major in Chemistry
... matter, measurements, calculations, elements, atomic theory, atomic mass, the mole, ionic and molecular compounds, types of bonding, mole calculations, types of reactions, limiting reagents, percent yield, solutions, gases, quantum mechanics, orbitals and electrons, electronic structure, atomic peri ...
... matter, measurements, calculations, elements, atomic theory, atomic mass, the mole, ionic and molecular compounds, types of bonding, mole calculations, types of reactions, limiting reagents, percent yield, solutions, gases, quantum mechanics, orbitals and electrons, electronic structure, atomic peri ...
Theories in the Evolution of Chemical Equilibrium: Impli
... pressure equal amounts of molecules decompose and unite by collision. That explanation required that not all molecules were in the same state of motion at a given temperature. That is, it was assumed that some of the molecules regularly diverged more or less widely from the average state, for only a ...
... pressure equal amounts of molecules decompose and unite by collision. That explanation required that not all molecules were in the same state of motion at a given temperature. That is, it was assumed that some of the molecules regularly diverged more or less widely from the average state, for only a ...
CHemiStrY - Cabrillo College
... Repeatability: May be taken a total of 1 time. Presents a one semester survey of general and organic chemistry as preparation for careers in the allied health sciences. It is not appropriate for premed, dental or veterinary students nor is it intended for allied health students requiring two semeste ...
... Repeatability: May be taken a total of 1 time. Presents a one semester survey of general and organic chemistry as preparation for careers in the allied health sciences. It is not appropriate for premed, dental or veterinary students nor is it intended for allied health students requiring two semeste ...
1 - Cathedral High School
... 3.2.1 Describe and explain the periodic trends in atomic radii, ionic radii, ionization energies, electronegativity and melting points for the alkali metals (Li Cs), halogens (F I) and period 3 elements (Na Ar). Cross reference with topics 2, 4 and 5. Data for all these properties are listed i ...
... 3.2.1 Describe and explain the periodic trends in atomic radii, ionic radii, ionization energies, electronegativity and melting points for the alkali metals (Li Cs), halogens (F I) and period 3 elements (Na Ar). Cross reference with topics 2, 4 and 5. Data for all these properties are listed i ...
Deans Community High School Intermediate 2 Revision Notes www
... A change in energy can also be shown by light being produced or a noise, e.g. an explosion. Reactions that give energy out are called EXOTHERMIC reactions. Reactions that take energy in are called ENDOTHERMIC reactions. ...
... A change in energy can also be shown by light being produced or a noise, e.g. an explosion. Reactions that give energy out are called EXOTHERMIC reactions. Reactions that take energy in are called ENDOTHERMIC reactions. ...
Chapter 7
... Break the problem into two steps: Raise the temperature of the liquid first then completely vaporize it. The total enthalpy change is the sum of the changes in each step. ...
... Break the problem into two steps: Raise the temperature of the liquid first then completely vaporize it. The total enthalpy change is the sum of the changes in each step. ...
Chapter 17: An Introduction to Organic Chemistry, Biochemistry, and
... into the millions. Fortunately, the task of studying them is not so daunting as their number would suggest, because organic compounds can be categorized according to structural similarities that lead to similarities in the compounds’ important properties. For example, you discovered in Section 3.3 t ...
... into the millions. Fortunately, the task of studying them is not so daunting as their number would suggest, because organic compounds can be categorized according to structural similarities that lead to similarities in the compounds’ important properties. For example, you discovered in Section 3.3 t ...
Chapter 7: Solutions
... • In all precipitation reactions, the reactants are always aqueous. • Use the Solubility Rules Table to determine the phase of the “possible” products. • If a compound is water soluble, it remains dissolved and we write “(aq).” • If a compound is water insoluble, it precipitates as a solid and w ...
... • In all precipitation reactions, the reactants are always aqueous. • Use the Solubility Rules Table to determine the phase of the “possible” products. • If a compound is water soluble, it remains dissolved and we write “(aq).” • If a compound is water insoluble, it precipitates as a solid and w ...
SIDE GROUP ADDITION TO THE POLYCYCLIC AROMATIC
... to previously reported alcohol (”OH) and ketone (>C»O) formation. This work represents the first experimental evidence that ice photochemistry may have contributed to the aromatics bearing carbon and nitrogen containing side groups that are detected in primitive meteorites and interplanetary dust par ...
... to previously reported alcohol (”OH) and ketone (>C»O) formation. This work represents the first experimental evidence that ice photochemistry may have contributed to the aromatics bearing carbon and nitrogen containing side groups that are detected in primitive meteorites and interplanetary dust par ...
Competition for Electrons
... track of electrons based on the arbitrary assumption that shared electrons belong to the more electronegative element n Rules for assigning oxidation numbers q Oxidation numbers for atoms that are free elements are always zero q The oxidation numbers of ions are the same as the charge on the ion q S ...
... track of electrons based on the arbitrary assumption that shared electrons belong to the more electronegative element n Rules for assigning oxidation numbers q Oxidation numbers for atoms that are free elements are always zero q The oxidation numbers of ions are the same as the charge on the ion q S ...
Materials Chemistry Prof. S. Sunder Manoharan Department of
... is easier for to make many materials. Therefore, these are grouped under 1 direct reaction of solids precursor methods, or wet combustion synthesis by this you can actually try to solids in bulk. And another group of compounds that you can make is through vapor transport synthesis, which is not a ve ...
... is easier for to make many materials. Therefore, these are grouped under 1 direct reaction of solids precursor methods, or wet combustion synthesis by this you can actually try to solids in bulk. And another group of compounds that you can make is through vapor transport synthesis, which is not a ve ...
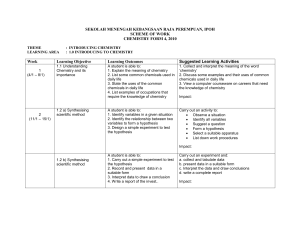
Comparison of 2008 to 2000 SCH3U_ud
... Sample issue: Base metal smelting produces useful metals such as zinc, lead, copper, and nickel directly from their ores. However, during smelting, harmful compounds can be released into the environment, including cadmium, arsenic, sulphur dioxide, and mercury, all of which can endanger the health a ...
... Sample issue: Base metal smelting produces useful metals such as zinc, lead, copper, and nickel directly from their ores. However, during smelting, harmful compounds can be released into the environment, including cadmium, arsenic, sulphur dioxide, and mercury, all of which can endanger the health a ...
physical setting chemistry
... Base your answers to questions 66 through 68 on the information below and on your knowledge of chemistry. Carbon dioxide, CO2, changes from the solid phase to the gas phase at 1 atm and 194.5 K. In the solid phase, CO2 is often called dry ice. When dry ice sublimes in air at 298 K, the water vapor i ...
... Base your answers to questions 66 through 68 on the information below and on your knowledge of chemistry. Carbon dioxide, CO2, changes from the solid phase to the gas phase at 1 atm and 194.5 K. In the solid phase, CO2 is often called dry ice. When dry ice sublimes in air at 298 K, the water vapor i ...
CHEM 1405 Practice Exam #2 (2015)
... 2) A)Cu, because it is not as active as Ni B) 1) [He] 2s2 2p4 2) [Ne] 3s2 3) A) Calcium Hydroxide B) Potassium Chloride C) Aluminum Bromide D) Sodium Fluoride ...
... 2) A)Cu, because it is not as active as Ni B) 1) [He] 2s2 2p4 2) [Ne] 3s2 3) A) Calcium Hydroxide B) Potassium Chloride C) Aluminum Bromide D) Sodium Fluoride ...
Section 5
... will depend on the polarity of the H-X bond (in most Brønsted acids, X = N, O, or a halogen) Electron-withdrawing groups attached to X will increase the quantity of partial positive charge on the H-atom, making it more susceptible to nucleophilic attack by a solvent (inductive effect) O ...
... will depend on the polarity of the H-X bond (in most Brønsted acids, X = N, O, or a halogen) Electron-withdrawing groups attached to X will increase the quantity of partial positive charge on the H-atom, making it more susceptible to nucleophilic attack by a solvent (inductive effect) O ...
Jon Abbatt - Earth, Atmospheric, and Planetary Physics
... Although sulfate aerosols all contain the SO42- ion, they are a highly heterogeneous mix of aerosol types. Formed from the oxidation of more chemically reduced sulfur compounds. Present in the nucleation and accumulation modes. Over the continents, the major sulfur source is SO2, formed from ...
... Although sulfate aerosols all contain the SO42- ion, they are a highly heterogeneous mix of aerosol types. Formed from the oxidation of more chemically reduced sulfur compounds. Present in the nucleation and accumulation modes. Over the continents, the major sulfur source is SO2, formed from ...
SEKOLAH MENENGAH KEBANGSAAN RAJA PEREMPUAN, IPOH
... 5. state the safety precautions when handling Group 1 elements ...
... 5. state the safety precautions when handling Group 1 elements ...
Appendix N CONCENTRATION UNITS
... in alloys. Certain generic names such as "stainless steel" imply the presence of certain minimum levels of different components in the steel. In this case, the definitions are not legal ones, but are standard compositions accepted by ...
... in alloys. Certain generic names such as "stainless steel" imply the presence of certain minimum levels of different components in the steel. In this case, the definitions are not legal ones, but are standard compositions accepted by ...
1 Mole
... Standard 3a: describing chemical reactions An equation describes a chemical reaction or ...
... Standard 3a: describing chemical reactions An equation describes a chemical reaction or ...
Inorganic chemistry

Inorganic chemistry deals with the synthesis and behavior of inorganic and organometallic compounds. This field covers all chemical compounds except the myriad organic compounds (carbon based compounds, usually containing C-H bonds), which are the subjects of organic chemistry. The distinction between the two disciplines is far from absolute, and there is much overlap, most importantly in the sub-discipline of organometallic chemistry. It has applications in every aspect of the chemical industry–including catalysis, materials science, pigments, surfactants, coatings, medicine, fuel, and agriculture.