Heat and Thermodynamics
... necessary. For example, a system could be as simple as a bicycle pump or as complex as the CANDU reactor itself. Parameters such as volume, temperature, pressure, internal energy, enthalpy, and entropy are used to define the state of a system. We will see how parameter changes impact our thermodynam ...
... necessary. For example, a system could be as simple as a bicycle pump or as complex as the CANDU reactor itself. Parameters such as volume, temperature, pressure, internal energy, enthalpy, and entropy are used to define the state of a system. We will see how parameter changes impact our thermodynam ...
Entropy generation minimization of one and two
... coefficient causes a temperature increase of hydrogen when subjected to a forced adiabatic expansion, as in the APRR-valve. The dominating contribution to the tank heating is the heat of compression[3] and the conversion of kinetic to internal energy[1][2]. The refuelling duration can be reduced wit ...
... coefficient causes a temperature increase of hydrogen when subjected to a forced adiabatic expansion, as in the APRR-valve. The dominating contribution to the tank heating is the heat of compression[3] and the conversion of kinetic to internal energy[1][2]. The refuelling duration can be reduced wit ...
Document
... between the system and surroundings Heat exchange occurs when system and surroundings have a difference in temperature Temperature is the measure of the amount of thermal energy within a sample of matter Heat flows from matter with high temperature to matter with low temperature until both objects r ...
... between the system and surroundings Heat exchange occurs when system and surroundings have a difference in temperature Temperature is the measure of the amount of thermal energy within a sample of matter Heat flows from matter with high temperature to matter with low temperature until both objects r ...
View/Open
... know that the amount of heat absorbed or evolved at constant temperature and pressure is called enthalpy. Therefore the amount of heat change during a reaction at constant temperature and pressure may also be called enthalpy change. Its value depends upon the number of moles of the reactants which h ...
... know that the amount of heat absorbed or evolved at constant temperature and pressure is called enthalpy. Therefore the amount of heat change during a reaction at constant temperature and pressure may also be called enthalpy change. Its value depends upon the number of moles of the reactants which h ...
book - University of Guelph Physics
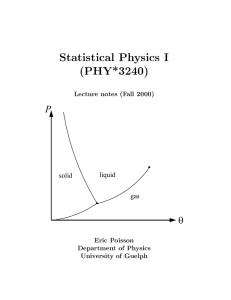
... We consider two thermodynamic systems, A and B, plus a third C, which will serve as our reference system. For concreteness, although this is not necessary for the discussion, we shall suppose that all three systems consist of fluids, so that P and V can be used as thermodynamic variables. (We could ...
... We consider two thermodynamic systems, A and B, plus a third C, which will serve as our reference system. For concreteness, although this is not necessary for the discussion, we shall suppose that all three systems consist of fluids, so that P and V can be used as thermodynamic variables. (We could ...
Chapter 16: Energy and Chemical Change
... heat. The plant is kept warm even when the temperature of the air drops to 0°C. Calculating heat evolved and absorbed If you’ve taken an early morning dive into a swimming pool similar to the one in Figure 16-3, you know that the water might be cold at that time of the day. Later in the day, especi ...
... heat. The plant is kept warm even when the temperature of the air drops to 0°C. Calculating heat evolved and absorbed If you’ve taken an early morning dive into a swimming pool similar to the one in Figure 16-3, you know that the water might be cold at that time of the day. Later in the day, especi ...
Episodic metamorphism and deformation in low
... the melting of the lower crust, with a melting interval between a solidus temperature Tsol = 700 oC and a liquidus temperature Tliq = 1300 oC, with a linear relation between melt volume and temperature in this interval. Although such a relation is not likely to hold for any specific bulk composition ...
... the melting of the lower crust, with a melting interval between a solidus temperature Tsol = 700 oC and a liquidus temperature Tliq = 1300 oC, with a linear relation between melt volume and temperature in this interval. Although such a relation is not likely to hold for any specific bulk composition ...
6 The ocean lithosphere
... Equation 6.3, derived entirely from theoretical considerations, is in excellent agreement with observed bathymetry of ocean lithosphere younger than about 80 Ma. Indeed, this remarkable agreement between observations and the age-heatflow-bathymetry relationships predicted by Equations 6.1 - 6.3 prov ...
... Equation 6.3, derived entirely from theoretical considerations, is in excellent agreement with observed bathymetry of ocean lithosphere younger than about 80 Ma. Indeed, this remarkable agreement between observations and the age-heatflow-bathymetry relationships predicted by Equations 6.1 - 6.3 prov ...
work is done - Portal UniMAP
... •Most reactions are not done in sealed containers: they are carried out in open vessels at constant pressure, with the volume capable of changing freely, especially if the reactants or products of the reaction involve gases. • In these cases, ΔV ≠ 0, and the energy change may be due to both heat tra ...
... •Most reactions are not done in sealed containers: they are carried out in open vessels at constant pressure, with the volume capable of changing freely, especially if the reactants or products of the reaction involve gases. • In these cases, ΔV ≠ 0, and the energy change may be due to both heat tra ...
Document
... Plan: We will use Hess’s law, summing the three equations or their reverses and multiplying each by an appropriate coefficient so that they add to give the net equation for the reaction of interest. At the same time, we keep track of the ΔH values, reversing their signs if the reactions are reversed ...
... Plan: We will use Hess’s law, summing the three equations or their reverses and multiplying each by an appropriate coefficient so that they add to give the net equation for the reaction of interest. At the same time, we keep track of the ΔH values, reversing their signs if the reactions are reversed ...
Chapter 06
... (Enthalpy is a state function. It doesn’t matter how you get there, only where you start and end.) Copyright©2000 by Houghton Mifflin Company. All rights reserved. ...
... (Enthalpy is a state function. It doesn’t matter how you get there, only where you start and end.) Copyright©2000 by Houghton Mifflin Company. All rights reserved. ...
Heat transfer

Heat transfer is the exchange of thermal energy between physical systems, depending on the temperature and pressure, by dissipating heat. The fundamental modes of heat transfer are conduction or diffusion, convection and radiation.Heat transfer always occurs from a region of high temperature to another region of lower temperature. Heat transfer changes the internal energy of both systems involved according to the First Law of Thermodynamics. The Second Law of Thermodynamics defines the concept of thermodynamic entropy, by measurable heat transfer.Thermal equilibrium is reached when all involved bodies and the surroundings reach the same temperature. Thermal expansion is the tendency of matter to change in volume in response to a change in temperature.