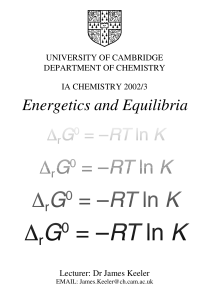do physics online space gravitational potential energy
... Hence, one can predict the velocity v of the object when it has fallen a distance h starting with an initial velocity u. Equation (2) and (3) are only applicable near the Earth’s surface where the value of the gravitational field strength (acceleration due to gravity) g is a constant. To find a mor ...
... Hence, one can predict the velocity v of the object when it has fallen a distance h starting with an initial velocity u. Equation (2) and (3) are only applicable near the Earth’s surface where the value of the gravitational field strength (acceleration due to gravity) g is a constant. To find a mor ...
Chapter4.Presentation.ICAM.Work,Power_and_Energy
... done during each, and adding them up. As the pieces become very narrow, the work done is the area under the force vs. distance curve. ...
... done during each, and adding them up. As the pieces become very narrow, the work done is the area under the force vs. distance curve. ...
Chapter 6 Work and Kinetic Energy
... Step 2: Free body diagram. There are two forces acting on the scale one upward due to gravity and one downward due to spring tension. Step 3: Apply Newton’s Laws / Work-energy theorem. From Third Law the force of woman acting on the spring and force of spring acting on woman are related ⃗ w on s = − ...
... Step 2: Free body diagram. There are two forces acting on the scale one upward due to gravity and one downward due to spring tension. Step 3: Apply Newton’s Laws / Work-energy theorem. From Third Law the force of woman acting on the spring and force of spring acting on woman are related ⃗ w on s = − ...
Work and Energy - University of Colorado Boulder
... This is called “The First Law of Thermodynamics”. Aside: Actually the First Law of Thermodynamics is this: “heat added plus work done equals change in energy” or Q + W = U . (Q is the symbol for heat). In this chapter we won’t consider adding heat to a system (like holding a flame under it), so Q = ...
... This is called “The First Law of Thermodynamics”. Aside: Actually the First Law of Thermodynamics is this: “heat added plus work done equals change in energy” or Q + W = U . (Q is the symbol for heat). In this chapter we won’t consider adding heat to a system (like holding a flame under it), so Q = ...
Energy, work, heat and chemical reactions
... total energy before reaction = total energy after reaction Reactions that absorb energy: Total bond energy of reactants ...
... total energy before reaction = total energy after reaction Reactions that absorb energy: Total bond energy of reactants ...
Seeing Energy in Everything
... They have developed telescopes with highly accurate polished lenses. 8. Draw a picture of light on a wall, what happens to the energy as it travels farther out from the center? As the energy travels outward its energy decreases. ...
... They have developed telescopes with highly accurate polished lenses. 8. Draw a picture of light on a wall, what happens to the energy as it travels farther out from the center? As the energy travels outward its energy decreases. ...
AH + B(-) A(
... AH + H2O A(-) + H3O(+) o o has an equilibrium constant associated with it Keq = [A][H3O]/[AH][H2O] o The terms in this equation are concentration. The molarity of water, [H2O] is 55 M or 55 moles/Liter o The acidity constant is ...
... AH + H2O A(-) + H3O(+) o o has an equilibrium constant associated with it Keq = [A][H3O]/[AH][H2O] o The terms in this equation are concentration. The molarity of water, [H2O] is 55 M or 55 moles/Liter o The acidity constant is ...
work
... Problem: A single conservative force of F = (3i + 5j) N acts on a 4.0 kg particle. Calculate the work done if the particle if the moves from the origin to r = (2i - 3j) m. Does the result depend on path? ...
... Problem: A single conservative force of F = (3i + 5j) N acts on a 4.0 kg particle. Calculate the work done if the particle if the moves from the origin to r = (2i - 3j) m. Does the result depend on path? ...