Chapter 9 - Ionic and Covalent Bonding 1. In which pair do both
... E) that bonding electrons are simultaneously attracted by more than one nucleus. ANS: D PTS: 1 DIF: moderate REF: 9.4 OBJ: Describe the formation of a covalent bond between two atoms. TOP: bonding | covalent bonding MSC: general chemistry 53. During the formation of a chemical bond between two hydro ...
... E) that bonding electrons are simultaneously attracted by more than one nucleus. ANS: D PTS: 1 DIF: moderate REF: 9.4 OBJ: Describe the formation of a covalent bond between two atoms. TOP: bonding | covalent bonding MSC: general chemistry 53. During the formation of a chemical bond between two hydro ...
Full-Text PDF
... and organic spacers in a complementary way. The inorganic component imparts magnetism, mechanical strength and thermal stability, while the organic part offers a way to improved luminescence, structural diversifications, and processability [13–16]. The effort exerted in this field promotes the under ...
... and organic spacers in a complementary way. The inorganic component imparts magnetism, mechanical strength and thermal stability, while the organic part offers a way to improved luminescence, structural diversifications, and processability [13–16]. The effort exerted in this field promotes the under ...
Natural Bond Orbital Analysis of [Fe(H2O)6]2+/3+ and N=0-4
... Metals are found in different forms in nature. One of the major forms is a molecule resulted from the coordination of metals with other molecules or ions. Such associations are often referred as coordination complexes [1]. These coordinated complexes play very important roles in the physiological ac ...
... Metals are found in different forms in nature. One of the major forms is a molecule resulted from the coordination of metals with other molecules or ions. Such associations are often referred as coordination complexes [1]. These coordinated complexes play very important roles in the physiological ac ...
Recent advances in understanding blue copper proteins
... as Pc and its function in tuning E0 into a physiological range for ET. 2. Active site ligand and metal perturbations 2.1. Ligand perturbations: S → Se T1 or BC proteins are now well understood from a geometric and electronic structure correlation perspective. A large majority of this understanding h ...
... as Pc and its function in tuning E0 into a physiological range for ET. 2. Active site ligand and metal perturbations 2.1. Ligand perturbations: S → Se T1 or BC proteins are now well understood from a geometric and electronic structure correlation perspective. A large majority of this understanding h ...
Coordination Compounds Coordination Compounds
... (ii) The ligands are then listed in alphabetical order. The placement of a ligand in the list does not depend on its charge. (iii) Polydentate ligands are also listed alphabetically. In case of abbreviated ligand, the first letter of the abbreviation is used to determine the position of the ligand i ...
... (ii) The ligands are then listed in alphabetical order. The placement of a ligand in the list does not depend on its charge. (iii) Polydentate ligands are also listed alphabetically. In case of abbreviated ligand, the first letter of the abbreviation is used to determine the position of the ligand i ...
The SimSoup Guide - Chris Gordon
... of the chemical processes in which they can participate. Although the conceptual model is an abstraction, it is intended to be realistic; much of the material that follows will be familiar to physical chemists. See for example Atkins and Paula (2006), Atkins (2001), and Pauling (1960). ...
... of the chemical processes in which they can participate. Although the conceptual model is an abstraction, it is intended to be realistic; much of the material that follows will be familiar to physical chemists. See for example Atkins and Paula (2006), Atkins (2001), and Pauling (1960). ...
http://doc.rero.ch
... plane compared to this direction, the counter anions lying only on one side of the chains. Indeed the coordination of the nitrate molecules is not distributed homogeneously around the silver atoms but they are found only on one side (Fig. 9b). In the 3-D structure of 2, the layers stack parallel and ...
... plane compared to this direction, the counter anions lying only on one side of the chains. Indeed the coordination of the nitrate molecules is not distributed homogeneously around the silver atoms but they are found only on one side (Fig. 9b). In the 3-D structure of 2, the layers stack parallel and ...
TERMS AND DEFINITIONS IN THERMOCHEMISTRY
... Note that the value for the C=C double bond is nearly twice that for the single CnC bond, and the value for the C/C bond nearly three times as much. This is what one would expect. Using these mean bond energies it is possible to estimate the enthalpy change for a reaction which cannot be studied in ...
... Note that the value for the C=C double bond is nearly twice that for the single CnC bond, and the value for the C/C bond nearly three times as much. This is what one would expect. Using these mean bond energies it is possible to estimate the enthalpy change for a reaction which cannot be studied in ...
support material
... According to this law equal volumes of gases at the same temperature and pressure should contain equal number of molecules. Dalton's Atomic Theory All substances are made up of tiny, indivisible particles called atoms. Atoms of the same element are identical in shape, size, mass and other properties ...
... According to this law equal volumes of gases at the same temperature and pressure should contain equal number of molecules. Dalton's Atomic Theory All substances are made up of tiny, indivisible particles called atoms. Atoms of the same element are identical in shape, size, mass and other properties ...
Document
... Since the MOs are irreducible representations of the molecule’s pointgroup, only certain atomic orbitals can interact together to form MOs with given bonding (or antibonding) character. The extent of the interaction between the atomic orbitals depends on the physical overlap as well as on their rela ...
... Since the MOs are irreducible representations of the molecule’s pointgroup, only certain atomic orbitals can interact together to form MOs with given bonding (or antibonding) character. The extent of the interaction between the atomic orbitals depends on the physical overlap as well as on their rela ...
M - Chemistry
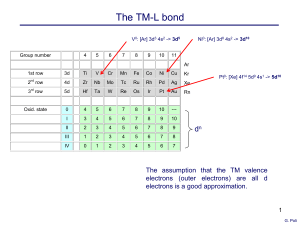
... the presence of the five d orbitals which can hold 10 more electrons relative to C, O, N, etc. Electron counting is the process of determining the number of valence electrons about a metal center in a given transition metal complex. Unfortunately, there are two methods of electron counting, the ioni ...
... the presence of the five d orbitals which can hold 10 more electrons relative to C, O, N, etc. Electron counting is the process of determining the number of valence electrons about a metal center in a given transition metal complex. Unfortunately, there are two methods of electron counting, the ioni ...
1 - American Chemical Society
... From this we conclude that the excited state is not delocalized over the Cu, unit or the aromatic rings. Murphy et al. have recently reported rmm temperature luminescence from tetrametallic complexes of ruthenium." They ascribed this to localization of the MLCT transition involved. This runs paralle ...
... From this we conclude that the excited state is not delocalized over the Cu, unit or the aromatic rings. Murphy et al. have recently reported rmm temperature luminescence from tetrametallic complexes of ruthenium." They ascribed this to localization of the MLCT transition involved. This runs paralle ...
NOBLE-GAS CHEMISTRY
... adducts of noble gases to a metal center, NgBe(II)O, where Ng = Ar, Kr, and Xe.31 The case of XeBeO is rather straightforward: a coordinatively unsaturated Be(II) cation exposes its empty (sp) hybrid, and is ready to bind whatever Lewis base you provide. Hence, it will bind a Xe atom with a surprisi ...
... adducts of noble gases to a metal center, NgBe(II)O, where Ng = Ar, Kr, and Xe.31 The case of XeBeO is rather straightforward: a coordinatively unsaturated Be(II) cation exposes its empty (sp) hybrid, and is ready to bind whatever Lewis base you provide. Hence, it will bind a Xe atom with a surprisi ...
Lecture 4- LIGANDS
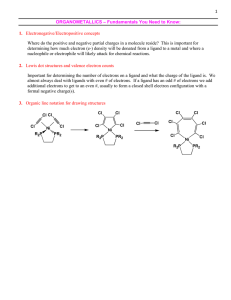
... CO groups have a high tendency to stabilize M−M bonds; not only are CO ligands relatively small but they also leave the metal atom with a net charge similar to that in its elemental form (electroneutrality principle). “Stable complexes are those with structures such that each atom has only a small e ...
... CO groups have a high tendency to stabilize M−M bonds; not only are CO ligands relatively small but they also leave the metal atom with a net charge similar to that in its elemental form (electroneutrality principle). “Stable complexes are those with structures such that each atom has only a small e ...
Hybridization of atomic orbitals
... Atomic orbitals are (energy) states or wave forms of electrons in the atom. If we insist on the particle nature of electrons, then the probability of finding an electron in an atomic orbital is proportional to the square of the wavefunction. The values of the wavefunction can be either positive or n ...
... Atomic orbitals are (energy) states or wave forms of electrons in the atom. If we insist on the particle nature of electrons, then the probability of finding an electron in an atomic orbital is proportional to the square of the wavefunction. The values of the wavefunction can be either positive or n ...
p-BLOCK ELEMENTS - einstein classes
... H3BO3 or (B(OH)3) cannot be titrated satisfactory with NaOH, as a sharp end point is not obtained. If certain organic polyhydroxy compounds such as glycerol, mannitol or sugars are added to the titration mixture, then B(OH)3 behaves as a strong monobasic acid. It can now be titrated with NaOH, and t ...
... H3BO3 or (B(OH)3) cannot be titrated satisfactory with NaOH, as a sharp end point is not obtained. If certain organic polyhydroxy compounds such as glycerol, mannitol or sugars are added to the titration mixture, then B(OH)3 behaves as a strong monobasic acid. It can now be titrated with NaOH, and t ...
co-ordination compounds
... Valence bond theory, primarily the work of Linus Pauling regarded bonding as characterized by the overlap of atomic or hybrid orbitals of individual atoms. The postulates of valence bond theory: 1) The central metal atom/ion makes available a number of vacant orbitals equal to its coordination numbe ...
... Valence bond theory, primarily the work of Linus Pauling regarded bonding as characterized by the overlap of atomic or hybrid orbitals of individual atoms. The postulates of valence bond theory: 1) The central metal atom/ion makes available a number of vacant orbitals equal to its coordination numbe ...
glossary of terms used in theoretical organic chemistry
... The Glossary may be considered as a supplement to the comprehensive compendium [38] of terminology traditionally established in physical organic chemistry. Therefore, the styles of the presentation of the material in both Glossaries are basically similar. The general criteria adopted for inclusion o ...
... The Glossary may be considered as a supplement to the comprehensive compendium [38] of terminology traditionally established in physical organic chemistry. Therefore, the styles of the presentation of the material in both Glossaries are basically similar. The general criteria adopted for inclusion o ...
tris(ethane-1,2–diammine)cobalt(III) sulphate
... In modern formulations, such spatial arrangements are called coordination polyhedra. The species within the square bracket are coordination entities or complexes and the ions outside the square bracket are called counter ions. He further postulated that octahedral, tetrahedral and square planar geom ...
... In modern formulations, such spatial arrangements are called coordination polyhedra. The species within the square bracket are coordination entities or complexes and the ions outside the square bracket are called counter ions. He further postulated that octahedral, tetrahedral and square planar geom ...
A QUALITATIVE ?C - MOLECULAR ORBITAL RATIONALE FOR H
... Simplified n-MO schemes for trans-[MLL'P 4 ] with other ligands L and L' which are isoelectronic with isonitriles [P 4 may stand for (dppe) 2 or other diphosphine, diarsine, mixed phosphine-arsine or monophosphine ligands][18], may be derived in a similar way as for the bisisonitrile complexes. Henc ...
... Simplified n-MO schemes for trans-[MLL'P 4 ] with other ligands L and L' which are isoelectronic with isonitriles [P 4 may stand for (dppe) 2 or other diphosphine, diarsine, mixed phosphine-arsine or monophosphine ligands][18], may be derived in a similar way as for the bisisonitrile complexes. Henc ...
Formation, photodissociation, and structure studies of group 14(Si
... Group 14(Si, Ge, Sn, and Pb)/P binary cluster ions have been produced by laser ablation, photodissociated by UV laser and detected with a tandem time-of-flight mass spectrometer. As expected on the double periodicities of group 14 elements, Si/P and Ge/P binary cluster ions, whereas Sn/P and Pb/P bi ...
... Group 14(Si, Ge, Sn, and Pb)/P binary cluster ions have been produced by laser ablation, photodissociated by UV laser and detected with a tandem time-of-flight mass spectrometer. As expected on the double periodicities of group 14 elements, Si/P and Ge/P binary cluster ions, whereas Sn/P and Pb/P bi ...
chm 205 - National Open University of Nigeria
... allotropic forms of carbon, viz., diamond and graphite are well-known. These are, in fact, giant macromolecules consisting of C atoms linked by a network of covalent bonds (Figs, 1.l and 1.2). Each carbon in diamond is tetrahedrally bonded to four other carbons with a bond distance of 154 pm. Here y ...
... allotropic forms of carbon, viz., diamond and graphite are well-known. These are, in fact, giant macromolecules consisting of C atoms linked by a network of covalent bonds (Figs, 1.l and 1.2). Each carbon in diamond is tetrahedrally bonded to four other carbons with a bond distance of 154 pm. Here y ...
CO ORDINATION COMPOUNDS
... up into its constituent ions in the dissolved state. These compounds exhibit individual properties of their constituents. For e.g.FeSO4.(NH4)2SO4.6H2Obreaks into Fe2+, NH4+, and SO42− ions. Hence, it gives a positive test for Fe2+ ions. A coordination compound is an addition compound which retains i ...
... up into its constituent ions in the dissolved state. These compounds exhibit individual properties of their constituents. For e.g.FeSO4.(NH4)2SO4.6H2Obreaks into Fe2+, NH4+, and SO42− ions. Hence, it gives a positive test for Fe2+ ions. A coordination compound is an addition compound which retains i ...
Lecture 1: Key Concepts in Stereoselective Synthesis
... Possible solution(s): Use of transition metal catalysts to lower the activation barrier - Chemoselectivity: Once the desired C-X bond is formed, this bond itself has a lower bond strength than the C-H bond before, and over-reactions such as catalyst inhibition can occur. Secondly, the introduction o ...
... Possible solution(s): Use of transition metal catalysts to lower the activation barrier - Chemoselectivity: Once the desired C-X bond is formed, this bond itself has a lower bond strength than the C-H bond before, and over-reactions such as catalyst inhibition can occur. Secondly, the introduction o ...
Novel Class of Heterometallic Cubane and Boride Clusters
... geometry of 5 can be viewed as cubane, made of four Mo and four B atoms, and out of the six square faces of the cube three of them are capped by BH units to build a [Mo4B7] core. Alternatively, the observed geometry of 5 can be viewed as a tetracapped tetrahedron in which four molybdenum atoms define ...
... geometry of 5 can be viewed as cubane, made of four Mo and four B atoms, and out of the six square faces of the cube three of them are capped by BH units to build a [Mo4B7] core. Alternatively, the observed geometry of 5 can be viewed as a tetracapped tetrahedron in which four molybdenum atoms define ...

![Natural Bond Orbital Analysis of [Fe(H2O)6]2+/3+ and N=0-4](http://s1.studyres.com/store/data/000268544_1-fea270dbb1c63563f568c2f83f2717db-300x300.png)