CHEMISTRY SEC 06 SYLLABUS
... The examination will consist of two written papers, each of two hours’ duration, and an assessment of practical work in chemistry. Questions will be set in English and must be answered in English. The examination will be structured as follows: Paper I consists of a written paper and a practical comp ...
... The examination will consist of two written papers, each of two hours’ duration, and an assessment of practical work in chemistry. Questions will be set in English and must be answered in English. The examination will be structured as follows: Paper I consists of a written paper and a practical comp ...
O A
... AgCl Electrode. The myoglobin showed quasireversible electrochemical behavior with a formal potential of -48 mV (vs Ag/AgCl), cathodic and anodic peaks were not observed using the bare graphite electrode. This shows that NiO-NPs acts as a facilitator of electron transfer from the redox species of my ...
... AgCl Electrode. The myoglobin showed quasireversible electrochemical behavior with a formal potential of -48 mV (vs Ag/AgCl), cathodic and anodic peaks were not observed using the bare graphite electrode. This shows that NiO-NPs acts as a facilitator of electron transfer from the redox species of my ...
Protein structure and function
... tissues their form and structural strength. The collagen superfamily of proteins includes more than 25 collagen types, and these types distributed in the tissues of the body according to their function. The most common collagen type is type I. ...
... tissues their form and structural strength. The collagen superfamily of proteins includes more than 25 collagen types, and these types distributed in the tissues of the body according to their function. The most common collagen type is type I. ...
Chemistry IGCSE
... expand. Until at some point they have enough energy to break the forces of attraction between them and the lattice turning into liquid. If you keep heating the liquid, particles will gain even more kinetic energy and start moving even faster, pushing each other away. The particles at the surface hav ...
... expand. Until at some point they have enough energy to break the forces of attraction between them and the lattice turning into liquid. If you keep heating the liquid, particles will gain even more kinetic energy and start moving even faster, pushing each other away. The particles at the surface hav ...
porphyrine, heme and..
... Heme itself, by feedback inhibition. Glucose and steroids. b- It is stimulated by : Certain drugs as phenobarbital and iron. ...
... Heme itself, by feedback inhibition. Glucose and steroids. b- It is stimulated by : Certain drugs as phenobarbital and iron. ...
BI0 120 cell and tissues
... A. adding phosphates, modifying sugars and forming glyceraldehyde-3-phosphate. B. oxidative steps, proton pumping, and reaction with oxygen. C. oxidation of glyceraldehyde-3-phosphate, and storage of energy. D. ATP synthesis by substrate-level phosphorylation, and formation of pyruvate. E. oxidation ...
... A. adding phosphates, modifying sugars and forming glyceraldehyde-3-phosphate. B. oxidative steps, proton pumping, and reaction with oxygen. C. oxidation of glyceraldehyde-3-phosphate, and storage of energy. D. ATP synthesis by substrate-level phosphorylation, and formation of pyruvate. E. oxidation ...
Sulfur Part II: Sulfur and Sulfur Compounds in the Human Body
... ** T – indicates high level of Thiol content, I – indicates high level of Isothiocyanate content. From the list above the most important source of organosulfur compounds in the human diet is from proteins. The organosulfur compounds found in protein come from the sulfur containing amino acids methio ...
... ** T – indicates high level of Thiol content, I – indicates high level of Isothiocyanate content. From the list above the most important source of organosulfur compounds in the human diet is from proteins. The organosulfur compounds found in protein come from the sulfur containing amino acids methio ...
RESPIRATION AND ELIMINATION OF NITROGENOUS WASTES
... (i) Breathing or pulmonary ventilation leading to exchange of oxygen and carbon dioxide between the atmospheric air and the lungs. (ii) Exchange of gases at the alveolar surface. (iii) Transport and exchange of gases in the tissues. (iv) Cellular respiration. 14.2.1 Breathing or pulmonary ventilatio ...
... (i) Breathing or pulmonary ventilation leading to exchange of oxygen and carbon dioxide between the atmospheric air and the lungs. (ii) Exchange of gases at the alveolar surface. (iii) Transport and exchange of gases in the tissues. (iv) Cellular respiration. 14.2.1 Breathing or pulmonary ventilatio ...
R Is for Arginine Metabolism of Arginine Takes off Again, in New
... concentrations may be limiting for NOS in specific subcellular locales. Moreover, the presence of endogenous competitive inhibitors of NOS (discussed below) may raise the effective intracellular arginine concentration needed for full enzyme activity. In any event, the action of NOS on l-arginine yie ...
... concentrations may be limiting for NOS in specific subcellular locales. Moreover, the presence of endogenous competitive inhibitors of NOS (discussed below) may raise the effective intracellular arginine concentration needed for full enzyme activity. In any event, the action of NOS on l-arginine yie ...
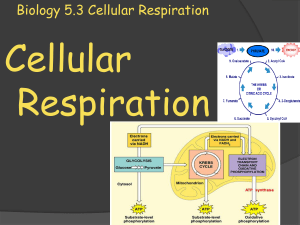
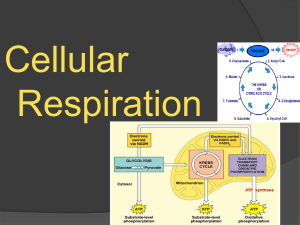
Biology 5.3 Cellular Respiration
... pyruvate during glycolysis. Glycolysis is an anaerobic process (no oxygen required), and it results in a gain of two ATP molecules. In the second stage of cellular respiration, the pyruvate passes through either aerobic respiration (requires oxygen) or fermentation. When oxygen is not present, ferme ...
... pyruvate during glycolysis. Glycolysis is an anaerobic process (no oxygen required), and it results in a gain of two ATP molecules. In the second stage of cellular respiration, the pyruvate passes through either aerobic respiration (requires oxygen) or fermentation. When oxygen is not present, ferme ...
R Is for Arginine
... concentrations may be limiting for NOS in specific subcellular locales. Moreover, the presence of endogenous competitive inhibitors of NOS (discussed below) may raise the effective intracellular arginine concentration needed for full enzyme activity. In any event, the action of NOS on l-arginine yie ...
... concentrations may be limiting for NOS in specific subcellular locales. Moreover, the presence of endogenous competitive inhibitors of NOS (discussed below) may raise the effective intracellular arginine concentration needed for full enzyme activity. In any event, the action of NOS on l-arginine yie ...
19. ch 18(361-383) RESPIRATION
... (sur-FAK-tant), a substance that reduces the surface tenAlveoli sion (“pull”) of the fluids that line the alveoli. This surface action prevents collapse of the alveoli and eases expansion of the lungs. There are about 300 million alveoli in the human lungs. The resulting surface area in contact with ...
... (sur-FAK-tant), a substance that reduces the surface tenAlveoli sion (“pull”) of the fluids that line the alveoli. This surface action prevents collapse of the alveoli and eases expansion of the lungs. There are about 300 million alveoli in the human lungs. The resulting surface area in contact with ...
Blood loss
... exchange of the anions HCO3‐ (bicarb) and Cl‐ between RBC and plasma. Exchange allows max. transport of CO2 in blood (as bicarb in solution (a) Basic point: Bicarb is much more soluble in plasma than CO2, so lots of bicarb (but not much CO2) can be carried in the blood. Therefore need to covert ...
... exchange of the anions HCO3‐ (bicarb) and Cl‐ between RBC and plasma. Exchange allows max. transport of CO2 in blood (as bicarb in solution (a) Basic point: Bicarb is much more soluble in plasma than CO2, so lots of bicarb (but not much CO2) can be carried in the blood. Therefore need to covert ...
Chemistry Test Ch 11 Stoichiometry
... If 18.5 grams of Fe2(SO4)3 are actually made what is the percent yield? 5. Use the following equation answer these questions: C12H22O11 + 12O2 ---> 12CO2 + 11H2O A. If 3.45 g CO2 is produced how many water molecules are also produced? B. If there are 10.0 g of C12H22O11 and 10.0 g of oxygen reacting ...
... If 18.5 grams of Fe2(SO4)3 are actually made what is the percent yield? 5. Use the following equation answer these questions: C12H22O11 + 12O2 ---> 12CO2 + 11H2O A. If 3.45 g CO2 is produced how many water molecules are also produced? B. If there are 10.0 g of C12H22O11 and 10.0 g of oxygen reacting ...
Antioxidant and Prooxidant Activities of
... other foreign molecules (Bastian and Hibbs, 1994). The important roles of NO in neurotransmission and regulation of blood pressure have been also well established (Ignarro, 1991; Prast and Philippu, 2001). Recent evidence obtained from nonphagocytic cells suggests that several cytokines, growth fact ...
... other foreign molecules (Bastian and Hibbs, 1994). The important roles of NO in neurotransmission and regulation of blood pressure have been also well established (Ignarro, 1991; Prast and Philippu, 2001). Recent evidence obtained from nonphagocytic cells suggests that several cytokines, growth fact ...
1. Introduction
... The synthesis and deposition of the sensing layer is obviously the most crucial part in the preparation of gas sensors. Three main groups can be distinguished: powder/slurry deposition, chemical vapour deposition (CVD) and physical vapour deposition (PVD) [8,14] (Table 2). The main difference betwee ...
... The synthesis and deposition of the sensing layer is obviously the most crucial part in the preparation of gas sensors. Three main groups can be distinguished: powder/slurry deposition, chemical vapour deposition (CVD) and physical vapour deposition (PVD) [8,14] (Table 2). The main difference betwee ...
Gas Stoichiometry
... Example of a Gas Stoichiometry Problem Airbags in automobiles contain sodium azide (NaN3), potassium nitrate, and silicon dioxide. (All are solids.) 1. Upon impact, the bag is inflated by the thermal decomposition of sodium azide (NaN3) to sodium metal and nitrogen gas. 2. Because sodium is toxic a ...
... Example of a Gas Stoichiometry Problem Airbags in automobiles contain sodium azide (NaN3), potassium nitrate, and silicon dioxide. (All are solids.) 1. Upon impact, the bag is inflated by the thermal decomposition of sodium azide (NaN3) to sodium metal and nitrogen gas. 2. Because sodium is toxic a ...
Appendices - Mattson Creighton
... 3. It is actually the syringe containing the gas that must be washed in order to remove all traces of chemicals that were used to make the gas. After that has happened, the syringe contains our desired gas (CO2), a little bit of air and a few droplets of water. 4. Use only the specified amounts of r ...
... 3. It is actually the syringe containing the gas that must be washed in order to remove all traces of chemicals that were used to make the gas. After that has happened, the syringe contains our desired gas (CO2), a little bit of air and a few droplets of water. 4. Use only the specified amounts of r ...
Biology 5.3 Cellular Respiration - Chemistry
... lactic acid. For example, during exercise, pyruvate in muscles is converted to lactate when muscles must operate without enough oxygen. ...
... lactic acid. For example, during exercise, pyruvate in muscles is converted to lactate when muscles must operate without enough oxygen. ...
Beneficial effects of L-arginine on reducing obesity
... primarily in white adipose tissue (WAT). Although obesity is recognized as a leading risk factor for insulin resistance, Type-2 diabetes, atherosclerosis, stroke, hypertension, and some types of cancer (including colon and breast cancers), there are few medications for treating this chronic disease ...
... primarily in white adipose tissue (WAT). Although obesity is recognized as a leading risk factor for insulin resistance, Type-2 diabetes, atherosclerosis, stroke, hypertension, and some types of cancer (including colon and breast cancers), there are few medications for treating this chronic disease ...
Respiration
... Aquatic respiratory organs increase the diffusion surface area by extensions of tissue, called gills, that project out into the water. Gills can be simple, as in the papulae of echinoderms (see figure 53.2c), or complex, as in the highly convoluted gills of fish (see figure 53.2e). The great increas ...
... Aquatic respiratory organs increase the diffusion surface area by extensions of tissue, called gills, that project out into the water. Gills can be simple, as in the papulae of echinoderms (see figure 53.2c), or complex, as in the highly convoluted gills of fish (see figure 53.2e). The great increas ...
File
... ending and replace with “ide.” For example: H2S is hydrogen sulfide. In this case, the amount of each element doesn’t affect the name of the compound. to name the following examples: ...
... ending and replace with “ide.” For example: H2S is hydrogen sulfide. In this case, the amount of each element doesn’t affect the name of the compound. to name the following examples: ...