W19 Aldehydes ketones I
... reaction scheme of aldehydes and ketones nucleophilic addition AN to C=O group: ...
... reaction scheme of aldehydes and ketones nucleophilic addition AN to C=O group: ...
Exam 2 Review A
... using an arrow-pushing mechanism to explain how the reaction works. a. Be able to explain the differences between this reaction and the reverse process, i.e., the acid-catalyzed dehydration of alcohols to form alkenes. 4. Be able to explain how alkenes can be hydrated in a Markovnikov fashion using ...
... using an arrow-pushing mechanism to explain how the reaction works. a. Be able to explain the differences between this reaction and the reverse process, i.e., the acid-catalyzed dehydration of alcohols to form alkenes. 4. Be able to explain how alkenes can be hydrated in a Markovnikov fashion using ...
Epoxidation and oxidation reactions using 1,4
... Solid-phase organic synthesis has been used as an efficient technique for the synthesis and screening of a large number of organic compounds.1 Over the past years, numerous techniques have been used to study polymeric resins commonly utilized in solid phase synthesis to allow greater understanding o ...
... Solid-phase organic synthesis has been used as an efficient technique for the synthesis and screening of a large number of organic compounds.1 Over the past years, numerous techniques have been used to study polymeric resins commonly utilized in solid phase synthesis to allow greater understanding o ...
19_03_05rw
... Second stage is restoration of C=O by elimination. Complicating features of each stage involve acid-base chemistry. ...
... Second stage is restoration of C=O by elimination. Complicating features of each stage involve acid-base chemistry. ...
NUCLEOPHILIC SUBSTITUTION & ELIMINATION ON Csp 3
... Rank the following SN2 rxn by expected rate; fastest=1st Identify and tag Cα in each substrate and classify it ...
... Rank the following SN2 rxn by expected rate; fastest=1st Identify and tag Cα in each substrate and classify it ...
An Overview of Carbonyl Compound Chemistry
... is applied to a reaction involving a carboxylic acid derivative, it will be most likely functioning as a catalyst. In acidic conditions, the C=O group will be protonated at first to make it more electrophilic, while in basic conditions, deprotonation will occur at first to make a nucleophile more nu ...
... is applied to a reaction involving a carboxylic acid derivative, it will be most likely functioning as a catalyst. In acidic conditions, the C=O group will be protonated at first to make it more electrophilic, while in basic conditions, deprotonation will occur at first to make a nucleophile more nu ...
Required Resources and Materials
... NB: Write down structures. Also write down explanation about the product (or lack of) Type of reaction: Alcohol oxidation Modelling: (Carbon-black; Hydrogen–white;Oxygen-Red) – model methylpropan-2-ol only but explain reaction ...
... NB: Write down structures. Also write down explanation about the product (or lack of) Type of reaction: Alcohol oxidation Modelling: (Carbon-black; Hydrogen–white;Oxygen-Red) – model methylpropan-2-ol only but explain reaction ...
Catalytic Functionalization of Methyl Group on Silicon: Iridium
... organosilicon compounds have been synthesized from methylchlorosilanes and utilized in organic and inorganic synthesis.2In these applications, the conversion of methylchlorosilanes is based on reactions at their Si−Cl bonds, which are efficiently converted into Si−O, Si−N, and Si−C bonds. In contrast, ...
... organosilicon compounds have been synthesized from methylchlorosilanes and utilized in organic and inorganic synthesis.2In these applications, the conversion of methylchlorosilanes is based on reactions at their Si−Cl bonds, which are efficiently converted into Si−O, Si−N, and Si−C bonds. In contrast, ...
Organometallics II
... formula C11H22O, are formed in the reaction of methyl lithium with 3-(R)-tertbutylcyclohexanone. These two alcohols are ...
... formula C11H22O, are formed in the reaction of methyl lithium with 3-(R)-tertbutylcyclohexanone. These two alcohols are ...
Chapter 18
... Before many spectroscopic tools were developed this was the only way to determine structure of unknowns, run reactions and systematically determine products ...
... Before many spectroscopic tools were developed this was the only way to determine structure of unknowns, run reactions and systematically determine products ...
Grignard Reagents brochure
... For direct metallation, relative reactivity of organohalides towards magnesium increases from the fluoride to the iodide. Fluorides are so unreactive that they can only be transferred to Grignard reagents under special conditions. Alkyl halides react easier with magnesium than Aryl halides. The gene ...
... For direct metallation, relative reactivity of organohalides towards magnesium increases from the fluoride to the iodide. Fluorides are so unreactive that they can only be transferred to Grignard reagents under special conditions. Alkyl halides react easier with magnesium than Aryl halides. The gene ...
Chapter 14
... and especially DO NOT distill these solvents that have been kept after long periods of time ...
... and especially DO NOT distill these solvents that have been kept after long periods of time ...
When 1°, 2°, aromatic amines or aryl amines . (Rand
... Sulphanilic acid (p-Aminobenzenesulphonic acid) is a salt of special type, called a dipolar ion* (sometimes called a Zwitterion). It is formed by the reaction of on acidic group (-S020H) and a basic group (-NH2) that are part of the same molecule, hence it is also called inner salt. It is interestin ...
... Sulphanilic acid (p-Aminobenzenesulphonic acid) is a salt of special type, called a dipolar ion* (sometimes called a Zwitterion). It is formed by the reaction of on acidic group (-S020H) and a basic group (-NH2) that are part of the same molecule, hence it is also called inner salt. It is interestin ...
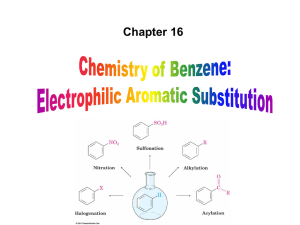
Practice Problem - HCC Southeast Commons
... intermediate loses H+ to regenerate the aromatic ring and yield a substitution product in which H+ is replaced by Br+ – It is similar to the 2nd step of an E1 reaction – The carbocation intermediate transfers a H+ to FeBr4- (from Br- and FeBr3) – This restores aromaticity (in contrast with addition ...
... intermediate loses H+ to regenerate the aromatic ring and yield a substitution product in which H+ is replaced by Br+ – It is similar to the 2nd step of an E1 reaction – The carbocation intermediate transfers a H+ to FeBr4- (from Br- and FeBr3) – This restores aromaticity (in contrast with addition ...
Document
... Addition, Substitution, and Elimination Reactions Addition reactions, substitution reactions, and elimination reactions are the three main types of organic reactions. Most organic reactions can be classified as one of these three types. Addition Reactions In an addition reaction, atoms are added to ...
... Addition, Substitution, and Elimination Reactions Addition reactions, substitution reactions, and elimination reactions are the three main types of organic reactions. Most organic reactions can be classified as one of these three types. Addition Reactions In an addition reaction, atoms are added to ...
Direct ester condensation catalyzed by bulky diarylammonium
... The ester condensation reaction is among the most fundamental organic transformations, and more environmentally benign alternative synthetic approaches to the ones currently used are in strong demand by the chemical industry1. Conventionally, the ester condensation reaction of carboxylic acids with ...
... The ester condensation reaction is among the most fundamental organic transformations, and more environmentally benign alternative synthetic approaches to the ones currently used are in strong demand by the chemical industry1. Conventionally, the ester condensation reaction of carboxylic acids with ...
f8560d95306293b
... • In order for ethers to undergo substitution or elimination reactions, their poor leaving group must first be converted into a good leaving group by reaction with strong acids such as HBr and HI. • HBr and HI are strong acids that are also sources of good nucleophiles (Br¯ and I¯ respectively). • W ...
... • In order for ethers to undergo substitution or elimination reactions, their poor leaving group must first be converted into a good leaving group by reaction with strong acids such as HBr and HI. • HBr and HI are strong acids that are also sources of good nucleophiles (Br¯ and I¯ respectively). • W ...
Normal and abnormal carbene complexes derived
... rather large and can be understood by the absence of ortho substituents in the carbene ligand in 3, thus contrasting the carbenes in 1 and 2. Structural data for unsubstituted thiazolium salts within their rings [19,20] indicate that the S–C2–N angle is wider than the S–C5–C4 angle. The normal thiaz ...
... rather large and can be understood by the absence of ortho substituents in the carbene ligand in 3, thus contrasting the carbenes in 1 and 2. Structural data for unsubstituted thiazolium salts within their rings [19,20] indicate that the S–C2–N angle is wider than the S–C5–C4 angle. The normal thiaz ...
Benzyne Mechanism
... Phenols and phenoxides are highly reactive. Only a weak catalyst (HF) required for FriedelCrafts reaction. Tribromination occurs without catalyst. Even reacts with CO2. O ...
... Phenols and phenoxides are highly reactive. Only a weak catalyst (HF) required for FriedelCrafts reaction. Tribromination occurs without catalyst. Even reacts with CO2. O ...
Elimination Reactions
... base (H2SO4, H3PO4). To favour SN1 over E1, use a good nucleophile. •To favour E2 over SN2, use a strong, bulky non-nucleophilic base. To favour SN2 over E2, use good nucleophiles that are relatively weak bases. •It is important to keep in mind that although you might choose reaction conditions that ...
... base (H2SO4, H3PO4). To favour SN1 over E1, use a good nucleophile. •To favour E2 over SN2, use a strong, bulky non-nucleophilic base. To favour SN2 over E2, use good nucleophiles that are relatively weak bases. •It is important to keep in mind that although you might choose reaction conditions that ...
Elimination Reactions
... base (H2SO4, H3PO4). To favour SN1 over E1, use a good nucleophile. •To favour E2 over SN2, use a strong, bulky non-nucleophilic base. To favour SN2 over E2, use good nucleophiles that are relatively weak bases. •It is important to keep in mind that although you might choose reaction conditions that ...
... base (H2SO4, H3PO4). To favour SN1 over E1, use a good nucleophile. •To favour E2 over SN2, use a strong, bulky non-nucleophilic base. To favour SN2 over E2, use good nucleophiles that are relatively weak bases. •It is important to keep in mind that although you might choose reaction conditions that ...
Haloalkanes and Haloarenes
... chlorofluoromethanes are gases at room temperature. Higher members are liquids or solids. As we have already learnt, molecules of organic halogen compounds are generally polar. Due to greater polarity as well as higher molecular mass as compared to the parent hydrocarbon, the intermolecular forces o ...
... chlorofluoromethanes are gases at room temperature. Higher members are liquids or solids. As we have already learnt, molecules of organic halogen compounds are generally polar. Due to greater polarity as well as higher molecular mass as compared to the parent hydrocarbon, the intermolecular forces o ...
Rhenium(VII) Catalysis of Prins Cyclization Reactions
... the reaction between isobutyraldehyde and alcohol 1. The expected product 8 is accompanied by side products 9 and 7 that arise from facile 2-oxonia-Cope rearrangements.8 The outcome with an electron-rich aromatic aldehyde is very different. Side-chain exchange products are much less prevalent, and t ...
... the reaction between isobutyraldehyde and alcohol 1. The expected product 8 is accompanied by side products 9 and 7 that arise from facile 2-oxonia-Cope rearrangements.8 The outcome with an electron-rich aromatic aldehyde is very different. Side-chain exchange products are much less prevalent, and t ...
Document
... dehydrohalogenation, the major product is the more stable stereoisomer. • A reaction is stereoselective when it forms predominantly or exclusively one stereoisomer when two or more are possible. • The E2 reaction is stereoselective because one stereoisomer is formed preferentially. Why? ...
... dehydrohalogenation, the major product is the more stable stereoisomer. • A reaction is stereoselective when it forms predominantly or exclusively one stereoisomer when two or more are possible. • The E2 reaction is stereoselective because one stereoisomer is formed preferentially. Why? ...
Stille reaction

The Stille reaction, or the Migita-Kosugi-Stille coupling, is a chemical reaction widely used in organic synthesis which involves the coupling of an organotin compound (also known as organostannanes) with a variety of organic electrophiles via palladium-catalyzed coupling reaction.The R1 group attached to the trialkyltin is normally sp2-hybridized, including alkenes, and aryl groups; however, conditions have been devised to incorporate both sp3-hybridized groups, such as allylic and benzylic substituents, and sp-hybridized alkynes. These organostannanes are also stable to both air and moisture, and many of these reagents are either commercially available or can be synthesized from literature precedent. However, these tin reagents tend to be highly toxic. X is typically a halide, such as Cl, Br, I, yet pseudohalides such as triflates and sulfonates and phosphates can also be used.The groundwork for the Stille reaction was laid by Colin Eaborn, Toshihiko Migita, and Masanori Kosugi in 1976 and 1977, who explored numerous palladium catalyzed couplings involving organotin reagents. John Stille and David Milstein developed a much milder and more broadly applicable procedure in 1978. Stille’s work on this area might have earned him a share of the 2010 Nobel Prize, which was awarded to Richard Heck, Ei-ichi Negishi, and Akira Suzuki for their work on the Heck, Negishi, and Suzuki coupling reactions. However, Stille died in the plane crash of United Airlines Flight 232 in 1989.Several reviews have been published on the Stille reaction.