Word Equations • a summary
... The general formula is an element reacting with a compound to produce a new element and a new compound. A metal (cation) can displace another metal (cation) or hydrogen: X + YZ Y + XZ eg. 1 Mg + ZnCl2 Zn + MgCl2 eg. 2 Mg + H2SO4 H2 + MgSO4 Try this…predict the product of this single displa ...
... The general formula is an element reacting with a compound to produce a new element and a new compound. A metal (cation) can displace another metal (cation) or hydrogen: X + YZ Y + XZ eg. 1 Mg + ZnCl2 Zn + MgCl2 eg. 2 Mg + H2SO4 H2 + MgSO4 Try this…predict the product of this single displa ...
last year`s April exam
... b) Draw a picture that shows how water molecules can interact with an aldehyde through H-bonding, showing all possible interactions. ...
... b) Draw a picture that shows how water molecules can interact with an aldehyde through H-bonding, showing all possible interactions. ...
Organic and Biological Molecules
... Optical Isomerism A molecule that exhibits optical isomerism is called chiral. Most biological molecules are chiral, often with one isomer performing important biological functions while the other may be biologically inactive. Although inorganic molecules can also be chiral, the chirality of organi ...
... Optical Isomerism A molecule that exhibits optical isomerism is called chiral. Most biological molecules are chiral, often with one isomer performing important biological functions while the other may be biologically inactive. Although inorganic molecules can also be chiral, the chirality of organi ...
Kwang-Ting Liu Department of Chemistry National Taiwan
... major in agricultural chemistry the latest edition of several popular ones were browsed. • In most books little attention was paid to the topics related to sustainable development, except ozone depletion and greenhouse gases. • The one by Solomons and Fryhle has discussion on the prohibition of usin ...
... major in agricultural chemistry the latest edition of several popular ones were browsed. • In most books little attention was paid to the topics related to sustainable development, except ozone depletion and greenhouse gases. • The one by Solomons and Fryhle has discussion on the prohibition of usin ...
Isomers
... Apart from their optical activity enantiomers generally have similar physical and chemical properties. The chemical properties may be significantly different when the enantiomers interact with other optically active compounds. Thalidomide has two optical isomers. One is a tranquilizer, the other is ...
... Apart from their optical activity enantiomers generally have similar physical and chemical properties. The chemical properties may be significantly different when the enantiomers interact with other optically active compounds. Thalidomide has two optical isomers. One is a tranquilizer, the other is ...
LOYOLA COLLEGE (AUTONOMOUS), CHENNAI – 600 034
... 5. Explain the change in the geometry of excited state molecule in a photochemical process and the variation in its physical property. 6. What is Norrish type I and II cleavage reactions? Give suitable examples. 7. What are the important guidelines to be followed while choosing alternate synthetic r ...
... 5. Explain the change in the geometry of excited state molecule in a photochemical process and the variation in its physical property. 6. What is Norrish type I and II cleavage reactions? Give suitable examples. 7. What are the important guidelines to be followed while choosing alternate synthetic r ...
Unit 8 Powerpoint
... 4. Balance the elements one at a time by using coefficients. Begin by balancing elements that appear only once on each side of the equation. Unwritten coefficients are assumed to be 1 Once you are certain you have the correct chemical ...
... 4. Balance the elements one at a time by using coefficients. Begin by balancing elements that appear only once on each side of the equation. Unwritten coefficients are assumed to be 1 Once you are certain you have the correct chemical ...
one
... • Step 2 – change one or more coefficients until the equation is balanced. – Start by balancing an element that appears in only one reactant and product. – Once one element is balanced, proceed to balance another, and another, until all elements are balanced. – Balance chemical formulas by placing c ...
... • Step 2 – change one or more coefficients until the equation is balanced. – Start by balancing an element that appears in only one reactant and product. – Once one element is balanced, proceed to balance another, and another, until all elements are balanced. – Balance chemical formulas by placing c ...
Key to Exam 3
... (50 pts) Draw the most likely product(s) to the right of the reactions and label them clearly A-J. Hints: A-B, and E-J have stereochemical issues; in some cases you have to evaluate which is the least hindered side of the molecules, in other cases cis versus trans etc. Pay attention to regiochemistr ...
... (50 pts) Draw the most likely product(s) to the right of the reactions and label them clearly A-J. Hints: A-B, and E-J have stereochemical issues; in some cases you have to evaluate which is the least hindered side of the molecules, in other cases cis versus trans etc. Pay attention to regiochemistr ...
Chapter 7 - Alkenes and Alkynes I less substituted alkene due to
... - Carrying out dehydrohalogenations with a bulky base, such as tert-butxide, favors the formation of the less substituted alkene due to sterics - When an elimination yields the less substituted alkene, it follows the Hofmann Rule - The anticoplanar transition state is preferred, but the syn coplanar ...
... - Carrying out dehydrohalogenations with a bulky base, such as tert-butxide, favors the formation of the less substituted alkene due to sterics - When an elimination yields the less substituted alkene, it follows the Hofmann Rule - The anticoplanar transition state is preferred, but the syn coplanar ...
Exam 1
... As mentioned in the text, diethyl ether, pentane, and 1-butanol have similar molar masses, but different physical properties. Boiling points are 35oC, 36oC, and 117oC, respectively. Their respective solubilities in water are 7.5g/100mL, insoluble, and 9g/100mL. (i) Draw structures for each of these ...
... As mentioned in the text, diethyl ether, pentane, and 1-butanol have similar molar masses, but different physical properties. Boiling points are 35oC, 36oC, and 117oC, respectively. Their respective solubilities in water are 7.5g/100mL, insoluble, and 9g/100mL. (i) Draw structures for each of these ...
Chapter 7 Alkenes and Alkynes I
... removing sterically hindered hydrogens and generally only react with more accessible hydrogens (e.g. primary hydrogens) ...
... removing sterically hindered hydrogens and generally only react with more accessible hydrogens (e.g. primary hydrogens) ...
Predicting Products online assistance #3
... 2. decomposition - one reactant decomposes, or breaks apart, into two or more products. 3. single replacement - an element replaces another in a compound. 4. double replacement - the elements in two compounds switch partners to form two new compounds. Writing Balanced Equations A chemical reaction i ...
... 2. decomposition - one reactant decomposes, or breaks apart, into two or more products. 3. single replacement - an element replaces another in a compound. 4. double replacement - the elements in two compounds switch partners to form two new compounds. Writing Balanced Equations A chemical reaction i ...
CHE-05 year 2004
... Answer sheets received after the due date shall not be accepted. We strongly suggest that you should retain a copy of your assignment. Wishing you all good luck. ...
... Answer sheets received after the due date shall not be accepted. We strongly suggest that you should retain a copy of your assignment. Wishing you all good luck. ...
Chemical Reactions
... (BrINClHOF) For example, Oxygen is O2 as an element. In a compound, it can’t be a diatomic element because it’s not an element anymore, it’s a compound! ...
... (BrINClHOF) For example, Oxygen is O2 as an element. In a compound, it can’t be a diatomic element because it’s not an element anymore, it’s a compound! ...
WM5 The synthesis of salicylic acid and aspirin
... What extra atoms need to be added to phenol? What conditions and reagents are needed? (research this); ...
... What extra atoms need to be added to phenol? What conditions and reagents are needed? (research this); ...
INTRODUCTION - Open Access Repository of Indian Theses
... 3. A Mild and Highly Efficient Synthesis of 3-Pyrrolyl-Indolinones and Pyrrolyl-Indeno[1,2-b]Quinoxalines using BiCl3 as a Catalyst An efficient synthesis of 3-pyrrolyl-indolinones and pyrrolyl-indeno[1,2-b] quinoxalines is described by the reaction of 4-hydroxyproline with isatin or indeno[1,2b]qui ...
... 3. A Mild and Highly Efficient Synthesis of 3-Pyrrolyl-Indolinones and Pyrrolyl-Indeno[1,2-b]Quinoxalines using BiCl3 as a Catalyst An efficient synthesis of 3-pyrrolyl-indolinones and pyrrolyl-indeno[1,2-b] quinoxalines is described by the reaction of 4-hydroxyproline with isatin or indeno[1,2b]qui ...
Catalytic asymmetric carbonyl addition reactions catalysed by group
... Catalytic asymmetric carbonyl addition reactions catalysed by group 10 metals The addition of a nucleophilic species to the carbonyl group is one of the most important methodology for carbon-carbon bond construction and various solutions have been offered to achieve an asymmetric version. ...
... Catalytic asymmetric carbonyl addition reactions catalysed by group 10 metals The addition of a nucleophilic species to the carbonyl group is one of the most important methodology for carbon-carbon bond construction and various solutions have been offered to achieve an asymmetric version. ...
Dess-Martin Oxidation
... • ^ Dess, D. B.; Martin, J. C. (1983). "Readily accessible 12-I-5 oxidant for the conversion of primary and secondary alcohols to aldehydes and ketones". J. Org. Chem. 48: 4155. doi:10.1021/jo00170a070. • Boeckman, R. J. In "Encyclopedia of Reagents for Organic Synthesis"; Paquette, L. A., Ed.; Wile ...
... • ^ Dess, D. B.; Martin, J. C. (1983). "Readily accessible 12-I-5 oxidant for the conversion of primary and secondary alcohols to aldehydes and ketones". J. Org. Chem. 48: 4155. doi:10.1021/jo00170a070. • Boeckman, R. J. In "Encyclopedia of Reagents for Organic Synthesis"; Paquette, L. A., Ed.; Wile ...
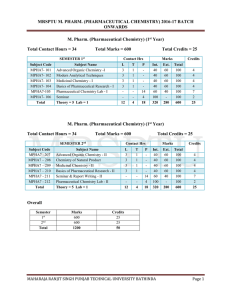
mrsptu m. pharm. (pharmaceutical chemistry) 2016
... John Wiley and Sons, New York. 2. L.G. Chatten, ‘Pharmaceutical Chemistry’, Vol. I & II, Marcel Dekker, New York. 3. W.D. James and H.T. Kenneth, ‘Analytical Chemistry by Open Learning: Thermal Methods. John Wiley and Sons, New York. 4. R.J. Abraham, J. Fisher and P. Bftus, ‘Introduction to NMR Spec ...
... John Wiley and Sons, New York. 2. L.G. Chatten, ‘Pharmaceutical Chemistry’, Vol. I & II, Marcel Dekker, New York. 3. W.D. James and H.T. Kenneth, ‘Analytical Chemistry by Open Learning: Thermal Methods. John Wiley and Sons, New York. 4. R.J. Abraham, J. Fisher and P. Bftus, ‘Introduction to NMR Spec ...
Enantioselective synthesis

Enantioselective synthesis, also called chiral synthesis or asymmetric synthesis, is defined by IUPAC as: a chemical reaction (or reaction sequence) in which one or more new elements of chirality are formed in a substrate molecule and which produces the stereoisomeric (enantiomeric or diastereoisomeric) products in unequal amounts.Put more simply: it is the synthesis of a compound by a method that favors the formation of a specific enantiomer or diastereomer.Enantioselective synthesis is a key process in modern chemistry and is particularly important in the field of pharmaceuticals, as the different enantiomers or diastereomers of a molecule often have different biological activity.