"Street Chemist" Part-12 - Sutter County Fire Department
... is toxic by ingestion and can cause blindness or death, with a TLV of 200 ppm. Ethyl alcohol or ethanol, also referred to as grain alcohol, is consumed in alcoholic beverages. It is classified as a depressant drug; too much of it can produce toxic effects and can lead to liver damage. The alcohol fu ...
... is toxic by ingestion and can cause blindness or death, with a TLV of 200 ppm. Ethyl alcohol or ethanol, also referred to as grain alcohol, is consumed in alcoholic beverages. It is classified as a depressant drug; too much of it can produce toxic effects and can lead to liver damage. The alcohol fu ...
Studying Sn1 and Sn2 reactions: Nucleophillic substitution
... The leaving group: A weak base The carbon group: unhindered by the presence of bulky groups is better for Sn2 reactions ...
... The leaving group: A weak base The carbon group: unhindered by the presence of bulky groups is better for Sn2 reactions ...
lec-2- 211(ES +Add)
... HX which can donate a proton, H2O should be able to add to alkenes in the same way as HBr, for example, resulting in the hydration of an alkene. However, for the addition of H2O to alkenes to occur acid catalysts are required. ...
... HX which can donate a proton, H2O should be able to add to alkenes in the same way as HBr, for example, resulting in the hydration of an alkene. However, for the addition of H2O to alkenes to occur acid catalysts are required. ...
CH 3 CH 2 CH 2 CH 2 CH 2 CH 2 OH
... points of the different hydrocarbons. fractional distillation ...
... points of the different hydrocarbons. fractional distillation ...
Polyesters are condensation polymers.
... Note that sometimes other molecules (HCl for eg) are lost in other condensation reactions. It is the elimination of a molecule which makes it a condensation reaction, not the loss of water. ...
... Note that sometimes other molecules (HCl for eg) are lost in other condensation reactions. It is the elimination of a molecule which makes it a condensation reaction, not the loss of water. ...
GRADE 11F: Chemistry 6
... a. For each of the reactions I, II and III give suitable reagents and conditions. b. If 2-methylpropan-2-ol was used as a starting material in a instead of propan-2ol, identify the organic products, if any, of reactions I, II and III. You should indicate if no reaction occurs. London (Nuffield) 1998 ...
... a. For each of the reactions I, II and III give suitable reagents and conditions. b. If 2-methylpropan-2-ol was used as a starting material in a instead of propan-2ol, identify the organic products, if any, of reactions I, II and III. You should indicate if no reaction occurs. London (Nuffield) 1998 ...
BioN02 Introduction to organic chemistry Summer 2014
... The hydroxyl group takes precedence over alkyl groups and halogen substituents, as well as double bonds, in the numbering of the parent chain Alcohols may also be classified as primary, 1º, secondary, 2º & tertiary, 3º (carbons attached to the carbon with –OH) ...
... The hydroxyl group takes precedence over alkyl groups and halogen substituents, as well as double bonds, in the numbering of the parent chain Alcohols may also be classified as primary, 1º, secondary, 2º & tertiary, 3º (carbons attached to the carbon with –OH) ...
The SN2 Reaction: 1
... prepared by heating the corresponding alcohol with (1) constantboiling hydrobromic acid (47% HBr), (2) an aqueous solution of sodium bromide and excess sulfuric acid, which is an equilibrium mixture containing hydrobromic acid, or (3) a solution of hydrobromic acid produced by bubbling sulfur dioxid ...
... prepared by heating the corresponding alcohol with (1) constantboiling hydrobromic acid (47% HBr), (2) an aqueous solution of sodium bromide and excess sulfuric acid, which is an equilibrium mixture containing hydrobromic acid, or (3) a solution of hydrobromic acid produced by bubbling sulfur dioxid ...
CaCl2.2H2O assisted oxidation of alcohols with (NH4)2Cr2O7
... dichromate in the presence of CaCl2.2H2O (Table I, Scheme I). The reaction is simply performed by stirring a mixture of alcohol, (NH4)2Cr2O7 and CaCl2.2H2O in an oil-bath (60°C) for the appropriate time (Table I). Alcohols were oxidized efficiently and the corresponding carbonyl compounds were isola ...
... dichromate in the presence of CaCl2.2H2O (Table I, Scheme I). The reaction is simply performed by stirring a mixture of alcohol, (NH4)2Cr2O7 and CaCl2.2H2O in an oil-bath (60°C) for the appropriate time (Table I). Alcohols were oxidized efficiently and the corresponding carbonyl compounds were isola ...
Bonds - MCAT Cooperative
... Substituents can occupy axial and equatorial positions. Axia (6) - perpendicular to the ring Equatorial (6)- roughly in the plane of the ring ...
... Substituents can occupy axial and equatorial positions. Axia (6) - perpendicular to the ring Equatorial (6)- roughly in the plane of the ring ...
Lab 7
... Oxidation reactions involve the addition of oxygen or the removal of hydrogen. First, we shall learn to identify structures that can undergo oxidation. Then, we shall learn the reagents that can oxidize the structures. Oxidation reactions require an “activated” carbon atom such as that shown in Figu ...
... Oxidation reactions involve the addition of oxygen or the removal of hydrogen. First, we shall learn to identify structures that can undergo oxidation. Then, we shall learn the reagents that can oxidize the structures. Oxidation reactions require an “activated” carbon atom such as that shown in Figu ...
10. Alkyl Halides - Clayton State University
... In organic chemistry, we say that oxidation occurs when a carbon or hydrogen that is connected to a carbon atom in a structure is replaced by oxygen, nitrogen, or halogen Not defined as loss of electrons by an atom as in inorganic chemistry Oxidation is a reaction that results in loss of electron ...
... In organic chemistry, we say that oxidation occurs when a carbon or hydrogen that is connected to a carbon atom in a structure is replaced by oxygen, nitrogen, or halogen Not defined as loss of electrons by an atom as in inorganic chemistry Oxidation is a reaction that results in loss of electron ...
CHEM 122: Introduction to Organic Chemistry Chapter 9: Aldehydes
... 22. Describe a simple chemical test by which you could distinguish between the members of each pair of compounds. a) Cyclohexanone and aniline b) Cyclohexene and cyclohexanol c) Benzaldehyde and cinnamaldehyde 23. 5-Hydroxyhexanal forms a six-membered cyclic hemiacetal, which predominates at equilib ...
... 22. Describe a simple chemical test by which you could distinguish between the members of each pair of compounds. a) Cyclohexanone and aniline b) Cyclohexene and cyclohexanol c) Benzaldehyde and cinnamaldehyde 23. 5-Hydroxyhexanal forms a six-membered cyclic hemiacetal, which predominates at equilib ...
13.2 Families of Compounds
... Using the information in the table on the previous page to predict the chemical family to which the following compounds are members: ...
... Using the information in the table on the previous page to predict the chemical family to which the following compounds are members: ...
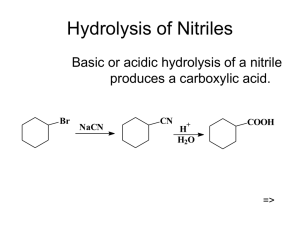
Hydrolysis of Nitriles
... Acid Chlorides An activated form of the carboxylic acid. Chloride is a good leaving group, so undergoes acyl substitution easily. To synthesize acid chlorides use thionyl chloride or oxalyl chloride with the acid. O ...
... Acid Chlorides An activated form of the carboxylic acid. Chloride is a good leaving group, so undergoes acyl substitution easily. To synthesize acid chlorides use thionyl chloride or oxalyl chloride with the acid. O ...
Exam 1
... Acid catalyzed ester & amide hydrolysis, Fisher esterification, trans esterification Hydroxide promoted ester hydrolysis Nucleophilic addition to aldehyde/ketone with Grignard reagents Determine reaction mechanisms from reaction outcome (see Mechanism work sheet). Be able to recognize the following ...
... Acid catalyzed ester & amide hydrolysis, Fisher esterification, trans esterification Hydroxide promoted ester hydrolysis Nucleophilic addition to aldehyde/ketone with Grignard reagents Determine reaction mechanisms from reaction outcome (see Mechanism work sheet). Be able to recognize the following ...
Alcohol

In chemistry, an alcohol is any organic compound in which the hydroxyl functional group (–OH) is bound to a saturated carbon atom. The term alcohol originally referred to the primary alcohol ethyl alcohol (ethanol), the predominant alcohol in alcoholic beverages.The suffix -ol appears in the IUPAC chemical name of all substances where the hydroxyl group is the functional group with the highest priority; in substances where a higher priority group is present the prefix hydroxy- will appear in the IUPAC name. The suffix -ol in non-systematic names (such as paracetamol or cholesterol) also typically indicates that the substance includes a hydroxyl functional group and, so, can be termed an alcohol. But many substances, particularly sugars (examples glucose and sucrose) contain hydroxyl functional groups without using the suffix. An important class of alcohols, of which methanol and ethanol are the simplest members is the saturated straight chain alcohols, the general formula for which is CnH2n+1OH.