handout alkenes from alcohols
... This procedure has been adapted from the microscale procedure described in the third edition of Macroscale and Microscale Organic Experiments by Kenneth L. Williamson (Houghton Mifflin, Boston, 1999). ...
... This procedure has been adapted from the microscale procedure described in the third edition of Macroscale and Microscale Organic Experiments by Kenneth L. Williamson (Houghton Mifflin, Boston, 1999). ...
ppt
... 2.Carbon dioxide has one carbon atom. How about forming CCO2 bond to synthesize potential organic compound ? ...
... 2.Carbon dioxide has one carbon atom. How about forming CCO2 bond to synthesize potential organic compound ? ...
Part 1
... Rule #2: Number the carbons in the main chain Number chain to minimize the position/number of the following in order of priority: a) thing you’re naming the compound after (double bond if alkene; -OH group if alcohol, etc) note: for multiple double bonds -diene, -triene, -tetraene b) first branch/s ...
... Rule #2: Number the carbons in the main chain Number chain to minimize the position/number of the following in order of priority: a) thing you’re naming the compound after (double bond if alkene; -OH group if alcohol, etc) note: for multiple double bonds -diene, -triene, -tetraene b) first branch/s ...
Chapter 7 Notes - Alcohols, Ethers, Thiols Functional groups alcohol
... CH3OH + NaNH2 ---> NH3 + CH3O- Na+ (sodium methoxide) most commonly made by direct reaction with active metals CH3OH + Na ---> 1/2 H2 + CH3O- Na+ (CH3)3COH + K ---> 1/2 H2 + (CH3)3CO-K+ ...
... CH3OH + NaNH2 ---> NH3 + CH3O- Na+ (sodium methoxide) most commonly made by direct reaction with active metals CH3OH + Na ---> 1/2 H2 + CH3O- Na+ (CH3)3COH + K ---> 1/2 H2 + (CH3)3CO-K+ ...
Crown ethers
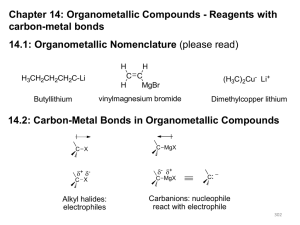
... These compounds contain carbon- metal (lithium) bond. They react in a similar manner to Grignard reagents, and are very useful in synthesis ...
... These compounds contain carbon- metal (lithium) bond. They react in a similar manner to Grignard reagents, and are very useful in synthesis ...
Organometallic Compounds - Reagents
... to a starting compound using known and reliable reactions. “it is a problem solving technique for transforming the structure of a synthetic target molecule (TM) to a sequence of progressively simpler structures along the pathway which ultimately leads to simple or commercially available starting mat ...
... to a starting compound using known and reliable reactions. “it is a problem solving technique for transforming the structure of a synthetic target molecule (TM) to a sequence of progressively simpler structures along the pathway which ultimately leads to simple or commercially available starting mat ...
Experiment 4- Alkene
... (ii) Reactions of Alkenes Alkenes, containing a site of unsaturation, undergo electrophilic addition reactions with several reagents such as halogens, oxidizing agents, and sulfuric, halogen, and hypohalous acids. In particular, bromine and oxidizing agents such as permanganate are widely used in qu ...
... (ii) Reactions of Alkenes Alkenes, containing a site of unsaturation, undergo electrophilic addition reactions with several reagents such as halogens, oxidizing agents, and sulfuric, halogen, and hypohalous acids. In particular, bromine and oxidizing agents such as permanganate are widely used in qu ...
Alcohol oxidation
... iodine, which can easily be observed by its violet color. For closer control of the reaction itself, an indicator such as Sudan Red III can be added to the reaction mixture. Ozone reacts with this indicator more slowly than with the intended ozonolysis target. After completing the addition a reagent ...
... iodine, which can easily be observed by its violet color. For closer control of the reaction itself, an indicator such as Sudan Red III can be added to the reaction mixture. Ozone reacts with this indicator more slowly than with the intended ozonolysis target. After completing the addition a reagent ...
Organic Chemistry III Laboratory
... These biocatalytic systems present some advantages because they can produce reactions under mild conditions with high enantio- or diastereoselectivity. Obtaining purified enzymes is costly, however, microorganisms can be easily obtained and exploited to carry out enzymatic reactions. A whole-cell sy ...
... These biocatalytic systems present some advantages because they can produce reactions under mild conditions with high enantio- or diastereoselectivity. Obtaining purified enzymes is costly, however, microorganisms can be easily obtained and exploited to carry out enzymatic reactions. A whole-cell sy ...
4.5 Topic Checklist Carbonyl Compounds
... appreciate the hazards of synthesis using HCN/KCN know that aldehydes can be reduced to primary alcohols and ketones to secondary alcohols using reducing agents such as NaBH4. Mechanisms showing H– are required (equations showing [H] as reductant are acceptable) understand the mechanism of the react ...
... appreciate the hazards of synthesis using HCN/KCN know that aldehydes can be reduced to primary alcohols and ketones to secondary alcohols using reducing agents such as NaBH4. Mechanisms showing H– are required (equations showing [H] as reductant are acceptable) understand the mechanism of the react ...
pptx
... provided that rigorously anhydrous conditions are employed. One disadvantage of the Collins reagent is that a large excess is often required in order to achieve good yields. This is particularly problematic given the toxicity and disposal issues surrounding Cr (VI) reagents. ...
... provided that rigorously anhydrous conditions are employed. One disadvantage of the Collins reagent is that a large excess is often required in order to achieve good yields. This is particularly problematic given the toxicity and disposal issues surrounding Cr (VI) reagents. ...
handout alkenes from alcohols
... This procedure has been adapted from the microscale procedure described in the third edition of Macroscale and Microscale Organic Experiments by Kenneth L. Williamson (Houghton Mifflin, Boston, 1999). ...
... This procedure has been adapted from the microscale procedure described in the third edition of Macroscale and Microscale Organic Experiments by Kenneth L. Williamson (Houghton Mifflin, Boston, 1999). ...
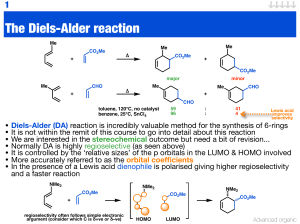
The Diels-Alder reaction
... • Last year (2005) saw the first examples of catalytic enantioselective Negishi couplings • The system still has some limitations but is an exciting development • On a practical note, many of the reactions above were run in air!!! Advanced organic ...
... • Last year (2005) saw the first examples of catalytic enantioselective Negishi couplings • The system still has some limitations but is an exciting development • On a practical note, many of the reactions above were run in air!!! Advanced organic ...
Grignard Reagents
... to a starting compound using known and reliable reactions. “it is a problem solving technique for transforming the structure of a synthetic target molecule (TM) to a sequence of progressively simpler structures along the pathway which ultimately leads to simple or commercially available starting mat ...
... to a starting compound using known and reliable reactions. “it is a problem solving technique for transforming the structure of a synthetic target molecule (TM) to a sequence of progressively simpler structures along the pathway which ultimately leads to simple or commercially available starting mat ...
91391 Demonstrate understanding of the properties of organic
... Fehling’s and Benedict’s. Reduction of aldehydes and ketones with NaBH4 elimination reactions using the following reagents: KOH in alcohol and concentrated H2SO4 (includes major and minor products from asymmetric alcohols and haloalkanes) polymerisation reactions involving formation of polyester ...
... Fehling’s and Benedict’s. Reduction of aldehydes and ketones with NaBH4 elimination reactions using the following reagents: KOH in alcohol and concentrated H2SO4 (includes major and minor products from asymmetric alcohols and haloalkanes) polymerisation reactions involving formation of polyester ...
91391 Demonstrate understanding of the properties of organic
... Fehling’s and Benedict’s. Reduction of aldehydes and ketones with NaBH4 elimination reactions using the following reagents: KOH in alcohol and concentrated H2SO4 (includes major and minor products from asymmetric alcohols and haloalkanes) polymerisation reactions involving formation of polyester ...
... Fehling’s and Benedict’s. Reduction of aldehydes and ketones with NaBH4 elimination reactions using the following reagents: KOH in alcohol and concentrated H2SO4 (includes major and minor products from asymmetric alcohols and haloalkanes) polymerisation reactions involving formation of polyester ...
2130404 - Gujarat Technological University
... List of Experiments and Open Ended Projects: Minimum 5 practicals to be performed and remaining Open-ended Projects / Study Reports / Latest outcomes in technology study :1. In the beginning of the academic term, faculties will have to allot their students at least one Projects / Study Reports / Lat ...
... List of Experiments and Open Ended Projects: Minimum 5 practicals to be performed and remaining Open-ended Projects / Study Reports / Latest outcomes in technology study :1. In the beginning of the academic term, faculties will have to allot their students at least one Projects / Study Reports / Lat ...
D:\Course Outline PDF F2015\Word Format\JAC Organic Chemistry Ix
... nucleophiles, radical elements, Lewis acids and bases. Acquire a working knowledge of ‘arrow-pushing’ to illustrate the mechanisms of common reactions. Write mechanisms for a wide variety of important chemical reactions. The reactions studied include some or all of the following; the synthesis of al ...
... nucleophiles, radical elements, Lewis acids and bases. Acquire a working knowledge of ‘arrow-pushing’ to illustrate the mechanisms of common reactions. Write mechanisms for a wide variety of important chemical reactions. The reactions studied include some or all of the following; the synthesis of al ...
Organometallic Chemistry between organic and inorganic
... olefin hydrogenation and CO oxidation. Organometallic compounds are used on a large scale as precursors for generation of semiconductors (AlN, GaAs, etc). Silicone rubbers are one of the few classes of organometallic compounds used as "final products". ...
... olefin hydrogenation and CO oxidation. Organometallic compounds are used on a large scale as precursors for generation of semiconductors (AlN, GaAs, etc). Silicone rubbers are one of the few classes of organometallic compounds used as "final products". ...
Full Article-PDF - UNC
... to -78°C. The resulting pale yellow solution was stirred 30 min. Methyl isobutyrate (15.00 g, 0.147 mol) in 50 mL THF was added dropwise over 30 min and the light yellow solution was stirred for an additional 1 h. Allyl bromide (19.54 g, 0.162 mol) in 50 mL THF was added dropwise and the solution wa ...
... to -78°C. The resulting pale yellow solution was stirred 30 min. Methyl isobutyrate (15.00 g, 0.147 mol) in 50 mL THF was added dropwise over 30 min and the light yellow solution was stirred for an additional 1 h. Allyl bromide (19.54 g, 0.162 mol) in 50 mL THF was added dropwise and the solution wa ...
Topics 10 and 20 Outline
... Use of data—much of the progress that has been made to date in the developments and applications of scientific research can be mapped back to key organic chemical reactions involving functional group interconversions. (3.1) Understandings: • Alkanes have low reactivity and undergo free-radical subst ...
... Use of data—much of the progress that has been made to date in the developments and applications of scientific research can be mapped back to key organic chemical reactions involving functional group interconversions. (3.1) Understandings: • Alkanes have low reactivity and undergo free-radical subst ...
Elias James Corey

Elias James ""E.J."" Corey (born July 12, 1928) is an American organic chemist. In 1990, he won the Nobel Prize in Chemistry ""for his development of the theory and methodology of organic synthesis"", specifically retrosynthetic analysis. Regarded by many as one of the greatest living chemists, he has developed numerous synthetic reagents, methodologies and total syntheses and has advanced the science of organic synthesis considerably.