certain substances, like hydrogen and oxygen, were combined to produce a new substance, the reactants (hydro ...
Atomic Theory and Periodic Table Review Multiple Choice Identify
... Complete each sentence or statement. 33. John Dalton concluded that all the atoms of a single ____________________ have the same mass. 34. Neutrons and ____________________ have almost the same mass. 35. The nuclei of isotopes contain different numbers of ____________________. 36. The ______________ ...
... Complete each sentence or statement. 33. John Dalton concluded that all the atoms of a single ____________________ have the same mass. 34. Neutrons and ____________________ have almost the same mass. 35. The nuclei of isotopes contain different numbers of ____________________. 36. The ______________ ...