Coordination Compounds: Chemistry and Application
... Coordination compounds are metal ions surrounded by ligands. Ligands are either anions or molecules that can donate electrons into the d-orbitals of the metal ion and form a bond. Examples of common ligands are chloride ion, cyanide ion, ammonia, ethylenediamine, and ethylenediaminetetraacetate ion ...
... Coordination compounds are metal ions surrounded by ligands. Ligands are either anions or molecules that can donate electrons into the d-orbitals of the metal ion and form a bond. Examples of common ligands are chloride ion, cyanide ion, ammonia, ethylenediamine, and ethylenediaminetetraacetate ion ...
Highly electron-deficient neutral and cationic zirconium complexes
... polymerizes olefins, whereas with the η6-[PhCH2B(C6F5)3]- counterion the system is inactive. Chelating dianionic diamido ligands are successfully being used as ancillary ligands for group 4 metals. Both bidentate1 and tridentate2 (amido-amine/amido-ether) varieties are known, and both types of ligan ...
... polymerizes olefins, whereas with the η6-[PhCH2B(C6F5)3]- counterion the system is inactive. Chelating dianionic diamido ligands are successfully being used as ancillary ligands for group 4 metals. Both bidentate1 and tridentate2 (amido-amine/amido-ether) varieties are known, and both types of ligan ...
Synthesis and Properties of a New Kind of One
... nitride, as well. But in the proposed new structure there are not so many restrictions limiting the number of possible compounds with good conductivity. Because much more metal atoms prefer an octahedral coordination as there are suitable atoms with a ds configuration available. In addition a greate ...
... nitride, as well. But in the proposed new structure there are not so many restrictions limiting the number of possible compounds with good conductivity. Because much more metal atoms prefer an octahedral coordination as there are suitable atoms with a ds configuration available. In addition a greate ...
Complex Ions and Free Energy
... Transition Metal Complexes • When transition metals bond they are the central atom and they bond extensively with numerous substances. • The complexes that form are known as metal complexes • The molecule that bond with metals are known as ligands ▫ A ligand is always a Lewis Base ...
... Transition Metal Complexes • When transition metals bond they are the central atom and they bond extensively with numerous substances. • The complexes that form are known as metal complexes • The molecule that bond with metals are known as ligands ▫ A ligand is always a Lewis Base ...
Chapter 21. Transition Metals and Coordination Chemistry
... Central idea: Formation of hybride atomic orbitals that are used to share electron pairs to form s bond between atoms ...
... Central idea: Formation of hybride atomic orbitals that are used to share electron pairs to form s bond between atoms ...
Lecture`23 - MSU Chemistry - Michigan State University
... 3. be)able)to)predict)the)range)of)possible)oxidation)states)for)the)firstJrow)transition) metals)and)the)types)of)bonding)associated)with)the)various)oxidation)states.) 4. understand)what)a)coordination)compound)is)and)how)coordinate)covalent)bonds)are) ...
... 3. be)able)to)predict)the)range)of)possible)oxidation)states)for)the)firstJrow)transition) metals)and)the)types)of)bonding)associated)with)the)various)oxidation)states.) 4. understand)what)a)coordination)compound)is)and)how)coordinate)covalent)bonds)are) ...
Handout-9
... In this case the alkyl anion is the best donor ligand compared to the more electronegative and poorly donating Cl anion. Note that the alkyl ligand (-CH2CH=CH2) initially coordinated to the Re after the oxidative addition is an η1-allyl ligand and that it can convert to the generally more stable η3 ...
... In this case the alkyl anion is the best donor ligand compared to the more electronegative and poorly donating Cl anion. Note that the alkyl ligand (-CH2CH=CH2) initially coordinated to the Re after the oxidative addition is an η1-allyl ligand and that it can convert to the generally more stable η3 ...
Many metal ions (usually transition metals but including a few others
... Copper(II) chloride dissociates in aqueous solution to give the blue color of [Cu(H2O)6]2+ and yellow or red color of the halide complexes of the formula [CuCl2+x]x-. Concentrated solutions of CuCl2 appear green because of the combination of these various chromophores. The color of the dilute soluti ...
... Copper(II) chloride dissociates in aqueous solution to give the blue color of [Cu(H2O)6]2+ and yellow or red color of the halide complexes of the formula [CuCl2+x]x-. Concentrated solutions of CuCl2 appear green because of the combination of these various chromophores. The color of the dilute soluti ...
(Marine Bioinorganic Chemistry) 12.755 Lecture 2
... Solubility Products: Example for Fe(OH)3(s) Ksp= [Fe][OH]3 = 1042.7 Stability constants for metal complexes (where L is ligand, M is Metal): K = [ML]/[M][L] Ligands can include inorganic chemical species: In oxic systems: OH-, CO32-,SO42-, Cl-, PO43-, In anoxic systems add: HS-,, S2Ligands can also ...
... Solubility Products: Example for Fe(OH)3(s) Ksp= [Fe][OH]3 = 1042.7 Stability constants for metal complexes (where L is ligand, M is Metal): K = [ML]/[M][L] Ligands can include inorganic chemical species: In oxic systems: OH-, CO32-,SO42-, Cl-, PO43-, In anoxic systems add: HS-,, S2Ligands can also ...
click here for example introspection
... Metal complexes are so complicated, yet they are so interesting. So far in class we have looked at these complexes in a variety of ways, specifically from the view of acid/base and crystal field theory. They are extremely different and yet have an enormous overlap, the metals themselves. First and f ...
... Metal complexes are so complicated, yet they are so interesting. So far in class we have looked at these complexes in a variety of ways, specifically from the view of acid/base and crystal field theory. They are extremely different and yet have an enormous overlap, the metals themselves. First and f ...
Dinitrogen Cleavage by a Molybdenum(III) Complex
... The nitrogenase enzyme system catalyzes the ATP (adenosine triphosphate)-dependent reduction of dinitrogen to ammonia during the process of nitrogen fixation. Nitrogenase consists of two proteins: the iron (Fe)-protein, which couples hydrolysis of ATP to electron transfer, and the molybdenum-iron (M ...
... The nitrogenase enzyme system catalyzes the ATP (adenosine triphosphate)-dependent reduction of dinitrogen to ammonia during the process of nitrogen fixation. Nitrogenase consists of two proteins: the iron (Fe)-protein, which couples hydrolysis of ATP to electron transfer, and the molybdenum-iron (M ...
InorgCh15.1
... c) Many organometallic complexes with ligands weaker than CO don’t follow the 18 electron rule, thus parallels to the main group fail d) Loss of CO is much easier than loss of outer atoms from main group element ...
... c) Many organometallic complexes with ligands weaker than CO don’t follow the 18 electron rule, thus parallels to the main group fail d) Loss of CO is much easier than loss of outer atoms from main group element ...
Ch 22 Transition complexes
... 7. A complex ion is a charged species consisting of a metal ion surrounded by [A] hydrogen ions. [B] ligands. [C] ligands and counter ions. [D] other transition metals. [E] none of these 8. What is the electron configuration of the Mn(II) ion? [A] [Ar] 4s23d3 [B] [Ar] 3d5 [C] [Ar] 4s23d5 [D] [Ar] 4s ...
... 7. A complex ion is a charged species consisting of a metal ion surrounded by [A] hydrogen ions. [B] ligands. [C] ligands and counter ions. [D] other transition metals. [E] none of these 8. What is the electron configuration of the Mn(II) ion? [A] [Ar] 4s23d3 [B] [Ar] 3d5 [C] [Ar] 4s23d5 [D] [Ar] 4s ...
5_slides_olefin_complexes_VIPEr
... Created by Margaret L. Scheuermann, Princeton University; Abby R. O’Connor, The College of New Jersey, [email protected]. Copyright Scheuermann and O’Connor, 2014. This work is licensed under the Creative Commons Attribution-NonCommercialShareAlike 3.0 Unported License. To view a copy of this licens ...
... Created by Margaret L. Scheuermann, Princeton University; Abby R. O’Connor, The College of New Jersey, [email protected]. Copyright Scheuermann and O’Connor, 2014. This work is licensed under the Creative Commons Attribution-NonCommercialShareAlike 3.0 Unported License. To view a copy of this licens ...
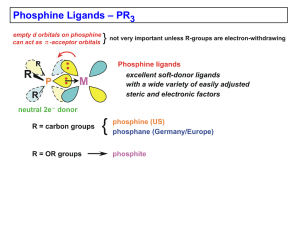
Chapter 4 (Phosphines)
... phosphides, PR2-, are very common). Phosphines generally tend to orient trans to one another in order to minimize steric interactions (especially true for bulky PR3). Chelating bisphosphine ligands are used to enforce cisoidal coordination geometries when needed. Some typical first row M-PR3 average ...
... phosphides, PR2-, are very common). Phosphines generally tend to orient trans to one another in order to minimize steric interactions (especially true for bulky PR3). Chelating bisphosphine ligands are used to enforce cisoidal coordination geometries when needed. Some typical first row M-PR3 average ...
Versatile Cooperative Ligand Effects in Group 9 Transition Metal
... Chemical production helps to sustain the modern society by providing people with energy, medicine and materials. Catalysis plays a central role in chemical processes, as it serves to provide costefficient, “green” and selective pathways for otherwise highly energy-consuming, environmentallyhazardous ...
... Chemical production helps to sustain the modern society by providing people with energy, medicine and materials. Catalysis plays a central role in chemical processes, as it serves to provide costefficient, “green” and selective pathways for otherwise highly energy-consuming, environmentallyhazardous ...
Metal Sequestration (English version)
... extraction, or technological process employing for example ion exchange or chelating resins. It can be affirmed that sequestration consists in the formation of some co-ordination state of the metal ion in solution which is stronger than the previous co-ordination condition. Therefore it can be assum ...
... extraction, or technological process employing for example ion exchange or chelating resins. It can be affirmed that sequestration consists in the formation of some co-ordination state of the metal ion in solution which is stronger than the previous co-ordination condition. Therefore it can be assum ...
Transition Metal Complexes
... The violet product consists of a pair of optical isomers. The green product is not optically active, as it has a mirror plane. ...
... The violet product consists of a pair of optical isomers. The green product is not optically active, as it has a mirror plane. ...
Topics • Introduction • Molecular Structure and Bonding • Molecular
... Sigma Bonding Ligands • All ligands so far have been using sigma bonding – donate electrons from lone pairs to the metal center typically from either s or p orbitals – typical examples amines, water, nitrite ion, alkyl ligands ...
... Sigma Bonding Ligands • All ligands so far have been using sigma bonding – donate electrons from lone pairs to the metal center typically from either s or p orbitals – typical examples amines, water, nitrite ion, alkyl ligands ...
Slide 1
... Problem: One could use electron transfer catalysis (ETC) to further activate the very inert trans-Cr(CO)3(PMe3)3 complex that we discussed earlier for another CO substitution. To initiate the ETC you can either oxidize the complex to [Cr(CO)3(PMe3)3]+ (17e-, half-empty orbital) or reduce it to [Cr( ...
... Problem: One could use electron transfer catalysis (ETC) to further activate the very inert trans-Cr(CO)3(PMe3)3 complex that we discussed earlier for another CO substitution. To initiate the ETC you can either oxidize the complex to [Cr(CO)3(PMe3)3]+ (17e-, half-empty orbital) or reduce it to [Cr( ...
Chapter 1: Fundamental Concepts
... • The oxidation state of the metal (charge!) D is greater for M3+ than for M2+ • The row of the metal in the periodic table (size!) for a given ligand and oxidation state of the metal, D increases going down in a group e.g. D is greater in Ru(NH3)63+ than in Fe(NH3)63+ Colors of metal complexes are ...
... • The oxidation state of the metal (charge!) D is greater for M3+ than for M2+ • The row of the metal in the periodic table (size!) for a given ligand and oxidation state of the metal, D increases going down in a group e.g. D is greater in Ru(NH3)63+ than in Fe(NH3)63+ Colors of metal complexes are ...
Full Text: PDF - Revistas USACH
... were calculated. The proton-ligand formation curves are obtained by plotting nA versus pH at constant temperature. It was found from these that the maximum nA value is 1.7 for HL1 indicating that the ligand has two pKa values. The first dissociation constant, pKa1 (3.94) is assigned to NH proton whi ...
... were calculated. The proton-ligand formation curves are obtained by plotting nA versus pH at constant temperature. It was found from these that the maximum nA value is 1.7 for HL1 indicating that the ligand has two pKa values. The first dissociation constant, pKa1 (3.94) is assigned to NH proton whi ...
Ligand
4-3D-balls.png?width=300)
In coordination chemistry, a ligand (/lɪɡənd/) is an ion or molecule (functional group) that binds to a central metal atom to form a coordination complex. The bonding between metal and ligand generally involves formal donation of one or more of the ligand's electron pairs. The nature of metal-ligand bonding can range from covalent to ionic. Furthermore, the metal-ligand bond order can range from one to three. Ligands are viewed as Lewis bases, although rare cases are known to involve Lewis acidic ""ligand.""Metals and metalloids are bound to ligands in virtually all circumstances, although gaseous ""naked"" metal ions can be generated in high vacuum. Ligands in a complex dictate the reactivity of the central atom, including ligand substitution rates, the reactivity of the ligands themselves, and redox. Ligand selection is a critical consideration in many practical areas, including bioinorganic and medicinal chemistry, homogeneous catalysis, and environmental chemistry.Ligands are classified in many ways like : their charge, their size (bulk), the identity of the coordinating atom(s), and the number of electrons donated to the metal (denticity or hapticity). The size of a ligand is indicated by its cone angle.