Electronic Structure
... Size of Atoms (atomic radius)- atomic size of atoms is measured in terms of bonding atomic radius. The bonding atomic radius- radius of the atom when it is bonded to another atom- is shorter than the nonbonding atomic radius. Think of an atom as a sphere. Atomic size tends to decrease as one goes fr ...
... Size of Atoms (atomic radius)- atomic size of atoms is measured in terms of bonding atomic radius. The bonding atomic radius- radius of the atom when it is bonded to another atom- is shorter than the nonbonding atomic radius. Think of an atom as a sphere. Atomic size tends to decrease as one goes fr ...
Script of atoms video
... For uncharged (electrically neutral) atoms: # of protons = # of electrons Re-cap Protons and neutrons are in the nucleus Their combined mass gives the mass of the atom Electrons “orbit” the nucleus Organized in energy shells Electron shells figure 2.5 e- are negatively charged -> attracted to nucleu ...
... For uncharged (electrically neutral) atoms: # of protons = # of electrons Re-cap Protons and neutrons are in the nucleus Their combined mass gives the mass of the atom Electrons “orbit” the nucleus Organized in energy shells Electron shells figure 2.5 e- are negatively charged -> attracted to nucleu ...
Atomic Structure
... Energy levels can be imagined like orbits Electrons can jump from orbit to orbit by either absorbing or emitting a specific energy photon This photon would have a specific energy and specific color, if ...
... Energy levels can be imagined like orbits Electrons can jump from orbit to orbit by either absorbing or emitting a specific energy photon This photon would have a specific energy and specific color, if ...
Chapter 2 Outline 3rd PERIOD
... Elements are unique substances that cannot be broken down into simpler substances by ordinary chemical methods Examples: oxygen, carbon, gold, copper, and iron Carbon, oxygen, hydrogen and nitrogen make up 96% of body weight All elements are shown on the periodic table Atoms are the building blocks ...
... Elements are unique substances that cannot be broken down into simpler substances by ordinary chemical methods Examples: oxygen, carbon, gold, copper, and iron Carbon, oxygen, hydrogen and nitrogen make up 96% of body weight All elements are shown on the periodic table Atoms are the building blocks ...
Unit 1 Atoms and Periodic Table Intro Periodic Table Notes
... Measured in AMU’s –Atomic Mass Unit • Protons plus neutrons in nucleus • Calculate number of neutrons by subtracting the atomic number from the atomic mass ...
... Measured in AMU’s –Atomic Mass Unit • Protons plus neutrons in nucleus • Calculate number of neutrons by subtracting the atomic number from the atomic mass ...
Chapter 4 Notes
... Development of Atomic Theory 1. Democritus named atoms from the Greek word that means ____________________. 2. During Aristotle’s it was believed that all substances were built from the four elements ________________, air, _____________________ and water. 3. By the 1800s Dalton’s ideas replaced the ...
... Development of Atomic Theory 1. Democritus named atoms from the Greek word that means ____________________. 2. During Aristotle’s it was believed that all substances were built from the four elements ________________, air, _____________________ and water. 3. By the 1800s Dalton’s ideas replaced the ...
Chapter 1 Notes: The Science of Chemistry
... o Number of waves (cycles) which pass a given point in one second Electron Configuration in the Atom Ground state- electrons in their lowest possible energy level around the atom (normal) Excited state- electrons that have absorbed energy and jumped to a higher energy level (will eventually drop ...
... o Number of waves (cycles) which pass a given point in one second Electron Configuration in the Atom Ground state- electrons in their lowest possible energy level around the atom (normal) Excited state- electrons that have absorbed energy and jumped to a higher energy level (will eventually drop ...
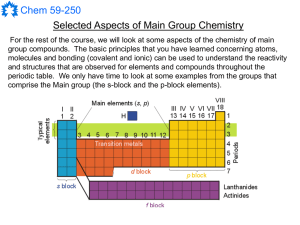
Main Group Notes 1
... Much of the important chemistry of the alkali and alkaline earth metals can be understood on the basis of their low ionization enthalpies (or electronegativities) and the favourability of ionic bonding. ...
... Much of the important chemistry of the alkali and alkaline earth metals can be understood on the basis of their low ionization enthalpies (or electronegativities) and the favourability of ionic bonding. ...
Notes on Atomic Theory
... All mass is made of atoms Atoms of same element have the same size, mass, and properties Atoms can’t be subdivided, created or destroyed Atoms of different element combine in whole number ratios to make compounds In chemical reactions, atoms are combined, separated, and rearranged. ...
... All mass is made of atoms Atoms of same element have the same size, mass, and properties Atoms can’t be subdivided, created or destroyed Atoms of different element combine in whole number ratios to make compounds In chemical reactions, atoms are combined, separated, and rearranged. ...
Unit 2 Notes Atomic
... amounts of _____________. ! The further from the nucleus, the more __________ an electron has. ...
... amounts of _____________. ! The further from the nucleus, the more __________ an electron has. ...
Atomic number
... decay and thereby lose energy. Why would nucleii tend to fall apart?? (Think about what protons do to each other) These unstable elements are called RADIOACTIVE. All elements with more than 83 protons are RADIOACTIVE. ...
... decay and thereby lose energy. Why would nucleii tend to fall apart?? (Think about what protons do to each other) These unstable elements are called RADIOACTIVE. All elements with more than 83 protons are RADIOACTIVE. ...
Chem 152 Chapter 4
... • A sample of an element contains only one type of atom. • There are over 100 known elements. ...
... • A sample of an element contains only one type of atom. • There are over 100 known elements. ...
John Dalton Dmitri Mendeleev JJ Thomson Ernest Rutherford
... Activity 2: What the scientists did. Cut each of the scientist rectangles on the 2nd sheet of paper Spread them out across your work table. These are going to be your headings. Next, cut each of the “things they did” descriptions. You are going to research each description and determine ...
... Activity 2: What the scientists did. Cut each of the scientist rectangles on the 2nd sheet of paper Spread them out across your work table. These are going to be your headings. Next, cut each of the “things they did” descriptions. You are going to research each description and determine ...
Atomic Structure
... He used a cathode ray tube to discover electrons. Sec. 1 - Atoms John Dalton and Democritus agreed that all elements are composed of _____________ particles called atoms. Unlike Democritus, Dalton performed ________________ to test and correct his theory. 100 000 000 copper atoms lined up would form ...
... He used a cathode ray tube to discover electrons. Sec. 1 - Atoms John Dalton and Democritus agreed that all elements are composed of _____________ particles called atoms. Unlike Democritus, Dalton performed ________________ to test and correct his theory. 100 000 000 copper atoms lined up would form ...
A quick summary about atoms: Atomic masses and/or hydrogen
... In 1905 Albert Einstein proved that atoms must exist (he showed that Brownian Motion would look different if there weren't really atoms and molecules shoving particles around). This later won the Nobel Prize for Jean Perrin in 1926. ...
... In 1905 Albert Einstein proved that atoms must exist (he showed that Brownian Motion would look different if there weren't really atoms and molecules shoving particles around). This later won the Nobel Prize for Jean Perrin in 1926. ...
Worksheet - Chapter 3A - Atomic Structure 2012 Atomic Theory
... He used a cathode ray tube to discover electrons. Sec. 1 - Atoms John Dalton and Democritus agreed that all elements are composed of _____________ particles called atoms. Unlike Democritus, Dalton performed ________________ to test and correct his theory. 100 000 000 copper atoms lined up would form ...
... He used a cathode ray tube to discover electrons. Sec. 1 - Atoms John Dalton and Democritus agreed that all elements are composed of _____________ particles called atoms. Unlike Democritus, Dalton performed ________________ to test and correct his theory. 100 000 000 copper atoms lined up would form ...
Periodic table
The periodic table is a tabular arrangement of the chemical elements, ordered by their atomic number (number of protons in the nucleus), electron configurations, and recurring chemical properties. The table also shows four rectangular blocks: s-, p- d- and f-block. In general, within one row (period) the elements are metals on the lefthand side, and non-metals on the righthand side.The rows of the table are called periods; the columns are called groups. Six groups (columns) have names as well as numbers: for example, group 17 elements are the halogens; and group 18, the noble gases. The periodic table can be used to derive relationships between the properties of the elements, and predict the properties of new elements yet to be discovered or synthesized. The periodic table provides a useful framework for analyzing chemical behavior, and is widely used in chemistry and other sciences.Although precursors exist, Dmitri Mendeleev is generally credited with the publication, in 1869, of the first widely recognized periodic table. He developed his table to illustrate periodic trends in the properties of the then-known elements. Mendeleev also predicted some properties of then-unknown elements that would be expected to fill gaps in this table. Most of his predictions were proved correct when the elements in question were subsequently discovered. Mendeleev's periodic table has since been expanded and refined with the discovery or synthesis of further new elements and the development of new theoretical models to explain chemical behavior.All elements from atomic numbers 1 (hydrogen) to 118 (ununoctium) have been discovered or reportedly synthesized, with elements 113, 115, 117, and 118 having yet to be confirmed. The first 94 elements exist naturally, although some are found only in trace amounts and were synthesized in laboratories before being found in nature. Elements with atomic numbers from 95 to 118 have only been synthesized in laboratories. It has been shown that einsteinium and fermium once occurred in nature but currently do not. Synthesis of elements having higher atomic numbers is being pursued. Numerous synthetic radionuclides of naturally occurring elements have also been produced in laboratories.