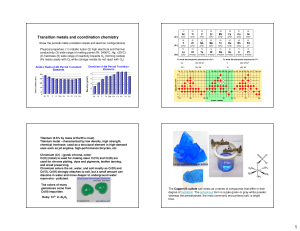
Experiment 4 Spectroscopic study of Cu(II) Complexes: Crystal Field
... Dissolve about 1.0 g of copper sulphate, CuSO4.5H2O, in 8.5 ml of 1 M HCl and add 700 mg of glycine (H2NCH2CO2H). Cautiously warm the mixture in the water bath or on hot plate for 30 minutes, and during this period, slowly add solid NaHCO3 to the warm solution until a crystalline precipitate is comp ...
... Dissolve about 1.0 g of copper sulphate, CuSO4.5H2O, in 8.5 ml of 1 M HCl and add 700 mg of glycine (H2NCH2CO2H). Cautiously warm the mixture in the water bath or on hot plate for 30 minutes, and during this period, slowly add solid NaHCO3 to the warm solution until a crystalline precipitate is comp ...
Concepts in Transition Metal Chemistry – Answers
... A moment of 4.8 µB would correspond to 4 or 3 unpaired electrons. The most stable oxidation states of Fe are +2 and +3. The +2 state has an electronic configuration of d6 and the +3 state a configuration of d5. In a weak field these would have 4 and 5 unpaired electrons respectively. For a strong fi ...
... A moment of 4.8 µB would correspond to 4 or 3 unpaired electrons. The most stable oxidation states of Fe are +2 and +3. The +2 state has an electronic configuration of d6 and the +3 state a configuration of d5. In a weak field these would have 4 and 5 unpaired electrons respectively. For a strong fi ...
MO Theory
... o double bonds do not allow rotation o triple bonds allow partial rotation o single bonds allow full rotation Arromatic molecules and delocalized bonds o C6H6 MO of bonds Cyclic molecules: o Energy level diagram Draw molecue with point down: Put one orbital for each apex Fill in electr ...
... o double bonds do not allow rotation o triple bonds allow partial rotation o single bonds allow full rotation Arromatic molecules and delocalized bonds o C6H6 MO of bonds Cyclic molecules: o Energy level diagram Draw molecue with point down: Put one orbital for each apex Fill in electr ...
Coordination Compounds
... in a square bracket, collectively termed as coordination sphere. The ionisable groups are written outside the bracket, called counter ions. For example, in the complex K4[Fe(CN)6] the coordination sphere is[Fe(CN)6]4–, and the counter ion is K+. (viii) Homoleptic and Heteroleptic complexes: Complexe ...
... in a square bracket, collectively termed as coordination sphere. The ionisable groups are written outside the bracket, called counter ions. For example, in the complex K4[Fe(CN)6] the coordination sphere is[Fe(CN)6]4–, and the counter ion is K+. (viii) Homoleptic and Heteroleptic complexes: Complexe ...
Transition metal Catalyzed Reactions
... from the metal, it is a neutral molecule with one lone pair of electrons. Therefore, as with the ionic model, ammonia is a neutral two electron donor. But we diverge from the ionic model when we consider a ligand such as methyl. When we remove it from the metal and make the methyl fragment neutral, ...
... from the metal, it is a neutral molecule with one lone pair of electrons. Therefore, as with the ionic model, ammonia is a neutral two electron donor. But we diverge from the ionic model when we consider a ligand such as methyl. When we remove it from the metal and make the methyl fragment neutral, ...
LOYOLA COLLEGE (AUTONOMOUS), CHENNAI – 600 034
... 17. Explain SN1CB mechanism with a specific example. 18. Discuss the synthesis and uses of cis -platin. 19. Explain photoisomerisation reaction with an example. 20. What is Ziegler-Natta catalyst? Mention its application. 21. What are blue copper proteins? Differentiate it from other copper proteins ...
... 17. Explain SN1CB mechanism with a specific example. 18. Discuss the synthesis and uses of cis -platin. 19. Explain photoisomerisation reaction with an example. 20. What is Ziegler-Natta catalyst? Mention its application. 21. What are blue copper proteins? Differentiate it from other copper proteins ...
Changing Coordination Numbers: Nickel Complexes
... A complex ion is a metal ion with Lewis bases attached to it through coordinate covalent bonds. A complex or coordination compound is a compound consisting either of complex ions with other ions of opposite charge or of a neutral complex species. Ligands are the Lewis bases attached to the metal ato ...
... A complex ion is a metal ion with Lewis bases attached to it through coordinate covalent bonds. A complex or coordination compound is a compound consisting either of complex ions with other ions of opposite charge or of a neutral complex species. Ligands are the Lewis bases attached to the metal ato ...
ppt
... Working out numbers of d-electrons from oxidation states: 1st: how many electrons are there in the shell? - count along the periodic table e.g. Mn = 7 electrons ...
... Working out numbers of d-electrons from oxidation states: 1st: how many electrons are there in the shell? - count along the periodic table e.g. Mn = 7 electrons ...
Elements, basic principles, periodic table
... - ion is larger than the neutral atom Ions behave the same as atoms across the periodic table (row vs column Importance of the radius: molecules can only “fit” certain sizes ...
... - ion is larger than the neutral atom Ions behave the same as atoms across the periodic table (row vs column Importance of the radius: molecules can only “fit” certain sizes ...
Answer - Assignment Expert
... For historic reasons, some coordination compounds are called by their common names. For example, Fe(CN)63- and Fe(CN)64- are named ferricyanide and ferrocyanide respectively, and Fe(CO)5 is called iron carbonyl. Examples 1. [Cr(NH3)3(H2O)3]Cl3 Name: triamminetriaquachromium(III) chloride Solution: ...
... For historic reasons, some coordination compounds are called by their common names. For example, Fe(CN)63- and Fe(CN)64- are named ferricyanide and ferrocyanide respectively, and Fe(CO)5 is called iron carbonyl. Examples 1. [Cr(NH3)3(H2O)3]Cl3 Name: triamminetriaquachromium(III) chloride Solution: ...
Chapter 7 Acids, bases and ions in aqueous solution
... solution containing the solute MY (the ion Mn+ is common to both salts), the presence of the dissolved Mn+ ions suppresses the dissolution of MX compared with that in pure water. ...
... solution containing the solute MY (the ion Mn+ is common to both salts), the presence of the dissolved Mn+ ions suppresses the dissolution of MX compared with that in pure water. ...
Review of Basic Principles: The following was adapted from The
... the CO-to-metal electron donation that constitutes the σ bond. One of the most direct arguments is structural. The M=C bond in metal carbonyls is usually substantially shorter than an M-C single bond. This is easiest to test when both types of bond are present in the same complex, such as CpMo(CO) 3 ...
... the CO-to-metal electron donation that constitutes the σ bond. One of the most direct arguments is structural. The M=C bond in metal carbonyls is usually substantially shorter than an M-C single bond. This is easiest to test when both types of bond are present in the same complex, such as CpMo(CO) 3 ...
UNIT 9: CO-ORDINATION COMPOUNDS
... Coordination isomerism: It arises from the interchange of ligands between cationic and anionic entities of different metal ions present in a complex . Example [Co(NH3)6][Cr(CN)6] & [Cr(NH3)6][Co(CN)6] STEREOISOMERISM: Stereo isomers have the same chemical formula and chemical bonds but they have dif ...
... Coordination isomerism: It arises from the interchange of ligands between cationic and anionic entities of different metal ions present in a complex . Example [Co(NH3)6][Cr(CN)6] & [Cr(NH3)6][Co(CN)6] STEREOISOMERISM: Stereo isomers have the same chemical formula and chemical bonds but they have dif ...
Electrochemical, in-vitro in-vivo study of Co (II)-ofloxacin complex Deepa Thakur ,Jyotsna Mishra,
... and diffusion controlled Polarographic wave which has revealed by the log plot slope and the plots of i d versus √h effective hight of mercury column respectively on the gradual addition of the ligand the E 1/2 of the metal shifted to more electronegative values indicating the formulation of Co (II) ...
... and diffusion controlled Polarographic wave which has revealed by the log plot slope and the plots of i d versus √h effective hight of mercury column respectively on the gradual addition of the ligand the E 1/2 of the metal shifted to more electronegative values indicating the formulation of Co (II) ...
Syntheses, Structures and Photophysical Properties of Metal
... From the fact that the LUMO’s for the new complexes involve metalmetal antibonding (possibility of fragmentation), the life-time of the excited state of these complexes is shortened compared to the luminophore itself. From the earlier discussion of the absorption/emission spectra, we know that there ...
... From the fact that the LUMO’s for the new complexes involve metalmetal antibonding (possibility of fragmentation), the life-time of the excited state of these complexes is shortened compared to the luminophore itself. From the earlier discussion of the absorption/emission spectra, we know that there ...
Ligand Field Theory - Sites at Penn State
... Assignment of Oxidation States ★ We can take the analysis of covalency one step further in order to „recover“ the concept of an oxidation state from our calculations. ★ This is even more approximate since it must be based on a subjective criterion ...
... Assignment of Oxidation States ★ We can take the analysis of covalency one step further in order to „recover“ the concept of an oxidation state from our calculations. ★ This is even more approximate since it must be based on a subjective criterion ...
Bioinorganic Chemistry
... The bound iron(III) ion at each binding site is surrounded by six donor atoms from various ligands. Thus, two oxygen atoms of a carbonate anion coordinate to the metal, and the following amino acid side chains from the protein primary structure also coordinate to the iron(III) ion with one potential ...
... The bound iron(III) ion at each binding site is surrounded by six donor atoms from various ligands. Thus, two oxygen atoms of a carbonate anion coordinate to the metal, and the following amino acid side chains from the protein primary structure also coordinate to the iron(III) ion with one potential ...
Shapes of d
... 1 Cl•Central metal ion or atom surrounded by a set of ligands •The ligand donates two electrons to the d-orbitals around the metal forming a dative or coordinate bond ...
... 1 Cl•Central metal ion or atom surrounded by a set of ligands •The ligand donates two electrons to the d-orbitals around the metal forming a dative or coordinate bond ...
Lecture notes for chapter 7
... far more readily to a metal for steric (more open, less hindered) and electronic (better donor, 2 or 4e- bonding) reasons. They are also quite a bit more reactive for doing C-C coupling and other reactions. When alkynes bridge, they almost always do so perpendicular to the MM axis, the parallel brid ...
... far more readily to a metal for steric (more open, less hindered) and electronic (better donor, 2 or 4e- bonding) reasons. They are also quite a bit more reactive for doing C-C coupling and other reactions. When alkynes bridge, they almost always do so perpendicular to the MM axis, the parallel brid ...
Group 14 - University of Ottawa
... compounds can react directly with dihalides to eliminate an alkyl halide: R3E + X2 R3EX2 (heat) R2EX + RX This intermediate is one route (metathesis) to ER5. The homoleptic +5 alkyls decompose on heating to produce the +3 alkyls, producing alkanes, alkenes, hydrogen. Aryl analogues are more stabl ...
... compounds can react directly with dihalides to eliminate an alkyl halide: R3E + X2 R3EX2 (heat) R2EX + RX This intermediate is one route (metathesis) to ER5. The homoleptic +5 alkyls decompose on heating to produce the +3 alkyls, producing alkanes, alkenes, hydrogen. Aryl analogues are more stabl ...
Transition Chemistry
... the transition metal elements are inner electrons: d electrons for the d-block transition metals and f electrons for the lanthanides and actinides. These inner d and f electrons cannot participate in bonding as readily as the valence s and p electrons. Thus, the chemistry of transition elements is n ...
... the transition metal elements are inner electrons: d electrons for the d-block transition metals and f electrons for the lanthanides and actinides. These inner d and f electrons cannot participate in bonding as readily as the valence s and p electrons. Thus, the chemistry of transition elements is n ...
General Properties of Transition Metals
... • Neutral molecule or ion having a lone electron pair that can be used to form a bond to a metal ion. Monodentate ligand – one bond to a metal ion Bidentate ligand (chelate) – two bonds to a metal ion Polydentate ligand – more than two bonds to a metal ion ...
... • Neutral molecule or ion having a lone electron pair that can be used to form a bond to a metal ion. Monodentate ligand – one bond to a metal ion Bidentate ligand (chelate) – two bonds to a metal ion Polydentate ligand – more than two bonds to a metal ion ...
Ligand
4-3D-balls.png?width=300)
In coordination chemistry, a ligand (/lɪɡənd/) is an ion or molecule (functional group) that binds to a central metal atom to form a coordination complex. The bonding between metal and ligand generally involves formal donation of one or more of the ligand's electron pairs. The nature of metal-ligand bonding can range from covalent to ionic. Furthermore, the metal-ligand bond order can range from one to three. Ligands are viewed as Lewis bases, although rare cases are known to involve Lewis acidic ""ligand.""Metals and metalloids are bound to ligands in virtually all circumstances, although gaseous ""naked"" metal ions can be generated in high vacuum. Ligands in a complex dictate the reactivity of the central atom, including ligand substitution rates, the reactivity of the ligands themselves, and redox. Ligand selection is a critical consideration in many practical areas, including bioinorganic and medicinal chemistry, homogeneous catalysis, and environmental chemistry.Ligands are classified in many ways like : their charge, their size (bulk), the identity of the coordinating atom(s), and the number of electrons donated to the metal (denticity or hapticity). The size of a ligand is indicated by its cone angle.